Team:Wash U/Protocol
From 2009.igem.org
(→Recipes) |
(→Tissue Flask Experiment) |
||
| (38 intermediate revisions not shown) | |||
| Line 365: | Line 365: | ||
<font size="4"> | <font size="4"> | ||
=='''Tissue Flask Experiment'''== | =='''Tissue Flask Experiment'''== | ||
| - | <font size="2"> | + | <font size="2">This experiment to monitor the growth and spectra of Rhodobacter shaeroides under anaerobic, photosynthetic conditions was performed as follows:<br> |
| - | : | + | |
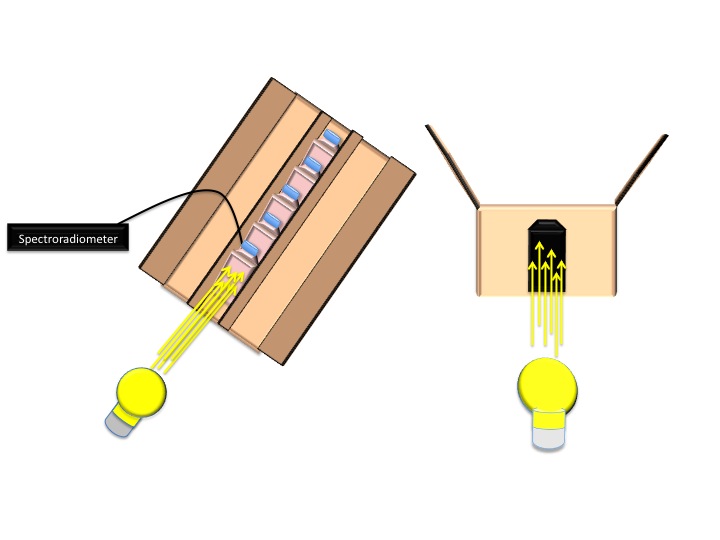
| - | + | -To ensure optimal growth conditions, an Excella E24 incubator shaker was used to maintain a temperature of 34° C and shaking at 160 rpm and the lid of the incubator was propped open 0.5 cm to prevent overheating. The incubator was covered by a black, opaque cloth to prevent external light from influencing culture growth. Tissue flasks were enclosed by 2 growth boxes inside the incubator, which allowed light to enter only through a tissue flask sized opening on the end nearest the light source. These boxes had the dimensions: 34.8 cm x 18.3cm x 16.8cm.<br> | |
| - | + | -5 BD Falcon ™ 50 ml 25 cm² Cell Culture Flasks with blue plug-seal screw caps were used to grow cultures within each box. Cell culture flasks were arranged in series with the first flask recessed 2.5 cm from of box opening and with 2.5 cm separations between each flask. The cell culture flasks were flanked on each side by an opaque barrier so that light incident on each flask was either from the light source, in the case of the 1st flask, or from the light transmittance from the preceding flask in the series<br> | |
| - | + | -A constant light source was centered in front of each of the growth boxes The Light source was a 40 watt incandescent light bulb located 10.8 cm from the growth box opening, as measured from the closest point to the box.<br> | |
| - | + | -Tissue culture flasks were inoculated with R. sphaeroides cultures in the exponential growth phase from a 50 ml inoculation culture grown at 34° C in the dark in M22 liquid media. <br> | |
| - | + | -The test tissue culture flasks were inoculated with Volume = 0.478ml / OD600nm of inoculation culture <br><br> | |
| - | + | [[Image:Innoculation culture growth a.png| 500px | center]]<br> | |
| + | -M22 liquid media was added to flasks for a final volume of 65ml and just after inoculation each flask had OD600nm = 0.011<br> | ||
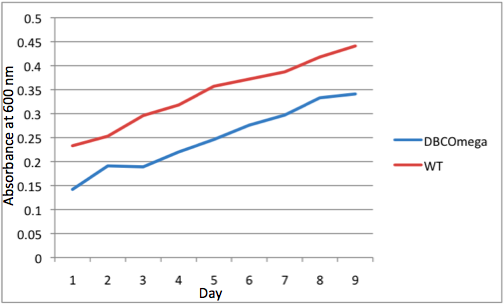
| + | -Cell culture growth was monitored by measuring OD600nm daily at a standard time for each tissue culture flask. To conduct this measurement,1ml of culture was extracted from each flask for measurement and 1ml M22 liquid media was added to each flask to replace the extracted volume and to ensure minimal oxygen exposure to the culture by reducing headroom. <br><br> | ||
| + | -Spectroradiometer readings were taken with an Ocean Optics QP-600-1-UV-Vis EOS25354-2 optic cable transmitting data to an Ocean Optics HR2000CG-UV-NIR High-Resolution Spectrometer. Absolute irradiance and transmittance data were recorded in front of the first flask, then behind this flask and each subsequent flask. The absolute irradiance data were calibrated to a Mikropack HL-2000 Halogen Light Source. The transmittance data were recorded relative to the reading at the front of the first flask. | ||
| + | |||
| + | [[Image:tissue flask.jpg| 700px | center]] | ||
| + | |||
