Team:EPF-Lausanne/Results/Mutations
From 2009.igem.org
(→ILE427 to PHE) |
(→ILE427 to PHE) |
||
| Line 77: | Line 77: | ||
The calculated ratios are: | The calculated ratios are: | ||
| + | |||
% of time the cystein is in the IN conformation = 55%. | % of time the cystein is in the IN conformation = 55%. | ||
Revision as of 15:22, 21 October 2009




Motivation
Cys450 side chain is involved in light state in bond formation with the cofactor. Cys450 can assume two conformational states, called here ON and OFF, and corresponding respectively, to the Sg being near or far from FMN.
The more populated conformer is the OFF, as showed by X-ray structures (OFF/ON=90/10 at low temperature and =70/30 at room temperature) but for the bond to form the Cys should switch the less favoured ON. The ON-OFF equilibrium is believed to be influenced by nearby residues. Moreover the steric hindrance of some nearby apolar residues is thought to have an effect also in the process of bond splitting. By looking at the structure and by examining previous mutation analysis we hypothesized that mutation favouring the OFF conformation of Cys450 in the dark state would likely result in favouring the splitting of the covalent adduct. On the other way round a strategy to increase the lifetime of covalent adduct could consist in mutating residue as to stabilize the ON conformation.
Since our simulation are capable of reproducing the experimental OFF/ON conformational equilibrium in the dark state (about 70/30) we decided to perform in-silico mutations, and to compute the OFF/ON ratios of mutants from MD simulations, as to have a guide to experimentally perform the mutations leading to lower OFF/ON ratios. Of course the OFF/ON conformational equilibrium is only indirectly connected to the energy barrier of the bond splitting, but some experimental evidences suggest a link between Cys conformational propensity and the splitting reaction.
We planned to perform QM calculations as to directly study the effect of specific mutations on the splitting reaction rate, but since those calcualtions require much more time we have at our disposal, we used for our mutations only chemical intuition and the results of the simulations of mutated systems.
Active site
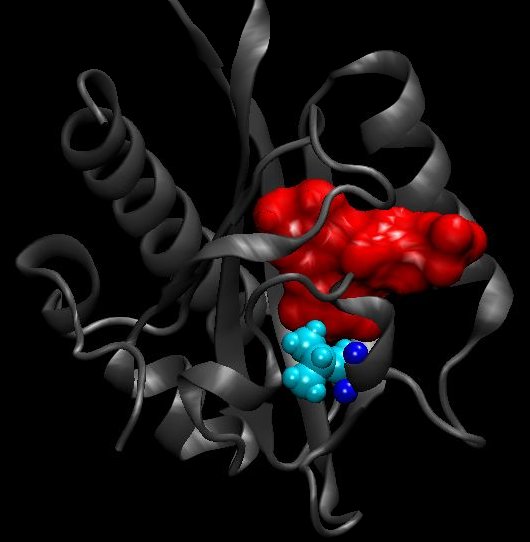
The selected residues for mutations are ILE427 in purple on the next video, LEU453 in orange and VAL416 in yellow.
Another video using the surf function shows better the steric interactions between those residues. The residues have the same color as before, but we added the CYS450 in blue.
ILE427 to PHE
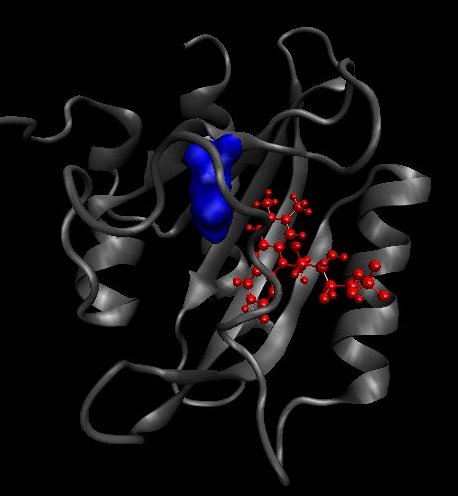
The isoleucine 427 is quite big. We tried in silico to change it to a cystein. As the cystein is a small residue that could lead some missfolding in vivo, we also planned to change it to valine in vivo. On the left, the ILE427 is colored in blue and on the right picture, this is the position of the protein having a mutation I427F after 35ns of simulation.
