Team:Bologna/Characterization
From 2009.igem.org
| HOME | TEAM | PROJECT | SOFTWARE | MODELING | WET LAB | PARTS | HUMAN PRACTICE | JUDGING CRITERIA |
|---|
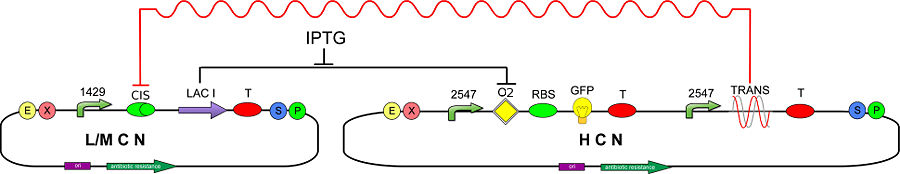
We decided to create and developed a biological circuit in which the TRANS-repressor, in the absence of IPTG, can inhibit the synthesis of LacI repressor protein, warranting the production of GFP:
To do that we initially needed to characterize some sub-circuits in order to obtain some information and values concerning the processes we were analyzing.
You can find here all our Wetlab Protocols
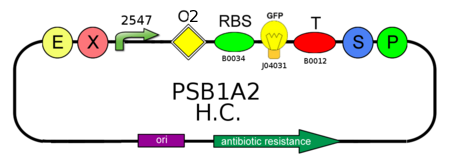
Plasmid copy number characterization
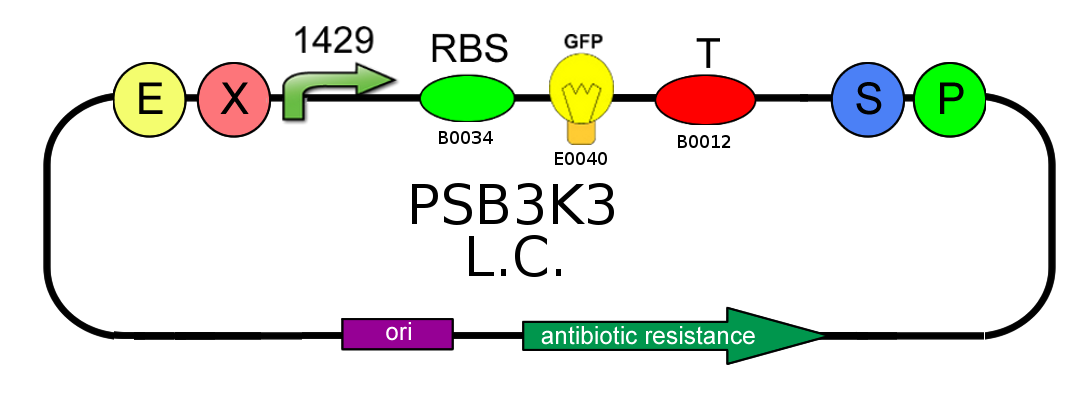
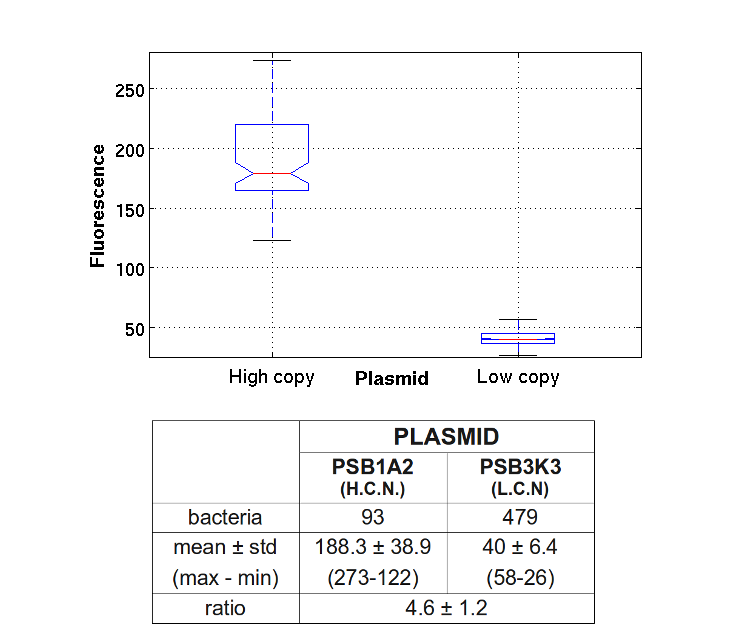
To test the ratio between the production of an high copy number plasmid (PSB1A2) and a low copy number one (PSB3K3), we assembled two circuits. The open loop GFP circuits are realized with a 1429 promotor and the standard biobrick I13504. From the Registry of Standard Biological Parts we knew that pSB1A2 is an high copy number plasmid while pSB3K3 is a low copy one, so the ideal ratio between their copy number is at least 10, but the best value that we reached with the spectrofluorimeter is about 3,3.
PSB1A2 with high copy number plasmid and a low copy number were transformed in DH5alfa bacterial cells according to the standard protocol.
