User:DavidC/7 October 2009
From 2009.igem.org
(→DNA electrophoresis) |
(→DNA electrophoresis) |
||
| (2 intermediate revisions not shown) | |||
| Line 44: | Line 44: | ||
| - | Samples: | + | Samples: Fusion protein report 1, 2, 3, 4. |
| + | <br> | ||
| + | |||
| + | [[image:W0710(2).png|center]] | ||
| + | <br> | ||
We obtain a fragment at 385bp, so the ligation does not work as expected. The fragment at 385bp is similar to the D protein, so there is not enough adenovirus 5 penton base into the sample or there is no DNA. <br><br> | We obtain a fragment at 385bp, so the ligation does not work as expected. The fragment at 385bp is similar to the D protein, so there is not enough adenovirus 5 penton base into the sample or there is no DNA. <br><br> | ||
| Line 79: | Line 83: | ||
<br> | <br> | ||
| - | [image:W0710.png|center]] | + | [[image:W0710.png|center]] |
<br> | <br> | ||
Latest revision as of 16:31, 21 October 2009
Wednesday the 7th
PCR
Trial for amplification of ligation between D protein and adenovirus 5 penton base
Sample:
D protein (D prt) and adenovirus 5 penton base (ADV5 pb) digest by Spe I and Xba I (samples created on the 2nd of October).
Primers:
Forward : Pref_ATG_Fw
Reverse : Suffixe_Rv
DNA:
5µL of each samples: first, second, third and fourth reports.
Cycles:
94°C 2minutes;
(94°C 1 minute;
59°C 1 minutes;
72°C 2 minute(s)) X 35;
72°C 5 minutes;
DNA electrophoresis
85 Volt, 15 minutes.
105 Volt, 40 minutes.
Ladder fermentas 1 Kb.
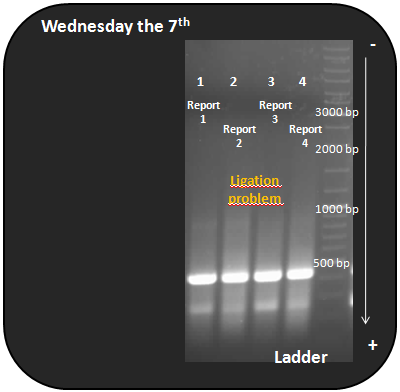
Samples: Fusion protein report 1, 2, 3, 4.
We obtain a fragment at 385bp, so the ligation does not work as expected. The fragment at 385bp is similar to the D protein, so there is not enough adenovirus 5 penton base into the sample or there is no DNA.
We will change the next protocol and try to integrate the D protein and the ADV5 pb into plasmid vector independently and join them as a plasmid vector and a insert.
Restriction digest
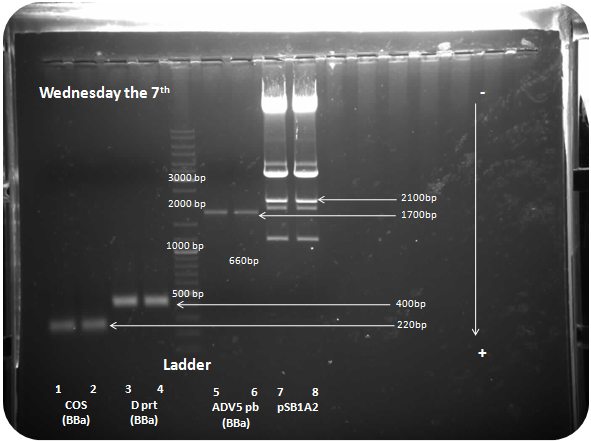
Restriction digest of adenovirus 5 penton base (1715bp), COS (250bp) and D protein (380bp) by Xba I and Pst I :
DNA (PCR) = 10µL
Buffer M (TAKARA) = 2µL
H20 = 6µL
Xba I (TAKARA) = 1µL
Pst I (TAKARA) = 1µL
1 hour of incubation at 37°C.
Restriction digest of pSB1A2 Xba I and Pst I (2079bp):
DNA (PCR) = 10µL
Buffer M (TAKARA) = 2µL
H20 = 6µL
Xba I (TAKARA) = 1µL
Pst I (TAKARA) = 1µL
1 hour of incubation at 37°C.
DNA electrophoresis
85 Volt, 15 minutes.
105 Volt, 40 minutes.
Ladder fermentas 1 Kb.
Samples: COS with Biobrick prefix and sufix (COS BBa)(250bp), D protein with Biobrick prefix and sufix (D prt BBa)(385bp), adenovirus penton base with Biobrick prefix and sufix (ADV5pb BBa)(1715bp), pSB1A2 (2079bp).
DNA purification
Kit Qiagen “gel extraction kit”, final volume = 50µL.
Ligation
Ligation between ADV5 pb and pSB1A2:
First report:
Plasmid (pSB1A2) = 2µL
Insert (ADV5 pb) = 6µL
Solution A = 1µL
Solution B = 1µL
Second report:
Plasmid (pSB1A2) = 3,5µL
Insert (ADV5 pb) = 10µL
Solution A = 2µL
Solution B = 2µL
2 hours of incubation at room temperature.
Same protocol for:
Ligation of COS with pSB1A2.
Ligation of D protein with pSB1A2.
 "
"