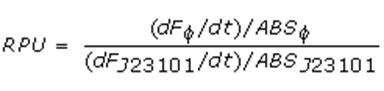Team:UNIPV-Pavia/Parts Characterization
From 2009.igem.org
(→Materials) |
(→BBa_F2620 - 3OC6HSL receiver device) |
||
| Line 273: | Line 273: | ||
==BBa_F2620 - 3OC6HSL receiver device== | ==BBa_F2620 - 3OC6HSL receiver device== | ||
| + | |||
| + | <html> | ||
| + | <table border=1 align="center" cellpadding=5px> | ||
| + | |||
| + | <tr> | ||
| + | <td rowspan=2 valign="bottom" align="center"><b>3OC6-HSL concentration<br></b></td> | ||
| + | <td colspan=2 align="center"><b>LB</b></td> | ||
| + | <td colspan=2 align="center"><b>M9 supplemented</b></td> | ||
| + | </tr> | ||
| + | |||
| + | <tr> | ||
| + | |||
| + | <td><b>Doubling time [minutes]</b></td> | ||
| + | <td><b>RPU</b></td> | ||
| + | <td><b>Doubling time [minutes]</b></td> | ||
| + | <td><b>RPU</b></td> | ||
| + | </tr> | ||
| + | |||
| + | <tr> | ||
| + | <td align="center">0 nM</td> | ||
| + | <td>Doubling time</td> | ||
| + | <td>RPU</td> | ||
| + | <td>Doubling time</td> | ||
| + | <td>RPU</td> | ||
| + | </tr> | ||
| + | |||
| + | <tr> | ||
| + | <td align="center">0.1 nM</td> | ||
| + | <td>Doubling time</td> | ||
| + | <td>RPU</td> | ||
| + | <td>Doubling time</td> | ||
| + | <td>RPU</td> | ||
| + | </tr> | ||
| + | |||
| + | <tr> | ||
| + | <td align="center">1 nM</td> | ||
| + | <td>Doubling time</td> | ||
| + | <td>RPU</td> | ||
| + | <td>Doubling time</td> | ||
| + | <td>RPU</td> | ||
| + | </tr> | ||
| + | |||
| + | <tr> | ||
| + | <td align="center">10 nM</td> | ||
| + | <td>Doubling time</td> | ||
| + | <td>RPU</td> | ||
| + | <td>Doubling time</td> | ||
| + | <td>RPU</td> | ||
| + | </tr> | ||
| + | |||
| + | <tr> | ||
| + | <td align="center">100 nM</td> | ||
| + | <td>Doubling time</td> | ||
| + | <td>RPU</td> | ||
| + | <td>Doubling time</td> | ||
| + | <td>RPU</td> | ||
| + | </tr> | ||
| + | |||
| + | <tr> | ||
| + | <td align="center">1 uM</td> | ||
| + | <td>Doubling time</td> | ||
| + | <td>RPU</td> | ||
| + | <td>Doubling time</td> | ||
| + | <td>RPU</td> | ||
| + | </tr> | ||
| + | |||
| + | <tr> | ||
| + | <td align="center">10 uM</td> | ||
| + | <td>Doubling time</td> | ||
| + | <td>RPU</td> | ||
| + | <td>Doubling time</td> | ||
| + | <td>RPU</td> | ||
| + | </tr> | ||
| + | |||
| + | </table> | ||
| + | </html> | ||
| + | |||
<div align="right"> | <div align="right"> | ||
Revision as of 11:35, 19 October 2009

|
|
|
Parts Characterization |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Here we describe the characterization results of 4 parts of our own design, 2 existing parts re-built because they were inconsistent and 7 existing parts taken from the Registry. When not reported differently, all the experiments have been performed according to Growth conditions and Data analysis sections.