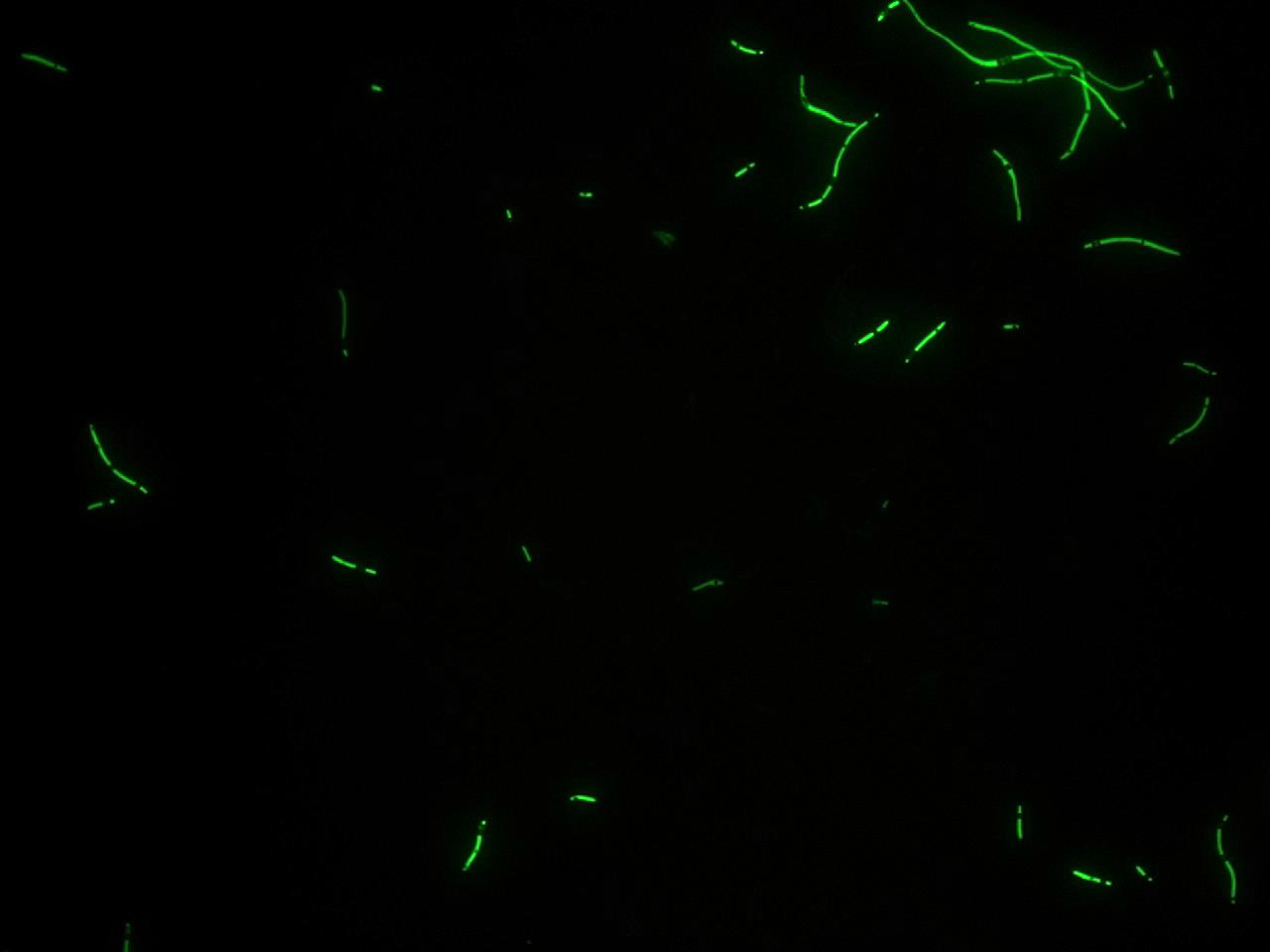
One colony from each plate was picked up and let grow overnight in M9 medium at 37°C. One milliliter for each of the two samples was collected by O/N cultures and spinned at 8000 rpm for a minute; another milliliter was used for measuring the optical density and estimate the growth of the sample. The supernatant was harvested and the pellet resuspended. Slides were prepared for the fluorescence bacteria image acquisition.
