Team:Groningen/Notebook/26 August 2009
From 2009.igem.org
(Difference between revisions)
m (→GVP Cluster) |
m (→GVP Cluster) |
||
| Line 91: | Line 91: | ||
'''Tranformation''' | '''Tranformation''' | ||
| - | :* add 10uL of the | + | :* add 10uL of the ligation product to 50uL competent E.coli TOP10 cells. |
''Incubate:'' | ''Incubate:'' | ||
:* 30 min @ ice | :* 30 min @ ice | ||
| Line 98: | Line 98: | ||
:* add 800uL LB-medium | :* add 800uL LB-medium | ||
:* incubate for 1 h at 37°C | :* incubate for 1 h at 37°C | ||
| - | :* plate on LB-amp<sub> | + | :* plate on LB-amp<sub>100</sub>/LB-kan<sub>50</sub> plates |
Revision as of 12:38, 26 August 2009
Wet
GVP Cluster
- → DONE Restriction of GVP and pSB1AC3-Lac/pBAD for assembly
- → DONE Gel purification of wanted fragments and nanodrop
- → DONE Clone GVP cluster behind Lac and Arabinose promoter for expression tests in pSB1AC3 plasmid
- → TODO Test promoter strenght compared to BBa_J23101 promoter (Sven)
- → TODO Enter sequences of constructs to Sandbox
Restriction for Assembly
The vector pSB1AC3 containing the LAC and pBAD inducible promoters were cut with PstI and SpeI to create correct ends for insert of GVP biobrick BBa_I750016, which was cut with XbaI and PstI.
- 3 to 16μL plasmid in MQ (1.0μg)
- 13μL MQ (end volume of 20μL)
- 2μL Fast digest buffer
- 1μL PstI fast digest enzyme
- 1μL SpeI/XbaI fast digest enzyme
Restriction was kept at 37C for 30 min. and put on ice until used for gel purification.
Purification
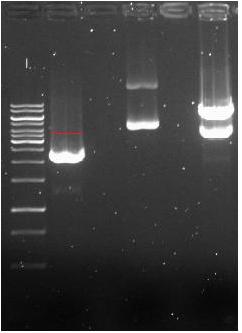
- → From left to right: 1kB ladder, pSB1AC3-LAC ,Empty Slot, pSB1AC3-pBAD-AraC , Empty Slot, GVP
- → The band for the pSB1AC3-LAC vector was expected to be at ~3700bp (red line), but instead it was at ~2500bp.
- → Comparison with the gel of Frans (constructed pSB1AC3-LAC) explained the band at a lower position (also seen on his gel), but not why it is so small. One option is use of pSB3K3 plasmid instead of pSB1AC3 plasmid, this would still give a band of ~3300bp and not ~2500bp!
A "Agarose Gel DNA Extraction Kit" Standard Protocol from Roche Applied Science.
- → In step ten, 30μL MQ was added to the dry pellet and incubated at room temperature.
Concentrations
| Plasmid | Conc. ng/μL | 260/280 | 260/230 | -20 box (michael | Restriction Control |
| pSB1AC3-LAC SpeI/PstI | 9.2 | 1.75 | 1.56 | N.S. | Gel |
| pSB1AC3-pBAD SpeI/PstI | 0.9 | 0.94 | 0.30 | N.S. | Gel |
| GVP XbaI/PstI restricted | 6.5 | 1.57 | 1.35 | N.S. | Gel |
Ligation
(1:6) (Done twice, old and new cut GVP insert)
- 4 uL Ligase buffer
- 2 ul T4 Ligase
- 8 uL plasmid pSB1AC3-LAC/pBAD digested with PstI and SpeI
- 8 uL insert GVP restricted with XbaI and PstI
(1:6)
- 4 uL Ligase buffer
- 2 ul T4 Ligase
- 8 uL plasmid pSB3K3-L digested with PstI and SpeI
- 8 uL insert GVP restricted with XbaI and PstI
Incubate:
- 25°C 50min.
- kept on ice for 10min.
Tranformation
- add 10uL of the ligation product to 50uL competent E.coli TOP10 cells.
Incubate:
- 30 min @ ice
- 90 sec 42°C
- 2 min @ ice
- add 800uL LB-medium
- incubate for 1 h at 37°C
- plate on LB-amp100/LB-kan50 plates
- → Positive control was BBa_J61002-J23101 plasmid, and negative control was MQ.
Transporters
Metal Accumulation
MBP-ArsR
- Transform ligation with inducible promotors
- Prepare glycerol stock without promotors
Vectors
Dry
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"