Team:Missouri Miners/Project
From 2009.igem.org
| Home | The Team | The Project | Parts Submitted to the Registry | Modeling | Notebook | Articles | Sponsors |
|---|
|
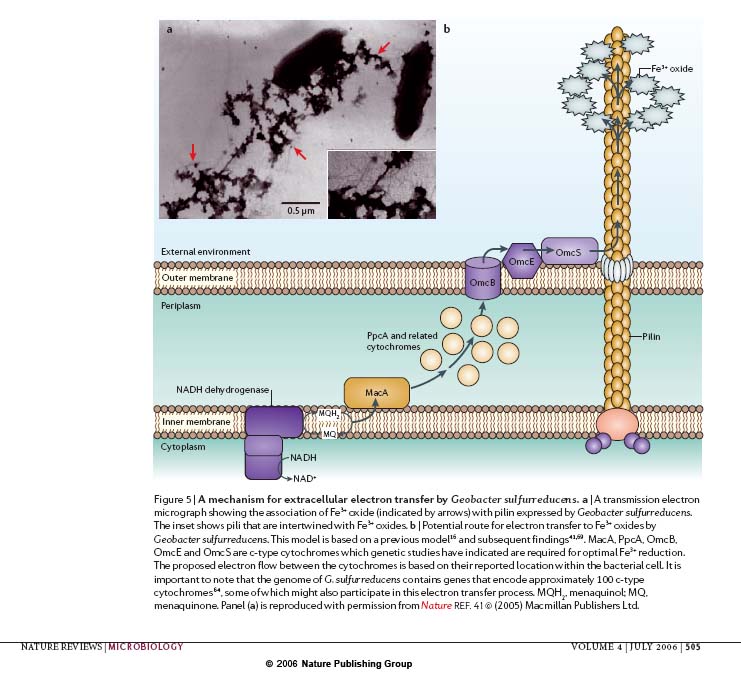
Current PROJECT - Microbial Fuel Cell: The goal of this research is to manipulate E.Coli; thus granting them the ability to release electrons in an aerobic environment. This project utilizes geobacter's cytocromes, which makes its extracellular electron transfer possible. OmcB,OmcE,OmcS, and MacA are our target genes, since their proteins are the major electron smugglers out of the cell. By isolating and placing these cytocromes into bricks, we hope to harness the ability to produce electricity in biological systems.
The purpose of this research is to use recombinant technology to culture yeast cells capable of determining the concentration of ethanol and using these cells to construct an ethanol sensor. Metabolic pathways exist for the metabolism of methanol and ethanol within some species of the Pichia taxa to include the yeast of our interest, Pichia pastoris. Alcohol oxidase (AO) appears to be the first and major enzyme produced in the methanol metabolic pathway of P. pastoris. However, if both ethanol and methanol are present, P. pastoris will utilize the ethanol before consuming the methanol. Consequently, the AOXI gene will not be expressed to produce the AO enzyme until the ethanol has been consumed. Fusing the AOXI gene promoter with the DNA sequence encoding a fluorescent protein will allow the expression of the AOXI gene to be detected. In supplying the yeast cells with ethanol and methanol simultaneously, the cells will produce the fluorescent protein once the ethanol is utilized. The concentration of ethanol can then be determined by measuring the time before fluorescence is detected.
Overall projectThe optimization of electron shuffle within bacteria to external surfaces such as anodes is one of the primary goals of our project. Geobacter sulfurreducens is the bacteria of choice due to its ability to efficiently export electrons outside of the cell. E. coli is our desired "skeleton" bacteria for our project due to the bacteria's genome already containing many key proteins in the desired pathway. Extracellular pilin, MacA, and many other cytochromes that E. coli does not have will be isolated from Geobacter sulfurreducens and introduced into E. coli to create the most optimal pathway for generating electronmotive force in a microbial fuel cell apparatus. A handful of problems have surfaced already in our project this year. Two of the most evident are correct plasmid engineering and Geobacter's anaerobic respiration contrasted to E. coli's aerobic respiration. Also, the role of Geobacter's pili in extracellular electron transfer is not clearly understood and could create a significant problem due to E. coli not having such pili. As a team, we will strive in the right direction harder than an emf on the internal resistivity of a toroid. Our team consists of many students working together from many diverse areas including Missouri S&T’s Electrical, Chemical, and Biological Engineering department along with the Biological Sciences. Project Details
Possible Applications: The applications of a interchangeable electron transfer gene part are limitless. The cytochrome cocktail could be used as a signal within bacteria. It would produce small currents which would be utilized in a similar fashion to fluorescent proteins as markers and indicators of cellular activity. Micro and nano-electronics could be adapted to use electron transfer modified bacteria as a cheap and effective power source. Medical research could benefit by using modified bacteria in areas of the body such as the stomach and intestines. The bacteria could be developed to act as a long term mobile monitor inside the human body. |
 "
"