Re-built existing parts (BBa_our part code/BBa_existing part code): Existing parts from the Registry:
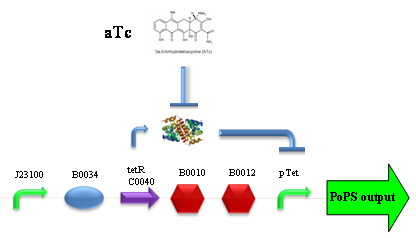
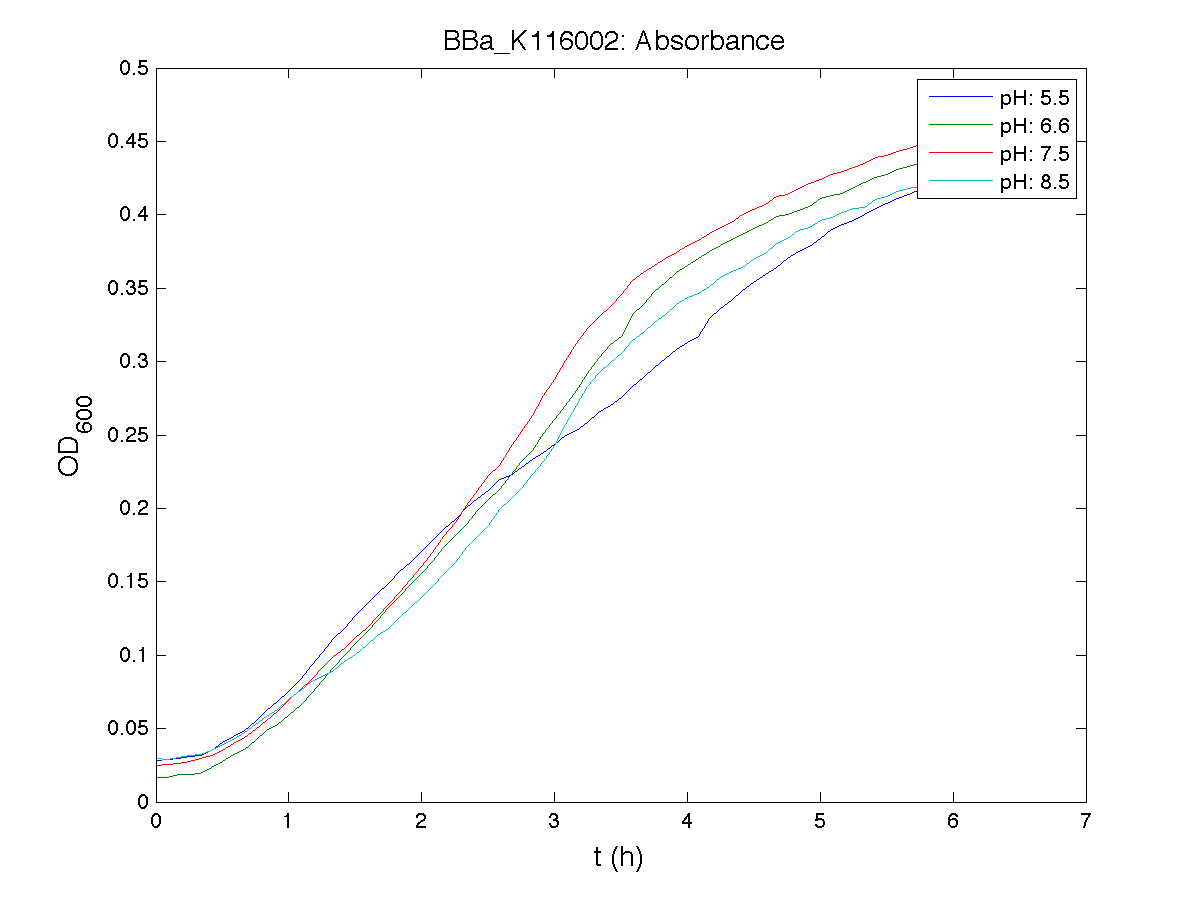
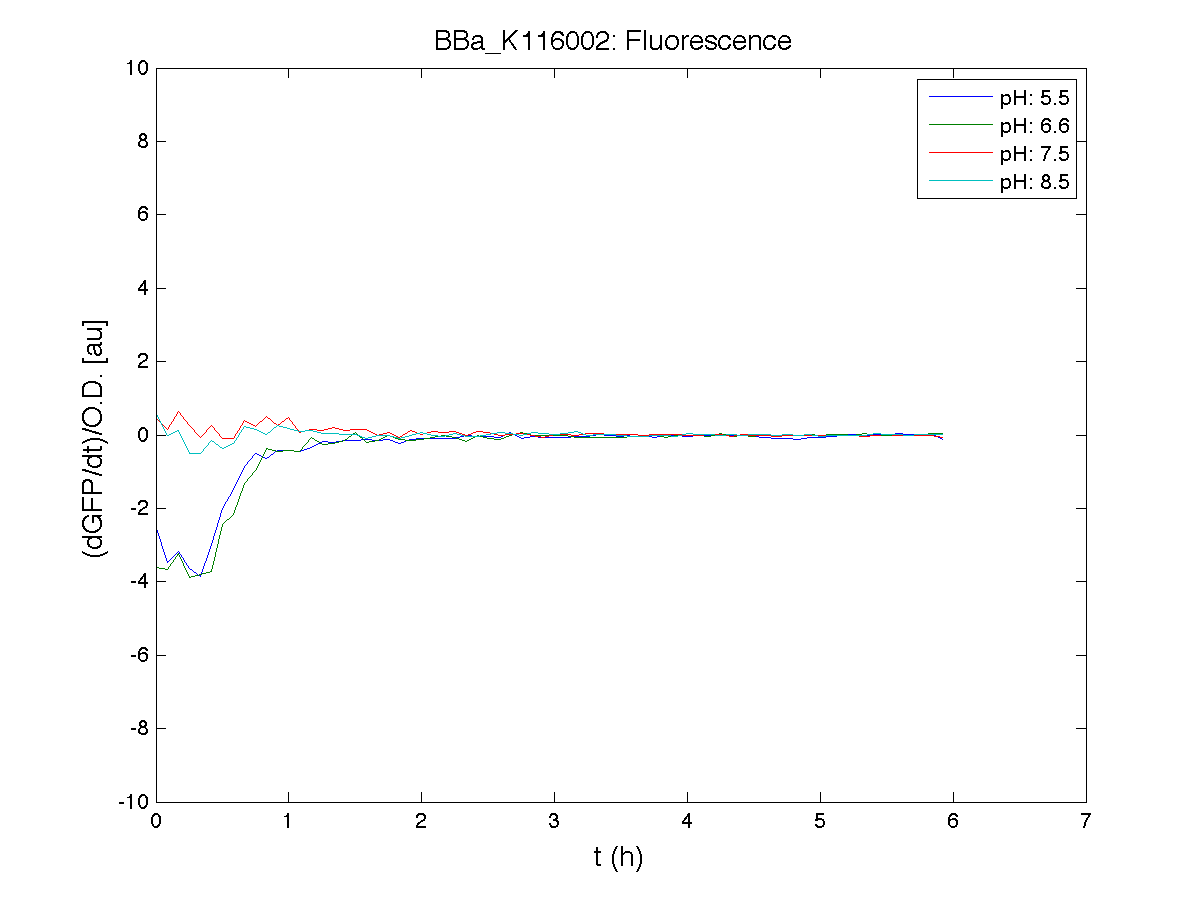
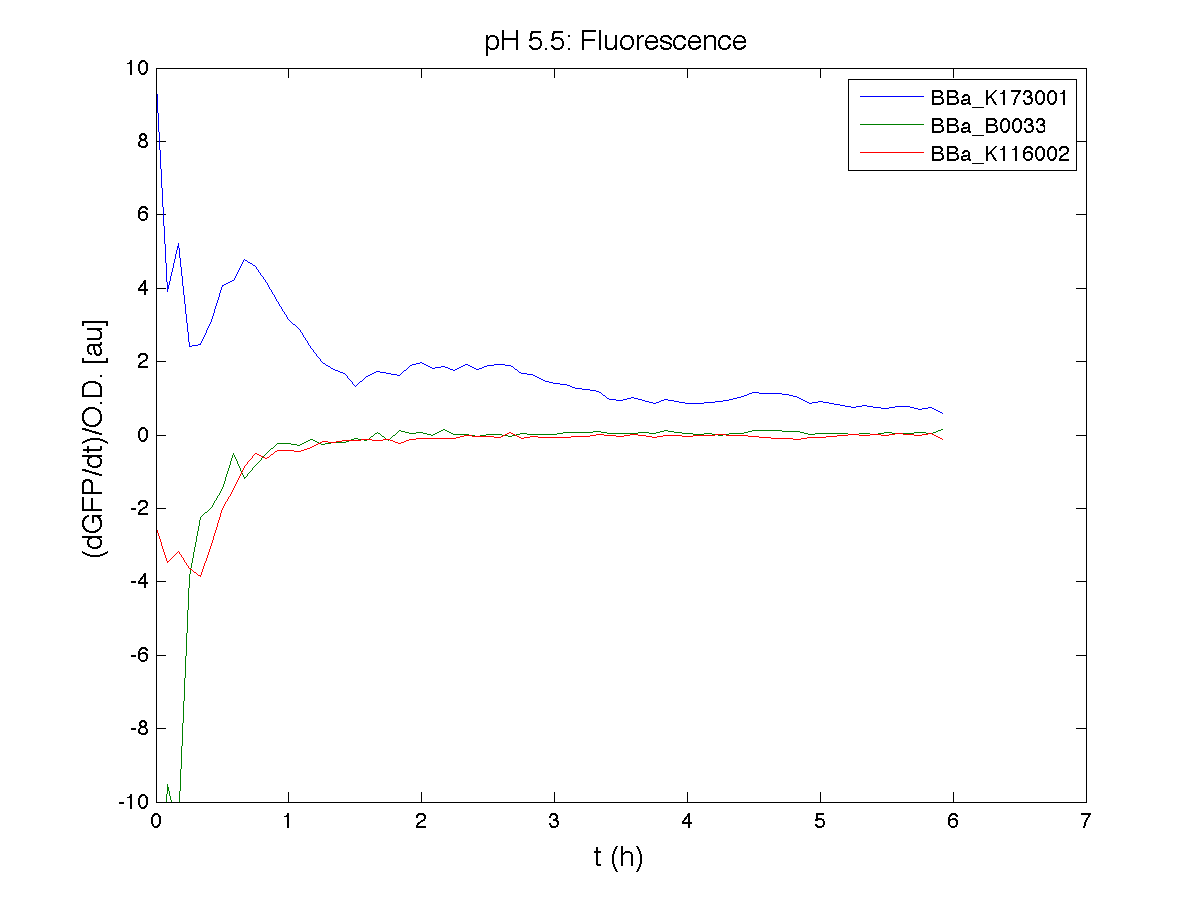
Our new partsBBa_K173003 - ethanol producing deviceBBa_K173007 - aTc inducible device with J23100 promoterDescriptionThis is an aTc sensing device. BBa_J23100 promoter drives the constitutive production of tetR repressor (BBa_C0040), which inhibits tetR promoter (BBa_R0040) activity. When aTc is added to the medium, it binds tetR