Team:TUDelft/ConjugationProtocol
From 2009.igem.org
Conjugation Protocol
Other Conjugation Protocols
This protocol was adapted from:
http://www.openwetware.org/wiki/Conjugation
and
http://openwetware.org/wiki/IGEM:Peking/2007/Count:Conjugation
TUDelft Conjugation Protocol
This is the Protocol Used by TUDelft iGEM 2009:
Day 1
- Get plates from fridge containing Donor and Recipient.
- Make 5 mL cultures with the appropriate antibiotics and leave overnight.
Day 2
- Prepare your plates (see tables at the bottom).
- Put 1 mL of the overnight cultures into 5 mL tubes. Usually 2 tubes of donors and 2 tubes of recipients.
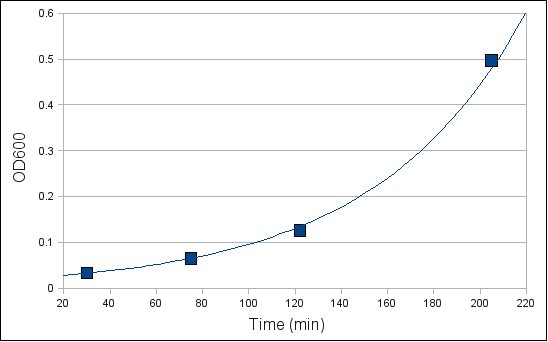
- Test the OD600. You can use the following graph to estimate the time needed for E.coli containing R751 to reach an OD600 value of 0.45 to 0.6.
- Place 500 μL of culture into enough 1.5mL Eppendorfs for the conjugation test.
- Prepare AB free plates for the conjugation. One plate for each set.
- Centrifuge 1 at 3000 rcf for 3 min and discard supernatant.
- Resuspend in 500 μL LB without AB.
- Centrifuge 2 at 3000 rcf for 2 min and discard supernatant.
- Resuspend in 500 μL LB without AB.
- Centrifuge 3 at 3000 rcf for 1 min and discard supernatant.
- Resuspend in 500 μL LB without AB.
- Place cells on ice.
- Mix 100 μL of donors and 100 μL recipients in an eppendorf tube and vortex gently.
- Centrifuge for 1 min and discard supernatant.
- Resuspend the cell pellet in 10 μL of LB.
- Put a PALL 0.2 μM filter on top of a AB free plate. Gently pipette the conjugation mix on the filter. Use good sterile technique.
- Incubate for 1 h at 37ºC.
- Pick the filter from the plate with sterilized tweezers and put it into an 50 mL assay tube with 2 mL of LB broth.
- Vortex to re-suspend the cells.
- Make serial dilutions in LB broth. Plate in selective antibiotics for donors, recipients and transconjugants.
- The following plates are usually made with R = Recipient, D = Donor, and T = Transconjugant.
| Set 1 | |||
| Plate ID | Antibiotics | Dilution | |
| D1 | TRI | 100 | |
| D2 | TRI | 10-1 | |
| D3 | TRI | 10-2 | |
| D4 | TRI | 10-3 | |
| D5 | TRI | 10-4 | |
| T1 | TRI + AMP | 100 | |
| T2 | TRI + AMP | 10-1 | |
| T3 | TRI + AMP | 10-2 | |
| T4 | TRI + AMP | 10-3 | |
| T5 | TRI + AMP | 10-4 | |
| T6 | TRI + AMP | 10-5 | |
| T7 | TRI + AMP | 10-6 | |
| R1 | AMP | 100 | |
| R2 | AMP | 10-1 | |
| R3 | AMP | 10-2 | |
| R4 | AMP | 10-3 |
| Set 2 | |||
| Plate ID | Antibiotics | Dilution | |
| D6 | TRI | 100 | |
| D7 | TRI | 10-1 | |
| D8 | TRI | 10-2 | |
| D9 | TRI | 10-3 | |
| D10 | TRI | 10-4 | |
| T8 | TRI + AMP | 100 | |
| T9 | TRI + AMP | 10-1 | |
| T10 | TRI + AMP | 10-2 | |
| T11 | TRI + AMP | 10-3 | |
| T12 | TRI + AMP | 10-4 | |
| T13 | TRI + AMP | 10-5 | |
| T14 | TRI + AMP | 10-6 |
| Set 3 | |||
| Plate ID | Antibiotics | Dilution | |
| D11 | TRI | 100 | |
| D12 | TRI | 10-1 | |
| D13 | TRI | 10-2 | |
| D14 | TRI | 10-3 | |
| D15 | TRI | 10-4 | |
| T15 | TRI + AMP | 100 | |
| T16 | TRI + AMP | 10-1 | |
| T17 | TRI + AMP | 10-2 | |
| T18 | TRI + AMP | 10-3 | |
| T19 | TRI + AMP | 10-4 | |
| T20 | TRI + AMP | 10-5 | |
| T21 | TRI + AMP | 10-6 |
 "
"