Team:TzuChiU Formosa/background
From 2009.igem.org
Background
The Light Sensor
.
Cph8 The light sensor is an artificial gene regulation system, which can be mediated by visible light. For our project the light sensor would activate downstream signal cascade and subsequently turn on expression of the reporter gene(aeguorin-GFP).
Plants and some bacteria use phytochromes to control photosynthesis. The light-sensitive protein pigment from cyanobacterium Synechocystis sp. PCC6803 consist a two-component protein. The one component is a cyanobacterial phytochrome 1(Cph1) which is a membrane bound sensor that responds to light. The other component is phycocyanobilin(PCB)which has iron molecule bind to the Cph1 and make the component to sensitize far red light. The photoreceptor are not found in Esherichia coil(E.coil), so a created light sensor was produced in 2005 by Austin iGEM team.
The Austin iGEM team created the Cph8 which was transformed to Cp919. Cp919 is an EnvZ knockout E.coil strain. The chimaera Cph8 was a fusion protein which had cyanobacterial phytochrome 1(Cph1)with E.coil intracellular histidine kinase domain(EnvZ). They also inserted two phycocyanobilin -biosynthesis gene(ho1 and pcyA)into phycocyanobilin. The part of the photoreceptor responds to light after PCB bind to cph1. The response-regulator affects transcription by the OmpR-dependent OmpC promoter and the aeguorin-GFP repoter. The cph8 is a bridge between light sensor and gene regulation.
In the present project, we transformed the reporter gene(aeguorin-GFP)to Cp919. Under light condition, the cph8 had little kinase activity and the OmpR remained unphosphorylated, and reporter gene would not expressed. On the other hand, the cph8 had activity and high levels of OmpR would be phosphorylated in dark. OmpR-p then would bind to ompC promoter, resulting in expression of high level of aeguorin-GFP .
The Light Expression
.
Aequorin and GFP
Aequorin and GFP(Green Fluorescent Protein)has existed in the world for more than one hundred millions years and were discovered in only one jellyfish specie-Aequorea Victoria. When Ca2+ ions bind on aequorin, the aequorin will emit a glowing blue light and GFP will be shifted to excited state and emit green fluorescence. The major excitation wavelength for GFP is 395nm, the second peak is at 475nm and emission peak is at 509nm.
Aequorin is composed of two distinct units: a) the apoprotein: apoaequorin and b) the prosthetic group: coelenterazine (a luciferin).
Aequorin activation is dependent on calcium influx. When Ca2+ stream flows in and binds to aequorin, the prosthetic group will be oxidized. The coelenterazine converts into the excited-state coelenteramide, and then shift back to the ground state. Then Aequorin can emit blue light (wavelenght= 469 nm) and provide energy for GFP expression.
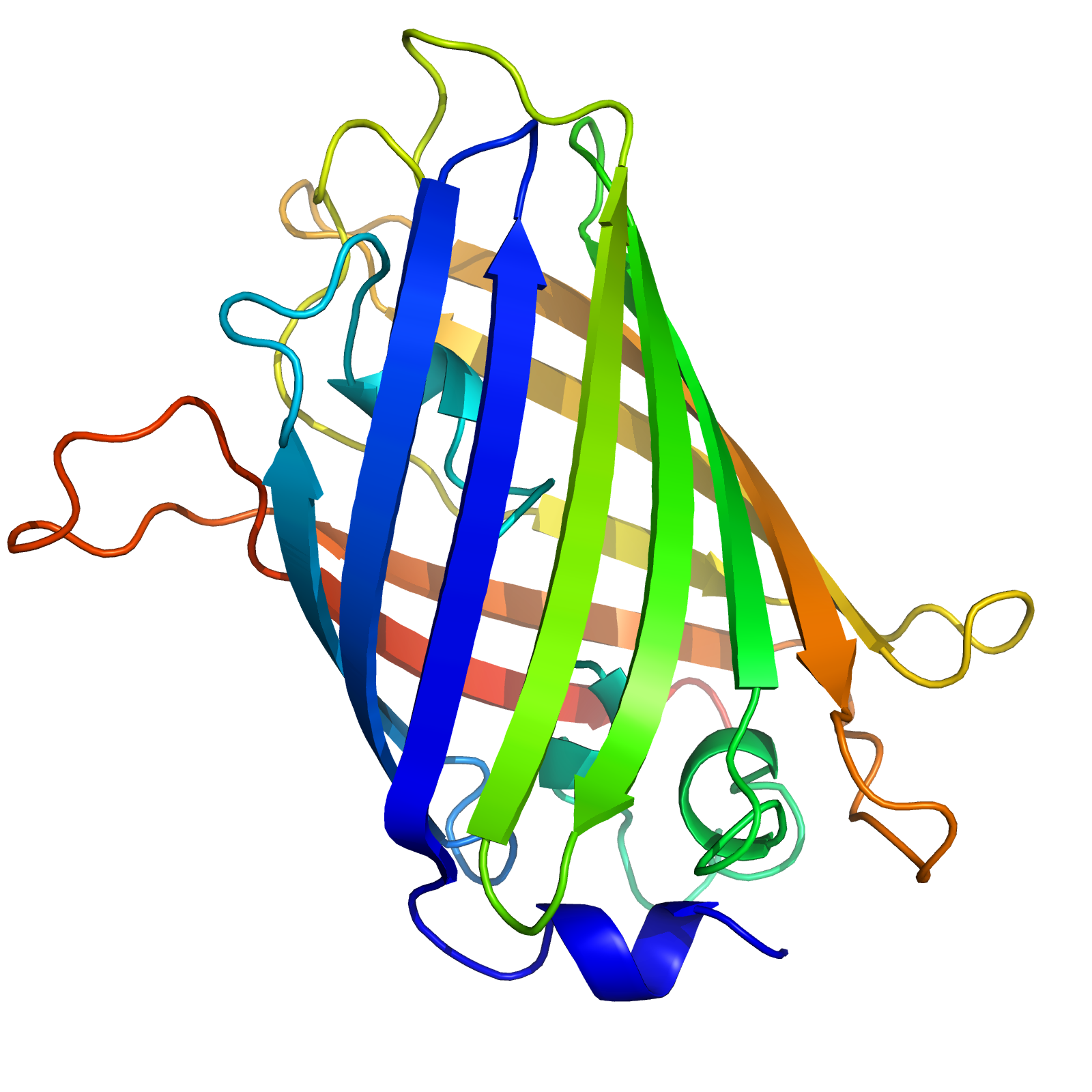
The structure of GFP is beta barrel, it constituted beta sheets with alpha helix and chromophore is located in the middle of the beta barrel which is responsible for fluorescence of GFP.
GFP has been widely used as a indicator to report level of gene expression. In our research, we hope to use illuminance of Aequorin-GFP fusion protein to substitute light sources generated by electric power.
 "
"