Team:HKU-HKBU/Speed Control Design
From 2009.igem.org
Contents |
Design
Speed control is a crucial feature of our Bactomotor and is indispensable for more advanced controllable applications. Devices equipped with the speed controllable Bactomotor open up many possibilities of non-invasive micro-surgery. Obviously, we may not expect the bacteria motor to behave exactly according to our will. After all, our motor is alive! It is subjected to numerous physical and physiological limitations. But the capacity of tweaking the swimming speed greatly promotes its usefulness. In the case of micro-surgery, we can slow down the Bactomotor in order to locate the pathologic tissue. We can then increase the bacteria to its full speed to bring desirable mechanical changes to the target area. Another example that will illustrate the importance of speed control is in the case of drug delivery. On one hand, we may wish to make the drug-loaded Bactomotor to swim faster than it normally does to overcome the resistance it encounters with in the capillaries during the process of delivery; on the other hand, we wish to slow down the Bactomotor in time to allow more accurate localization of drug.
Escherichia coli or Salmonella typhimurium can swim around by rotating the flagella. When the flagella rotate in a counterclockwise fashion, the bactomotor gathers momentum and produces non-random movement. When the rotation is in the clockwise direction, the bactomotor will tumble in one place and stop 'swimming'.

Speed control is not achieved by a single bacterium; On the contrary, it is the result of the collaborative change in the swimming behavior of a population of bacteria that are attached onto the silicon nano-scale motor via biotin-streptavidin interaction. The aim is achieved by regulation of the expression level of cheZ gene. The gene of cheZ plays the key role here as it controls the phosphorylation level of cheY. CheZ protein can dephosphorylate CheY. High levels of phosphorylation of CheY protein in E. coli or S.typhimurium leads to tumbling movement while low levels of phosphorylation switch the flagella to its non-tumbling mode and enable the bacteria to swim. Therefore, an increase in the expression level of cheZ gene allows us to reduce the tumbling movement, which in turn can increase the swimming speed of the bacteria to achieve manipulation of speed.
Step 1--cheZ knockout
By using λ red recombination system, recombineering was applied to knock out the cheZ gene in the chromosome of E. coli or S.typhimurium. Homologous arms (about 50-bp) were placed inside the cheZ gene. The cheZ gene was substituted by a chloramphenicol resistance gene after recombination.
Step 2--Controllable cheZ expression
An inducible cheZ plasmid ( [http://partsregistry.org/wiki/index.php?title=Part:BBa_K283002 BBa_K283002] ) was tranformed into cheZ knockout strains. Therefore, by controlling cheZ expression level, we can implement the adjustable control over the speed of the bacteria and hence the motor.
There are two designs for cheZ plasmid.
Original Design
The original design ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K283016 BBa_K283016]) is to use LacI as a repressor to prevent the occurrence of leaky expression in the absence of the inducer, which in this case is IPTG (Isopropyl β-D-1-thiogalactopyranoside). We predict that the bacterium will swim at a lower speed when it is in a tumbling mode. When the bacteria are treated with IPTG (switch-on), the expression level of cheZ could be regulated according to inducer's concentration and hence swimming speed of the bacteria.
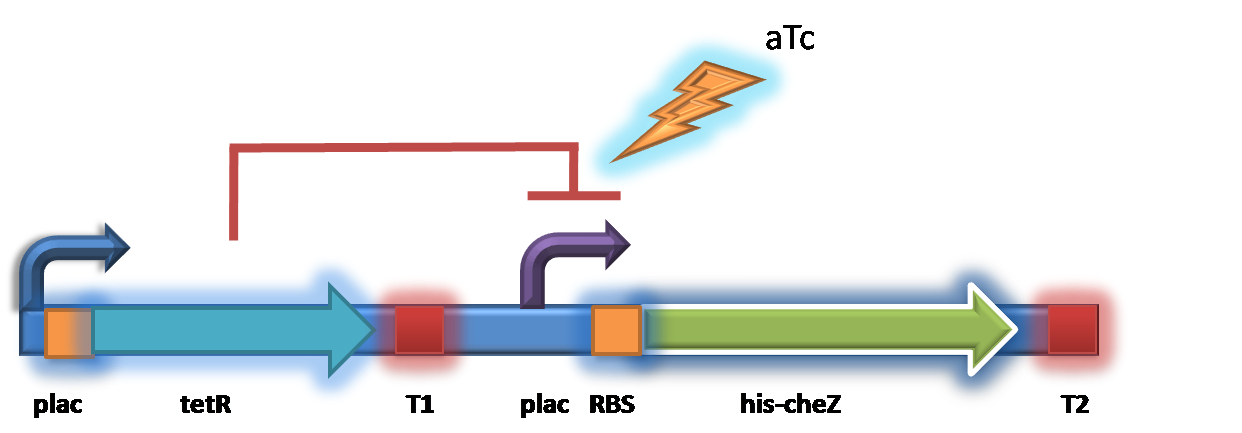
Backup Design
The backup design ( [http://partsregistry.org/wiki/index.php?title=Part:BBa_K283002 BBa_K283002] ) is to use tetR as a repressor and pTet as the regulator, which tetracycline (or aTc)-inducible. We suppose that by changing the concentration of tetracycline, the expression amount of protein CheZ will be altered, resulting in the acceleration and deceleration.
 "
"