Team:IPN-UNAM-Mexico/Notebook/August
From 2009.igem.org
August
03-Agust-2009
Results of previous transformations:
| F1610 | Worked | |
| K091146 | Worked | |
| K081016 | Worked | |
| K081009 | Didn't work | |
| R0051 | Worked | |
| K081018 | Didn't work |
04-Agust-2009
Only the transformed cells from yesterday's experiments are kept in incubation, being prepared for tomorrow’s extraction.
05-Agust-2009
We made mini preps ( see protocol in protocols section) and did the follow restrictions:
| Number | Biobrick | Restriction |
|---|---|---|
| 2 | BBa_R0079 | S & P |
| 3 | BBa_F1610 | X & P |
| 4 | BBa_K091146 | S & P |
| 5 | BBa_K093005 | X & P |
| 6 | BBa_K081016 | X & P |
| 7 | BBa_EC840 | X & P |
| 8 | BBa_K081009 | X & P |
| 9 | BBa_R0051 | S &P |
| 10 | BBa_J06800 | X & P |
| 11 | BBa_Q04121 | X & P |
| 12 | BBa_B0034 | S & P |
| 13 | BBa_C0079 | E & S |
| 14 | BBa_C0179 | E & S |
| 15 | BBa_B0015 | E & X |
| 16 | BBa_K081018 | E & S |
| 17 | BBa_K116640 | X & P |
| 23 | BBa_S03154 | X & P |
| 24 | BBa_k145201 | E & S |
06-Agust-2009
- We prepared 5ml falcon tubes containing liquid agar and Ampr for inoculation.
- We designed primers for 11 (Bba_Q04121) & 12 (BBa_B0034) we niknamed as AMPLICON.
07-Agust-2009
- Miniprep next clones: BBa_K081018, BBa_K081009, Bba_S03154, BBa_K145201, and prepare glycerol stocks.
10-Agust-2009
We made DNA purification from the Gel an we are planing start ligations this week.
11-Agust-2009
- Digest wit ECORI we use this method to check out if is the plasmid correct and begin ligations.
12-Agust-2009
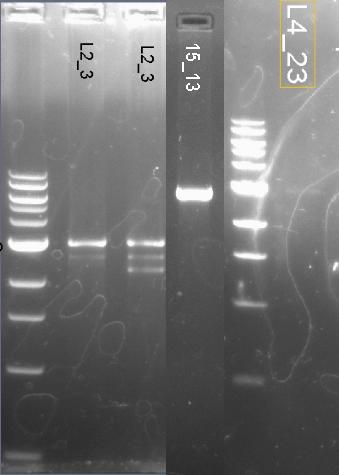
We start with ligations 15-13 (BBa_K266002), 2-3 (BBa_K266000) and 4_23 (BBa_K266005).
In this step we use 2 as backbone and 3 as insert, on the same 15 as backbone and 13 as insert, for 2-3 (BBa_K266000) we expect 3034 bp band as shown below, for 15-13 4074 bp. 4 as backbone 23 as insert. For ligation we use purifed DNA from agarosa Gel.
| Mix Ligation | Per reaction |
|---|---|
| Plasmidic DNA | 5μl |
| Insert DNA | 25μl |
| T4 DNA Ligasa Buffer | 3μl |
| T4 DNA ligasa | 2μl |
| ATP | 3μl |
The ligation left 17 hours overnight in 14ºC.
13-Agust-2009
We transform the ligation by thermic shock and plate. We received primers for 11 and 12.
14-Agust-2009
The transformation was successful we get colonies, we picket out for plasmid and left in 37ºC overnight.
15-Agust-2009
Only take off culture from camera and prepare for monday mini prep.
17-Agust-2009
We did mini prep and run out on a gel, we expect 3034 bp for L2_3(BBa_K266000) and 4074 bp for L15_13 (Bba_K266002) this are successful not so for 4_23 this is unsuccessful and proceed to repeat the ligation.
18-Agust-2009
The next step is make a ligation Amplicon – 15_13 (Bba_K266003) from the 15_13 DNA purification we left 17 hours ligation (overnight)
19-Agust-2009
Do double digestion for 15_13 as insert and aplicon as backbone overnight digest.
20-Agust-20009
Purificated from gel for begin ligation.
21-Agust-2009
The transformation was through thermick shok and plate the weight we expect is amplicon 1515 bp 15_13 4074 bp total for Bba_K266003 5574 bp.

22-Agust-2009
The transformation was successful and we get colonies picket out for midi prep. For ligations we're use midi in this step we need more DNA for ligations. The plasmid weight grow up.
23-Agust-2009
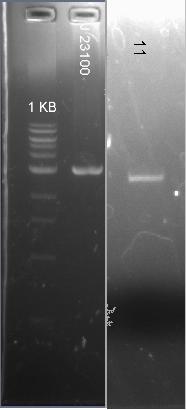
Meeting; we are detected a mistake on the primer, our primer musted include J23100 promotor now we have to do a extra ligation, and use J23100 as backbone.
24-Agust-2009
We going to do double digest for J23100 ECORI and SPEI and 11 like insert 17 hours digest overnight.
25-Agust-2009
Carried out on the digest on a gel and we watch correct weight we going to proceed to ligation.
For ligation we going to do a mix and leave 17 hours in 14ºC camera.
26-Agust-2009
We transform by thermic shock and plate.
27-Agust-2009
The transformation was unsuccessful we haven't colonies. We going to repeat the ligation and left overnight incubation 14ºC.





 "
"