Team:Groningen/Notebook/20 July 2009
From 2009.igem.org
(→Vectors) |
(→Dry) |
||
| (15 intermediate revisions not shown) | |||
| Line 4: | Line 4: | ||
===GVP Cluster=== | ===GVP Cluster=== | ||
| + | |||
| + | |||
| + | '''Precipitation of the restriction fragments from GVP, J23109, J23100 and J23106''' | ||
| + | |||
| + | To concentrate [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23109 BBa_J23109], [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23100 BBa_J23100], [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23106 BBa_J23106] and [http://partsregistry.org/wiki/index.php?title=Part:BBa_I750016 GVP] it is first precipitated and then MQ is added. | ||
| + | |||
| + | Procedure: | ||
| + | * 100 μl absolute ethanol is added to ~50 μl Restriction fragment of GVP (12.1 ng/μL),J23109 (13.1 ng/μl), J23100 (11.9 ng/μl) and J23106 (11.1 ng/μl). | ||
| + | * Incubation -80°C for 1 h | ||
| + | * Centrifugation 30 min 0°C (14000rpm) | ||
| + | * Supernatant is removed | ||
| + | * Wash with 1000 μl 96% Ethanol (added ethanol and inverted a couple of times) | ||
| + | * Centrifugation 10 min 4°C (14000rpm) | ||
| + | * o/n air dried on bench top | ||
| + | |||
| + | |||
| + | '''Preparation of 1-14N and 1-1D for glycerol stocks, restiction analysis and 3A assembly''' | ||
| + | |||
| + | 1-14N (<partinfo>BBa_I0500</partinfo>) and 1-1D (<partinfo>BBa_R0010</partinfo>) are inducible promoters, induced by L-arabinose and lactose (or IPTG) respectively. | ||
| + | |||
| + | * DNA from iGEM plates resuspended in 15 μL MQ and stored at -20°C | ||
| + | |||
===Transporters=== | ===Transporters=== | ||
| - | Below the PCR of 17/07, There seems to be a vague band at ~1150 in 1. | + | Below the PCR of 17/07, There seems to be a vague band at ~1150 in lane 1. |
| Line 36: | Line 58: | ||
|} | |} | ||
| + | |width="10"| | ||
| + | | | ||
<!--Tabel 2 hier--> | <!--Tabel 2 hier--> | ||
| Line 72: | Line 96: | ||
|4° | |4° | ||
|Forever | |Forever | ||
| + | |} | ||
| + | |||
|} | |} | ||
| Line 79: | Line 105: | ||
Positive control for the psB1AC3 vector done with both new and last years primers. | Positive control for the psB1AC3 vector done with both new and last years primers. | ||
| - | + | {| | |
| + | | | ||
<!--Tabel 1 hier--> | <!--Tabel 1 hier--> | ||
| + | |width="10"| | ||
| + | | | ||
{| border="1" | {| border="1" | ||
| Line 104: | Line 133: | ||
<!--tabel 2 hier--> | <!--tabel 2 hier--> | ||
| + | |width="10"| | ||
| + | | | ||
| + | |||
{|border="1" | {|border="1" | ||
| - | |+ Primers | + | |+ '''Primers''' |
!Cup nr | !Cup nr | ||
!F primer | !F primer | ||
| Line 137: | Line 169: | ||
<!--Tabel 2 hier--> | <!--Tabel 2 hier--> | ||
| - | + | |width="10"| | |
| - | | | + | | |
| - | + | ||
| + | {| | ||
| + | |+'''VR VF2 program''' | ||
| + | !Stage | ||
! Temperature | ! Temperature | ||
! Time | ! Time | ||
| Line 155: | Line 190: | ||
|- | |- | ||
|Annealing | |Annealing | ||
| - | | | + | |61° |
|20 sec | |20 sec | ||
|- | |- | ||
| Line 174: | Line 209: | ||
|} | |} | ||
| + | |} | ||
| + | |||
| + | The gel shown below shows bands at about 1000 which is not what would be expected. Band of 315 were expected for lanes 1-6. '''--> contains ccdB gene, which would increase the expected fragment size to 991bp! This is about the size which is seen on gel....''' | ||
[[image:F102471_2009-07-20_08hr_30min_noted.JPG]] | [[image:F102471_2009-07-20_08hr_30min_noted.JPG]] | ||