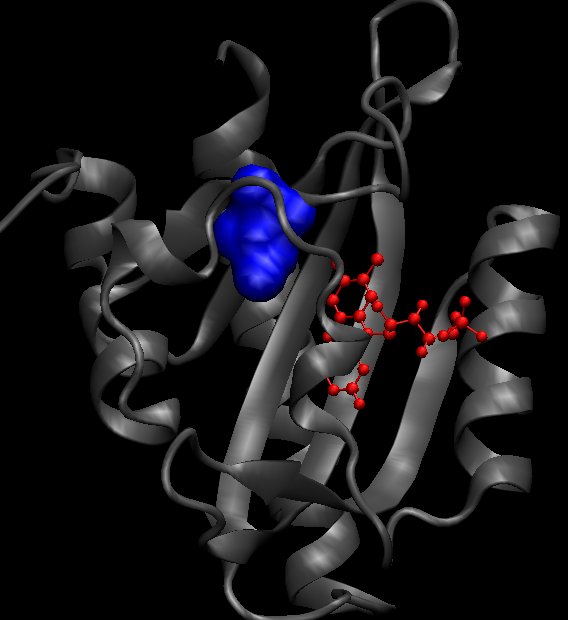
| 
|
|---|
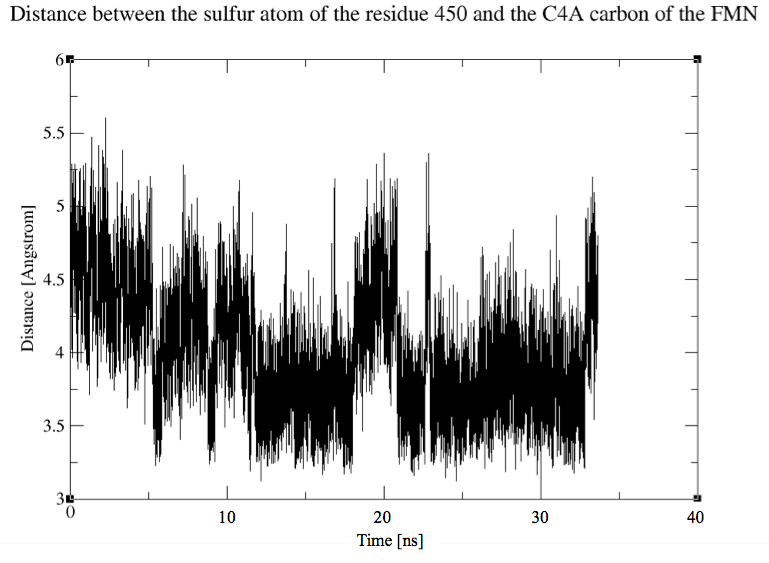
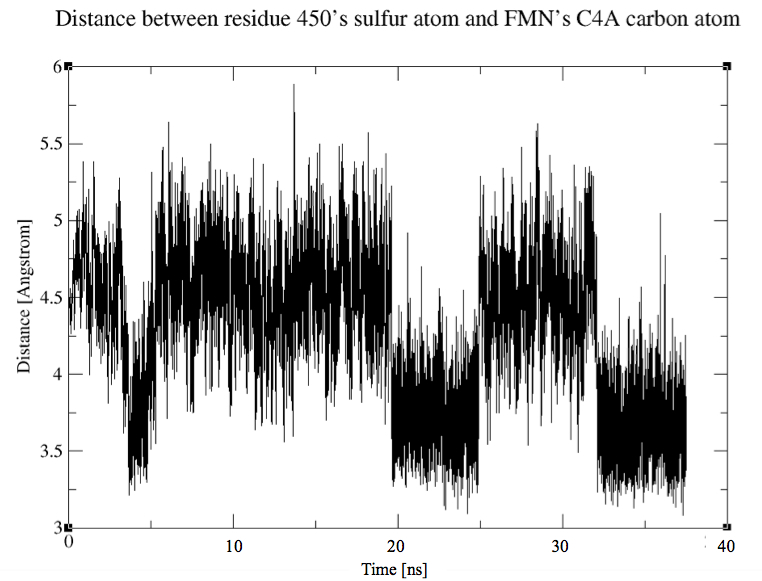
After 35 ns of simulation, we already see a clear difference in the ON/OFF ratio see the comparison with the wild type dark state result.