| + | [[Image:tissue flask 0981 b.jpg| 450px | left]][[Image:tissue flask 00973.jpg| 450px | right]] | ||
| + | <br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br> | ||
[https://2009.igem.org/Team:Wash_U/Protocol Back To Top] | [https://2009.igem.org/Team:Wash_U/Protocol Back To Top] | ||
<font size="4"> | <font size="4"> | ||
| + | |||
== '''Recipes''' == | == '''Recipes''' == | ||
<font size="2"> | <font size="2"> | ||
| Line 441: | Line 450: | ||
== '''''R. sphaeroides'' Protocols and Recipes''' == | == '''''R. sphaeroides'' Protocols and Recipes''' == | ||
| - | + | '''Optimal Growth Conditions''' | |
| - | + | <font size="2"> | |
| - | + | ||
| - | ''' | + | |
| - | <font size="2"> | + | |
| - | + | ||
| - | + | ||
| - | + | ||
*M22 Media, anaerobic | *M22 Media, anaerobic | ||
*34º C | *34º C | ||
*Incandescent Light | *Incandescent Light | ||
| - | + | '''Recipes''' | |
| + | <font size="2"><br><br> | ||
| + | '''M22 Growth Media'''<br> | ||
| + | M22 is a Minimal Media that promotes photosynthetic growth. The media is essential for tripaternal mating protocol where it prevents growth of E. coli.<br> | ||
10X Stock: makes up 4 Liters | 10X Stock: makes up 4 Liters | ||
*Potassium dihydrogen orthophosphate KH2PO4 122.4g | *Potassium dihydrogen orthophosphate KH2PO4 122.4g | ||
| Line 464: | Line 470: | ||
*Aspartic acid DL – aspartic acid 1.6g | *Aspartic acid DL – aspartic acid 1.6g | ||
*Solution C 800ml | *Solution C 800ml | ||
| - | |||
Make up to 2-3 litres, pH to 6.8 and then make up to 4 Litres. | Make up to 2-3 litres, pH to 6.8 and then make up to 4 Litres. | ||
Autoclave as 10 X 500ml flats. | Autoclave as 10 X 500ml flats. | ||
| + | 1X M22: | ||
| + | To make up 2 Litres | ||
| + | *10X stock M22 200ml | ||
| + | *CAA 40ml | ||
| + | *Water 1760ml | ||
| - | '''Solution C''' | + | '''Solution C'''<br> |
| - | + | Makes up to 4 Liters | |
| - | Makes up to 4 | + | |
*Nitrilotriacetic acid 40g | *Nitrilotriacetic acid 40g | ||
*Magnesium Chloride MgCl2 96g | *Magnesium Chloride MgCl2 96g | ||
| Line 490: | Line 499: | ||
To make 1 Litre | To make 1 Litre | ||
*Casein Hydrosylate acid 50g | *Casein Hydrosylate acid 50g | ||
| - | |||
Makes up 5% solution to be aliquotted into 200ml. | Makes up 5% solution to be aliquotted into 200ml. | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | + | Batches: | |
| + | *1.5 Liters in 2 Liter flasks | ||
| + | *12 X 80ml in 100ml flasks | ||
| + | *100 X 10ml in universals. | ||
| + | *For M22 agar add 1.5g agar to 100ml of M22 with no CAA in, store in 300ml flats. | ||
| + | '''Vitamin Solution'''<br> | ||
prepare a 10,000 times stock solution of vitamins as follows. | prepare a 10,000 times stock solution of vitamins as follows. | ||
| Line 523: | Line 525: | ||
aliquots you aren't using and keep your working stock in the fridge. | aliquots you aren't using and keep your working stock in the fridge. | ||
| - | + | [https://2009.igem.org/Team:Wash_U/Protocol Back To Top] | |
| Line 551: | Line 553: | ||
6) Allow cells to dry into media for 8hrs at 34º C in the dark<br> | 6) Allow cells to dry into media for 8hrs at 34º C in the dark<br> | ||
7) Scrape dried cells of M22 plate and streak onto M22 step 5ug/ml tet 5ug/ml | 7) Scrape dried cells of M22 plate and streak onto M22 step 5ug/ml tet 5ug/ml | ||
| + | |||
| + | [https://2009.igem.org/Team:Wash_U/Protocol Back To Top] | ||
{{WashUbottom}} | {{WashUbottom}} | ||
Latest revision as of 03:49, 22 October 2009

 "
"