Team:DTU Denmark/theory
From 2009.igem.org

| Home | The Team | The Project | Parts submitted | Modelling | Notebook |
|
The redoxilator - Theoretical background - Yeast as a model organism - Practical approach The USER assembly standard - Principle - Proof of concept - Manual - Primer design software |
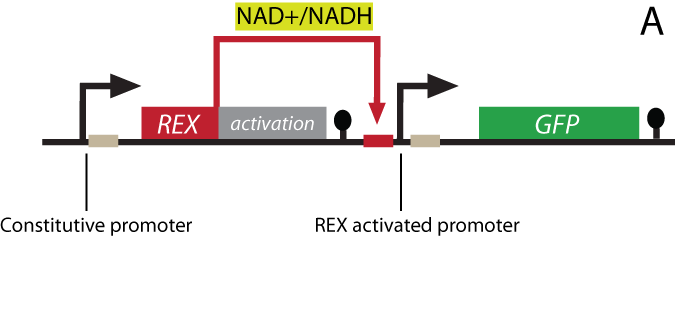
The project Theoretical background The NAD+/NADH ratio sensor-protein Rex (Redox regulator) has been discovered in the bacterium Streptomyces coelicolor. In its host organism, the sensor works as a repressor and controls the gene expression of a large number of genes by recognizing and binding to a specific DNA-sequence termed ROP (Rex OPerator). NAD+ and NADH compete for Rex binding, and the protein binds the ROP DNA-sequence only when NAD+ is bound. Our synthetic biology project: The Redoxilator To achieve a system that senses changing levels in the NAD+/NADH ratio in the eukaryote S. cerevisiae, the gene encoding the Rex protein will be fused to a yeast activator domain, resulting in a new synthetic protein: the Redoxilator. The ROP sequence - the DNA binding site Rex can bind to - will be inserted into a yeast promoter, resulting in a promoter activated by the Redoxilator.
Figure 1 - Gene design and redox regulation
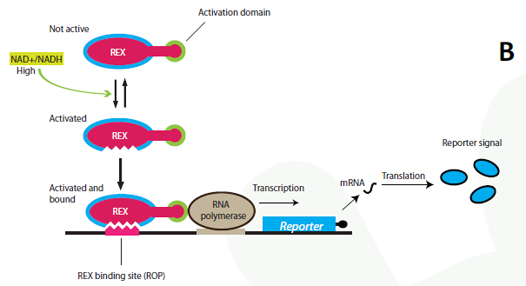
A certain NAD+/NADH ratio will activate the Redoxilator to recognize the ROB promoter resulting in transcription of a downstream gene. In this way the ROB promoter and the Redoxilator comprises the complete sensing system. The system can be coupled to the expression of virtually any gene of interest; making transcription solely dependent on the ratio of NAD+/NADH in the cell. In our iGEM project, the system will be used for two selected applications considered highly relevant: i) in vivo monitoring of NAD+/NADH in yeast, and ii) NAD+/NADH ratio regulated production of yeast products in chemostat processes. i) Reporter gene expression regulated by the Rexivator – an in vivo redox sensorThe gene encoding green fluorescent protein (GFP) is widely used as a reporter gene in molecular biology. By placing the ROB promoter upstream of a GFP gene on a plasmid, and transforming the whole system into a yeast cell, GFP will be expressed at certain NAD+/NADH levels. When the Rexivator is bound to DNA, GFP expression will produce a visible and quantitatively measurable signal, which will be an indirect measure of the NAD+/NADH ratio.
Figure 2 – Schematic overview of overall approach. |
The yeast metabolic cycle It has recently been shown by Tu et al. and Klevecz et al. that the expression of at least half of the genes monitored on a standard yeast gene chip will oscillate in a coordinated manner when grown under glucose limited conditions. The cells will shift between oxidative and reductive metabolism in a synchronized metabolic cycle with three phases: oxidative, reductive/building and reductive/ charging. As oxygen will only be consumed in the oxidative phase, the dissolved oxygen will oscillate. Many metabolites and cofactors including NADH and NAD+ will also oscillate during this cycle as NADH is converted to NAD+ when oxygen is consumed. |
| Comments or questions to the team? Please Email us -- Comments of questions to webmaster? Please Email us |
 "
"