Team:British Columbia/Project
From 2009.igem.org
(Difference between revisions)
(→'Overview of the Traffic Light Biosensor: A flexible, modular, and transparent system for multi-level assessment of variable inputs.') |
(→Traffic Light Overview) |
||
| (28 intermediate revisions not shown) | |||
| Line 2: | Line 2: | ||
<!--- The Mission, Experiments ---> | <!--- The Mission, Experiments ---> | ||
| - | == | + | ==Traffic Light Overview== |
| + | Depending on the concentration of a particular substrate in the medium, E. coli will respond accordingly by producing different coloured fluorescence proteins. A diagram would look like this: | ||
| + | [[Image:E_coli_Traffic_Light_Subprojects.png|center|thumb||600px|The ''E. coli'' Traffic Light Biosensor is composed of three major subparts: variable arabinose-inducible promoters, RNA lock and key system, and reverse antisense promoters for input detection, color activation and traffic light switching respectively.]] | ||
| - | + | Here is what's happening inside our traffic light: | |
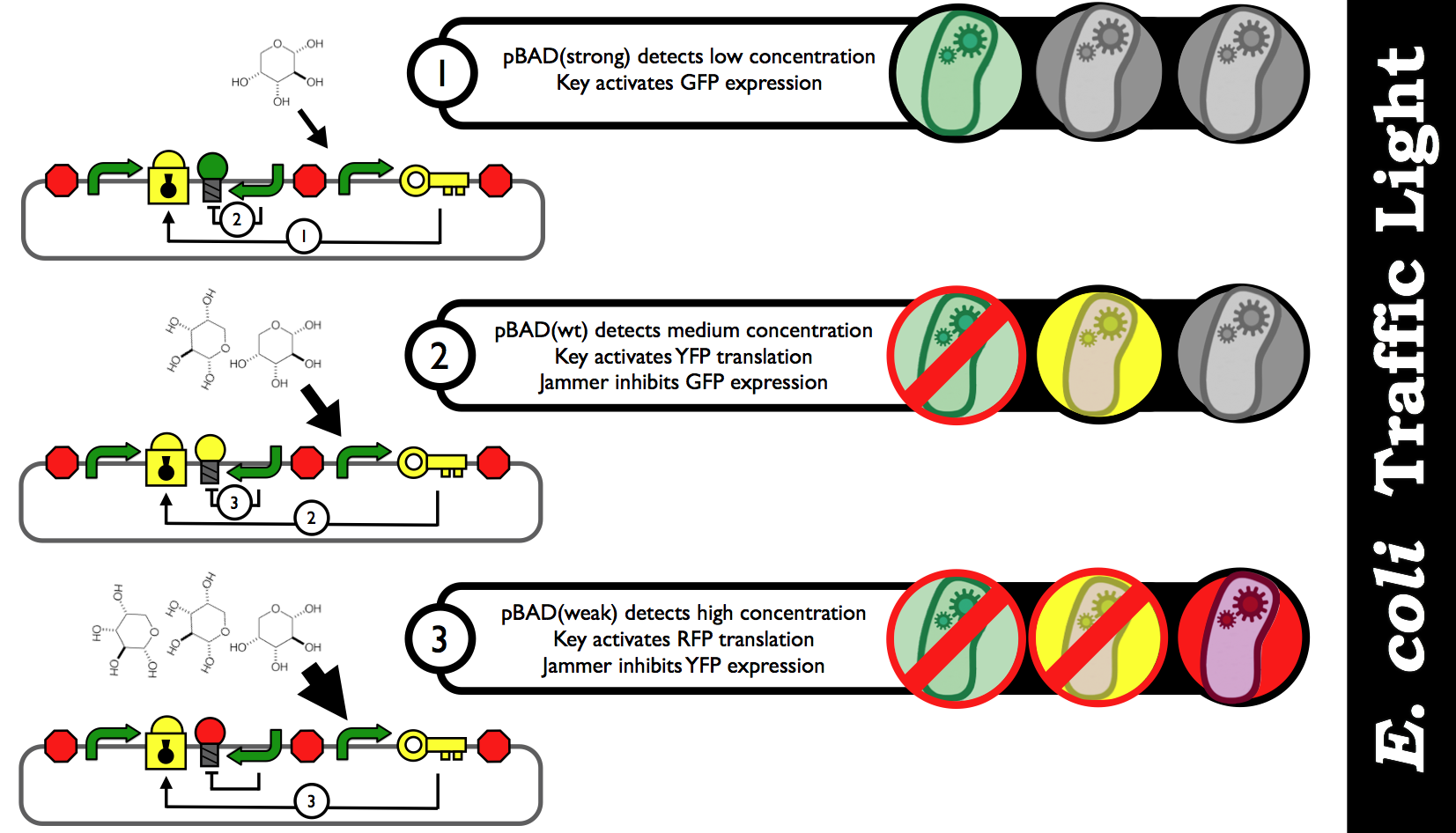
| - | + | [[Image:E_coli_Traffic_Light_Step_by_Step.png|thumb|center|850px|Schematic black-box representation of the E. coli Biosensor that detects various concentration inputs and color outputs. The idea is discrete analog outputs based on a user-specified threshold for each range of concentration.]] | |
| - | + | For our ideas to work, we will need:<br> | |
| - | 1. A variable sensitivity biosensor<br> | + | 1. [https://2009.igem.org/Team:British_Columbia/pBAD A variable sensitivity biosensor]<br> |
| - | 2. A lock-and-key logic gate system | + | 2. [https://2009.igem.org/Team:British_Columbia/LockandKey A lock-and-key logic gate system]]<br> |
| + | 3. [https://2009.igem.org/Team:British_Columbia/Jammer An antisense "off" switch] | ||
<!-- !!!!!!!make sure the spacer is enough so that the image doesn't run into the next section --> | <!-- !!!!!!!make sure the spacer is enough so that the image doesn't run into the next section --> | ||
| - | + | <br><br><br> | |
| - | + | <!-- | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | <!-- | + | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
== Logic Gates == | == Logic Gates == | ||
| Line 38: | Line 29: | ||
[[Image:UBC2009-Key-lock_roadmap.jpg|700x700px|center]] | [[Image:UBC2009-Key-lock_roadmap.jpg|700x700px|center]] | ||
| - | |||
==Lock and Key== | ==Lock and Key== | ||
| Line 54: | Line 44: | ||
As of the 2009 Jamboree, not all aspects of our project have been completed. Looking forward, we intend to complete, test, and characterize our lock, key and jammer. Once this is done we intend to do the same for a second set of lock, key and jammer that have different sequences and will therefore not interfere with the first set. This second set will be configured to detect a second molecule, distinct from the first. When this is completed, we will try adding logic gates, allowing us to address nine different combinations: high, medium and low concentrations of arabinose combined with high, medium and low concentrations of whatever we choose as our second molecule, possibly an antibiotic. When plated on media with two perpendicular gradients, we should be able to independently control each square of a 3x3 grid, possibly displaying 9 different colors of fluorescent proteins, or generating other metabolites, such as indigo. | As of the 2009 Jamboree, not all aspects of our project have been completed. Looking forward, we intend to complete, test, and characterize our lock, key and jammer. Once this is done we intend to do the same for a second set of lock, key and jammer that have different sequences and will therefore not interfere with the first set. This second set will be configured to detect a second molecule, distinct from the first. When this is completed, we will try adding logic gates, allowing us to address nine different combinations: high, medium and low concentrations of arabinose combined with high, medium and low concentrations of whatever we choose as our second molecule, possibly an antibiotic. When plated on media with two perpendicular gradients, we should be able to independently control each square of a 3x3 grid, possibly displaying 9 different colors of fluorescent proteins, or generating other metabolites, such as indigo. | ||