| + | |||
| + | ==vectors== | ||
| + | Colony PCR on vectors for checking if promotors were inserted | ||
| + | |||
| + | - expected lengths (primer VF2 & VR) on: | ||
| + | |||
| + | psb1AC3 316 bp | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
==Dry== | ==Dry== | ||
| + | Jasper looked at GlpF import, trying to create a prototype model in Simbiology by playing with the parameters. However, it turns out that Simbiology doesn't allow reactions using reactants from different compartments, so using a Michaelis-Menten equation (instead of using more fine-grained reactions) is pretty much the only option for transporters. Also, after discovering that it is quite hard to directly derive the required constants from the given graph manually (due to the different capacities of the cell and the solution) a table was made of the uptake graph (Fig. 1B) in [[Team:Groningen/Literature#Meng2004|Meng2004]] by importing the graph in Inkscape and aligning the axes with its rulers. | ||
{{Team:Groningen/Notebook/Day/Footer}} | {{Team:Groningen/Notebook/Day/Footer}} | ||
Latest revision as of 09:33, 19 August 2009
Wet
GVP Cluster
Precipitation of the restriction fragments from GVP, J23109, J23100 and J23106
To concentrate BBa_J23109, BBa_J23100, BBa_J23106 and GVP it is first precipitated and then MQ is added.
Procedure:
- 100 μl absolute ethanol is added to ~50 μl Restriction fragment of GVP (12.1 ng/μL),J23109 (13.1 ng/μl), J23100 (11.9 ng/μl) and J23106 (11.1 ng/μl).
- Incubation -80°C for 1 h
- Centrifugation 30 min 0°C (14000rpm)
- Supernatant is removed
- Wash with 1000 μl 96% Ethanol (added ethanol and inverted a couple of times)
- Centrifugation 10 min 4°C (14000rpm)
- o/n air dried on bench top
Preparation of 1-14N and 1-1D for glycerol stocks, restiction analysis and 3A assembly
1-14N () and 1-1D () are inducible promoters, induced by L-arabinose and lactose (or IPTG) respectively.
- DNA from iGEM plates resuspended in 15 μL MQ and stored at -20°C
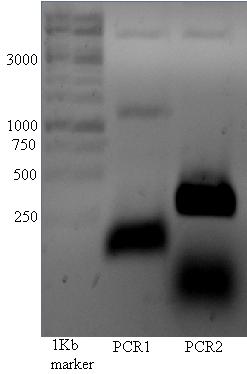
Transporters
Below the PCR of 17/07, There seems to be a vague band at ~1150 in lane 1.
The band at ~1150 kb was cut out of the gel and used for pcr described below.
|
|
Metal Accumulation
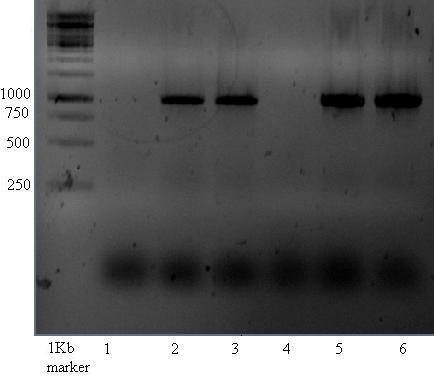
Vectors
Positive control for the psB1AC3 vector done with both new and last years primers.
|
|
|
The gel shown below shows bands at about 1000 which is not what would be expected. Band of 315 were expected for lanes 1-6. --> contains ccdB gene, which would increase the expected fragment size to 991bp! This is about the size which is seen on gel....
vectors
Colony PCR on vectors for checking if promotors were inserted
- expected lengths (primer VF2 & VR) on:
psb1AC3 316 bp
Dry
Jasper looked at GlpF import, trying to create a prototype model in Simbiology by playing with the parameters. However, it turns out that Simbiology doesn't allow reactions using reactants from different compartments, so using a Michaelis-Menten equation (instead of using more fine-grained reactions) is pretty much the only option for transporters. Also, after discovering that it is quite hard to directly derive the required constants from the given graph manually (due to the different capacities of the cell and the solution) a table was made of the uptake graph (Fig. 1B) in Meng2004 by importing the graph in Inkscape and aligning the axes with its rulers.
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"