The calculated ratios are:
% of time the cystein is in the IN conformation = 55%.
% of time the cystein is in the OUT conformation = 45%.
These results are very encouraging because it proves that the cystein is "pushed" in the direction of the FMN by the steric interaction of the phenylalanine with the csytein.
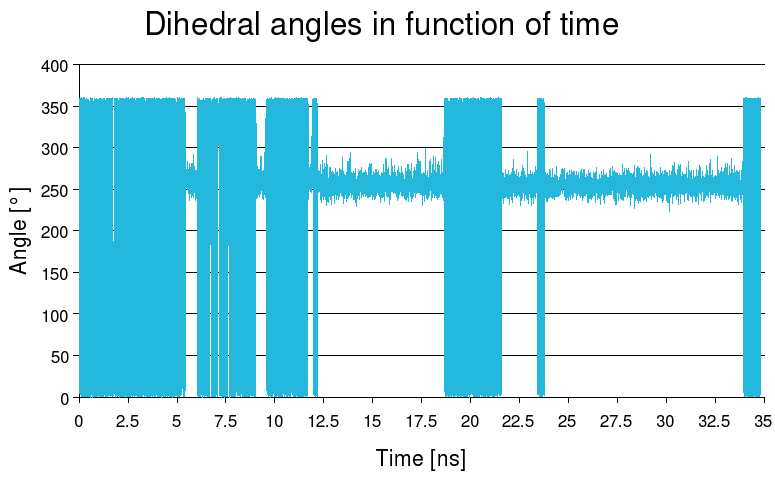
Here is a plot of the evolution of dihedral angle during the simulation:
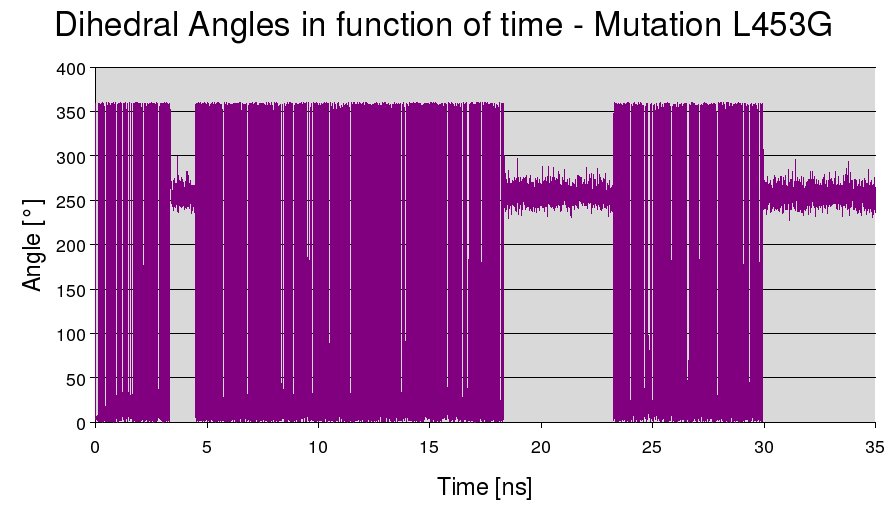
LEU453 to GLY
The light blue is the isoleucine and the dark blue is the new cystein, which is really much smaller, is in dark blue. This mutation gave good results in previous articles.
Here is the plot of the dihedral angles :
%IN = 31.5% %OUT = 68.5%
VAL416 to ILE
 "
"