| - | + | --> | |
| - | + | == Tools used and produced == | |
| + | |||
| + | To assist our project, we produced a Biobrick digestion engine and Biobrick picture maker to help out the project: | ||
| - | |||
:http://www.pkts.ca/bb - Biobrick digestion engine - enter the name of a biobrick plasmid and biobrick insert, and this will show you the product of an EcoRI and PstI digestion/ligation as a FASTA file (suitable for viewing in your favorite program). | :http://www.pkts.ca/bb - Biobrick digestion engine - enter the name of a biobrick plasmid and biobrick insert, and this will show you the product of an EcoRI and PstI digestion/ligation as a FASTA file (suitable for viewing in your favorite program). | ||
:http://www.pkts.ca/brickedit/ - Biobrick picture maker - enter a sequence of letters corresponding to the icons, and the program will produce a concatenated file of the Biobrick. | :http://www.pkts.ca/brickedit/ - Biobrick picture maker - enter a sequence of letters corresponding to the icons, and the program will produce a concatenated file of the Biobrick. | ||
| - | = | + | Also, we generated a handy Fasta file containing every biobrick from [http://partsregistry.org/cgi/partsdb/pgroup.cgi?pgroup=List Here]: |
| + | :[[Media:Biobricks.zip|Biobricks.zip]] - Fasta file containing every biobrick | ||
| + | |||
| + | We also found the following tools very helpful: | ||
| - | http://rna.tbi.univie.ac.at/ - a package of prediction tools for RNA structures; we used RNAfold to annotate the key and lock structures | + | :http://rna.tbi.univie.ac.at/ - a package of prediction tools for RNA structures; we used RNAfold to annotate the key and lock structures |
| - | http://mobyle.pasteur.fr/cgi-bin/portal.py - a set of web-accessible bioinformatics tools including Mfold, which determines 2D RNA structure and draws it | + | :http://mobyle.pasteur.fr/cgi-bin/portal.py - a set of web-accessible bioinformatics tools including Mfold, which determines 2D RNA structure and draws it |
| - | http://frodo.wi.mit.edu/ - Primer3, a primer design program | + | :http://frodo.wi.mit.edu/ - Primer3, a primer design program |
Latest revision as of 03:08, 22 October 2009
Traffic Light Overview
Depending on the concentration of a particular substrate in the medium, E. coli will respond accordingly by producing different coloured fluorescence proteins. A diagram would look like this:
Here is what's happening inside our traffic light:
For our ideas to work, we will need:
1. A variable sensitivity biosensor
2. A lock-and-key logic gate system]
3. An antisense "off" switch
Tools used and produced
To assist our project, we produced a Biobrick digestion engine and Biobrick picture maker to help out the project:
- http://www.pkts.ca/bb - Biobrick digestion engine - enter the name of a biobrick plasmid and biobrick insert, and this will show you the product of an EcoRI and PstI digestion/ligation as a FASTA file (suitable for viewing in your favorite program).
- http://www.pkts.ca/brickedit/ - Biobrick picture maker - enter a sequence of letters corresponding to the icons, and the program will produce a concatenated file of the Biobrick.
Also, we generated a handy Fasta file containing every biobrick from Here:
- Biobricks.zip - Fasta file containing every biobrick
We also found the following tools very helpful:
- http://rna.tbi.univie.ac.at/ - a package of prediction tools for RNA structures; we used RNAfold to annotate the key and lock structures
- http://mobyle.pasteur.fr/cgi-bin/portal.py - a set of web-accessible bioinformatics tools including Mfold, which determines 2D RNA structure and draws it
- http://frodo.wi.mit.edu/ - Primer3, a primer design program
 "
"