Team:ULB-Brussels/Project/Perspectives
From 2009.igem.org
GluColi: a future alternative to toxic glues?
Glue has become a material of everyday life but we have to know that common glues contain Volatile Organic Compounds (VOC). These substances are air pollutants and toxic.(19) (20) (21) For example, cyanoacrylate commonly sold under trade names like super glue can cause various toxic issues depending on their precise composition, (22).
According to Yves Brun, Indiana University biologist, concerning Caulobacter crescentus’s glue: ”This natural, non-toxic glue [...], has the strongest adhesion force of any known natural material. Unlike commercial super glues, which are often toxic, it sticks well under water, even salt water, and, thus, has a wide range of potential applications ”, (13).
Indeed, succeeding in the production of our GluColi could offer a wide range of useful applications.
That could cover fields ranging from medical purposes to ship repairing and automotive or aeronautic industry.
Bone reconstructive surgery has lead to a large demand for bone graft, (26), (27). Consequently, synthetic bone graft substitutes have been developped with mixed success and surgical acceptance. Bone engineering has still lots of challenges to cope with. Surgical adhesives are part of this field of research but toxic issues are sometimes reported. Looking for new adhesives is today of great interest (28). In that, GluColi may play an interesting role.
GluColi and a free-antibiotics alternative
There is no Ethic Comity at the Institute of Molecular Biology and Medecine. However the governing body of the ULB supported us in the achievment of our project.
In this work, we intended to be as far as possible aware of some ethical issues, in particular the use of antibiotics and the toxicity of the current adhesives. Antibiotics are used to select all types of plasmidic vectors in bacteria which is a significant concern in industrial cultures producing recombinant proteins or DNA. As plasmid-free cells grow faster than plasmid-carrying cells, the yield and the production reproducibility of recombinant molecules are significantly lowered in the absence of plasmid selection by the appropriate antibiotic.
To overcome the plasmid instability, many antibiotic resistance genes are used as selectable markers in fermentation processes. Antibiotics are expensive and they pose safety problems: they contaminate the production product. (23) (24) The Belgian company Delphi Genetics has developed a strategy called StabyTM system which relies on the use of poison-antidote genes.
The StabyTM system is based on the poison-antidote ccd module. Poison-antidote modules are found in natural plasmid in which they serve to the plasmid maintenance. The CcdB protein (poison) is cytotoxic and poisons DNA-gyrase complexes. Expression of this gene in the absence of its cognate ccdA antidote leads the death of the bacteria. The product of the ccdA gene (antidote) antagonizes this toxic activity by forming a poison-antidote protein complex.
If a plasmid carrying the ccd module is lost at cell division, the plasmid-free bacteria contain poison and antidote proteins in their cytoplasm. Since the antidote is unstable and degraded by a host protease, the poison will be free and able to poison DNA-gyrase complexes. This will lead eventually to cell death.
In theStabyTM system,the antidote gene (ccdA) is introduced in the plasmid DNA under the control of a weak constitutive promoter: the mob gene promoter, which originates from a broad host range plasmid (pBHR1). On the other hand, the toxic gene (ccdB) is introduced in the E. coli chromosome of the bacteria. Expression of the poison gene is under the control of a promoter strongly repressed in the presence of the plasmid. Practically, 100 % of the bacteria will carry the vector. If they lose the vector, they will not obtain a growth advantage, but will die. Upon induction, all the bacteria will start producing the recombinant protein. It will lead to higher yields of the recombinant protein and less background caused by unwanted proteins. Therefore, higher plasmid stability means higher protein of interest production.
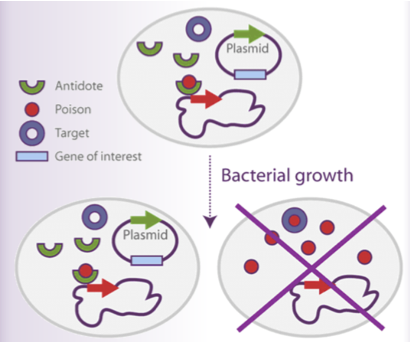
We think it could be relevant to implement this antibiotic free expression strategy in our project. This is the reason why we designed 2 bricks containing the ccdA gene. In order to carry out transformation, bacteria should be furnished with the ccdB gene in their genome (i.e. by Delphi Genetics).
Conclusions
In this report, we describe a new way to produce a strong bioadhesive. We transferred part of Caulobacter crescentus’s holdfast system in Escherichia coli which produced an adhesive and was able to stick.
We conducted a few experiments in order to characterize the glue. The glue did not show all the expected adhesive properties. However they could be improved by purifying the glue.
In addition, we designed a biological circuit that would allow us to integrate the glue synthesis into a useful application. The circuit described in this report should activate the glue synthesis only in specific spots where repairs are needed. This is achieved by attracting GluColi bacteria to the break and inducing the glue synthesis once the adequate cell density is reached (i.e. thanks to quorum sensing).
For the mathematical modeling part of our project we built a theoretical system which is able to produce the glue. However, we found two main improvements to increase the robustness of the system. Firstly, by considering the complete dynamics of the Quorum Sensing system, we showed that it is possible to controle the glue production only with the quantity of inductor. Secondly, we increased the robustness of our system by adding a toggle switch. This last improvement establishes a connection between the initial concentration of the c2 repressor and the IPTG value we put in the system to start the glue production process.
 "
"