Finally, images were elaborated with the fluorescence visualization software and these are the results:
Promoter characterization
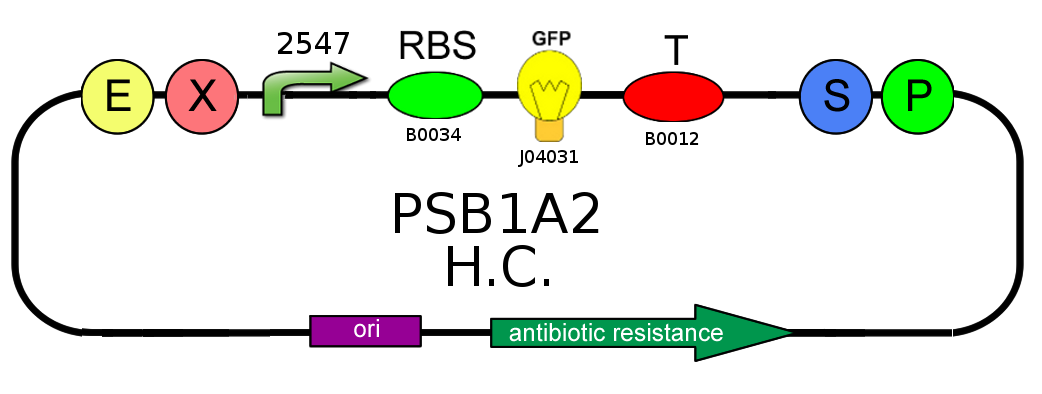
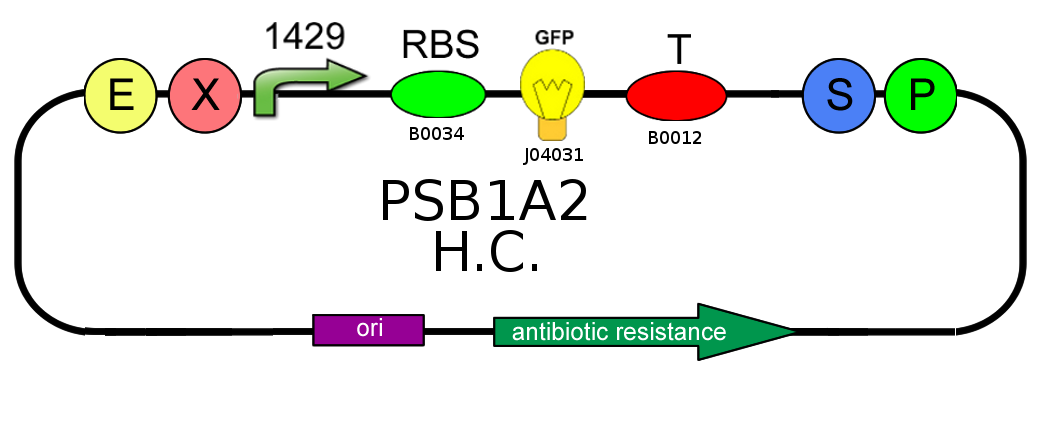
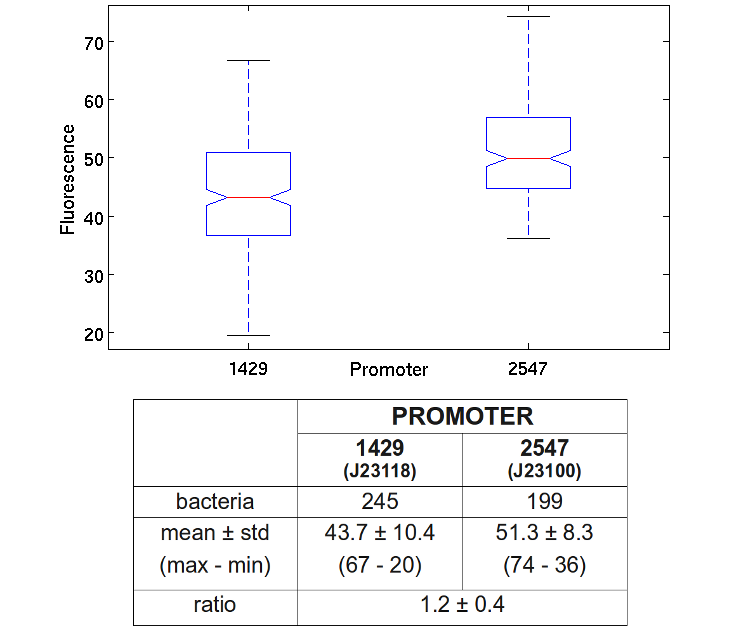
In order to estimate the difference in strength of the two promoters J23100 (2547) and J23118 (1429), we realized two circuits. Both were composed by an open loop GFP mounted on a high copy number plasmid (PSB1A2), the only different element was the promoter.
To do this we transformed those constructs in bacterial cells; we picked up a colony from each plate and we inoculated it in M9 medium. After growing all night at 37°C we took a milliliter of each sample and we measured their optical density; than we prepared slides for the fluorescence bacteria images acquisition, following the same steps of the previous test. The images, acquired during some repetitions of the test, was elaborated with the fluorescence visualization software (VIFluoR) giving out those results:
From the registry of standard parts we learnt that the strengths of J23100 and J23118 are respectively 2547 and 1429, so the ratio between them is about 1.78. Experimentally we have achieved the value of 1.2; for this reason we can say that this prove has gone well.
GFP production in absence/presence of operator O2
With this trial we aimed to demonstrate that, in absence of LacI protein, the operator O2 doesn't affect the bacterial production of GFP.
To do that we realized four circuits: the first was composed by the GFP protein under the control of the J23100 promoter mounted on the pSB1A2 plasmid; the second circuit differed from the first because it contained also the operator O2. Similarly the third and the fourth circuits differed only for the presence or the absence of the operator, but they mounted the J23118 promoter instead of the J23100.
Following the steps of the previous tests we obtained those results:
 "
"