Team:Minnesota/Notebook
From 2009.igem.org
| Home | The Team | The Project | SynBioSS Designer | Modeling | Experimental | Competition Requirements |
|---|
Contents |
Overview
This year’s AND gate project is a continuation of previous years. In the past different combinations of Tet, Lac, and null operator sites were made to produce an AND gate. From last year results, we decided to use the Tet Tet Lac promoter because it was the least leaky. We decided to mutate the palindromic sequence in the Tet operator site. This would alter the binding affinity of TetR for the operator site. By doing this, we can have the modeling team more accurately predict the behavior of our promoter. We also made and tested the TNN and TTN constructs in the hopes that the models would add together thermodynamically to be equivalent to our AND gate data.
Standard Protocols We Used in the Wet Lab
These link to pdf files:
Transformation of Chemically Competent Cells
Polymerase Chain Reaction (PCR)
Procedure
- Prepare Competent Cells for TOP10 and DH5αPro
- Design the primers so that the proper mutations exist in the palindromic sequence of the Tet operator site. Design for each construct: TNN, TTN, and TTL.
- Combine Soeing PCR reagents with primers and place in a thermocycler.
- Amplify the insert using PCR
- Purify the PCR product using QIAquick PCR purification
- LigateLigation the insert into pGLOTopo
- The plasmid is transformed into TOP10 cells
- The transformants are plated then screened screened using a florescent camera
- Once the colonies have been screened, positive colonies have their plasmids isolatedplasmids isolated.
- The plasmids are then sequenced to ensure the correct sequence was obtained.
- Transform the cells into DH5αPro and TOP10 and perform sample collection
- The samples are left at 4 °C for 24 hours.
- The relative GFP intensity is taken with FACSCalibar flow cytometer.
Discussion of General Trends Found
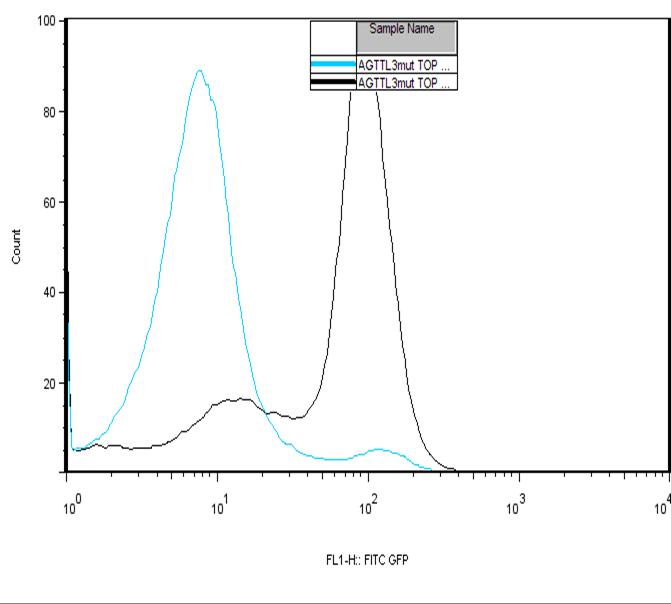
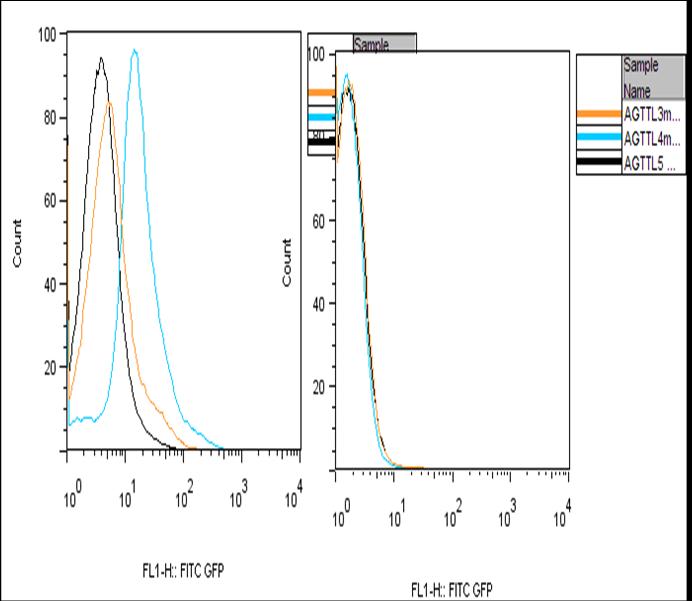
The positive control showing a weak signal can be explained by the GFP forming inclusion bodies in high concentrations. The inclusion bodies would be fluorescently inactive and show a negative GFP signal. This theory is also support by Figure 1.C which shows two peaks for two time points. At the earlier time point, the negative GFP peak is weak while the positive gives a strong signal, however at the later time point the negative GFP signal is stronger and the positive GFP. There is also roughly the same area under both peaks meaning that it is likely that the counts from one event are being transferred to the other. Since time is the only factor and GFP accumulates over time, it can be inferred that at high concentration the cell forms the fluorescently inactive inclusion bodies. The large increase from an aTc concentration of 10 ng to 50 ng that is observed is likely explained by the saturation of TetR. With the TetR bound to its inducers, it wouldn’t bind to the operator site and produce a greater GFP intensity. However, the rate of GFP production in the TTL construct seems to be governed more strongly by the Lac operator site. This is likely due to LacR being a very large molecule which prevents polymerase from binding to the operator site. The mutations in the Tet operator site were made to change the binding affinity of TetR for the operator site. These alterations not only affect the binding affinity of just the repressor protein but also the inducer-repressor protein complex. This means that not only the leakiness of the promoter will be affected. The efficiency of repression is also affected. Our results from the AND gate expression did not support what was found when just studying just mutations in the Tet operator . The highest expression was observed with mutant 4 while mutant 3 and 5 had a slight decrease in efficiency of transcription.
Safety
1. Would any of your project ideas raise safety issues in terms of:
researcher safety, public safety, or environmental safety?
No, our constructs are simple promoter regulatory elements, built from well characterized lactose and tetracycline operon components, and characterized in non-pathogenic strain of E. coli.
2. Is there a local biosafety group, committee, or review board at your institution?
Yes, there is an Institutional Biosafety Committee at the University of Minnesota. We have described our work and gotten permission to work in a wet lab (IBC Code Number: 0706H11321).
3. What does your local biosafety group think about your project?
We use standard molecular biology techniques. IBC readily approved a continuing review.
4. Do any of the new BioBrick parts that you made this year raise any safety issues? If yes, did you document these issues in the Registry?
No, none of the parts we built raise any safety issues.
Notebook
This calendar contains a day-by-day catalog of what we did in the wet lab for our project and parts characterization. The calendar for computational work can be found below and on the Modeling page. Please click on each event to see a detailed description of what we did.
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 "
"