and inhibits it. So, the PoPS output is a function of the aTc concentration. A tight regulation is expected for this inducible system because BBa_J23100 is a strong promoter and so tetR repressor shold be produced at extremely high levels. Characterization
ConclusionsWe demonstrated that this part works as expected, sensing the aTc concentration provided in the culture medium. The transfer function of this sensor has been characterized in standard units (RPUs) in two different growth media (LB and M9 supplemented with glycerol), as well as the metabolic burden (in terms of doubling time) which affects E. coli bearing this part. On the other hand, we did not expect to have a higher GFP synthesis rate per cell after the exponential growth phase than in the exponential phase itself (as reported in the 3rd plot). BBa_K173011 - aTc inducible device with J23118 promoterDescriptionThis is an aTc sensing device. BBa_J23118 promoter drives the constitutive production of tetR repressor (BBa_C0040), which inhibits tetR promoter (BBa_R0040) activity. When aTc is added to the medium, it binds tetR and inhibits it. So, the PoPS output is a function of the aTc concentration. A less tight regulation is expected for this inducible system than in BBa_K173007 because BBa_J23118 promoter is weaker than BBa_J23100 and so tetR repressor shold be produced at lower levels than in the other sensor. Characterization
ConclusionsBBa_K173010 - lactose/IPTG inducible device with J23118 promoterRe-built existing partsBBa_K173004/BBa_I732019 - beta-galactosidase protein generatorBBa_K173005/BBa_Q04400 - tetR QPIExisting parts from the RegistryBBa_J23100, BBa_J23101, BBa_J23118 - constitutive promoter family membersBBa_F2620 - 3OC6HSL receiver device
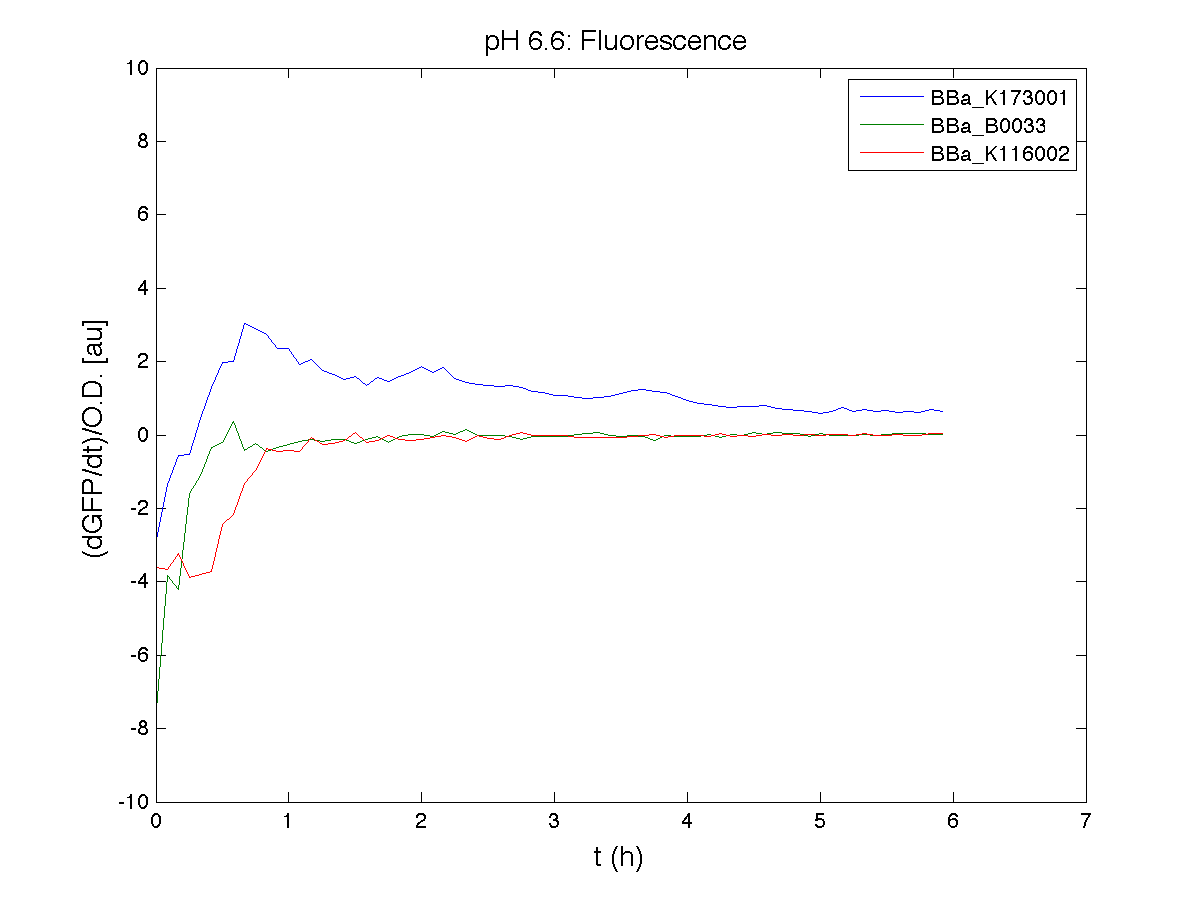
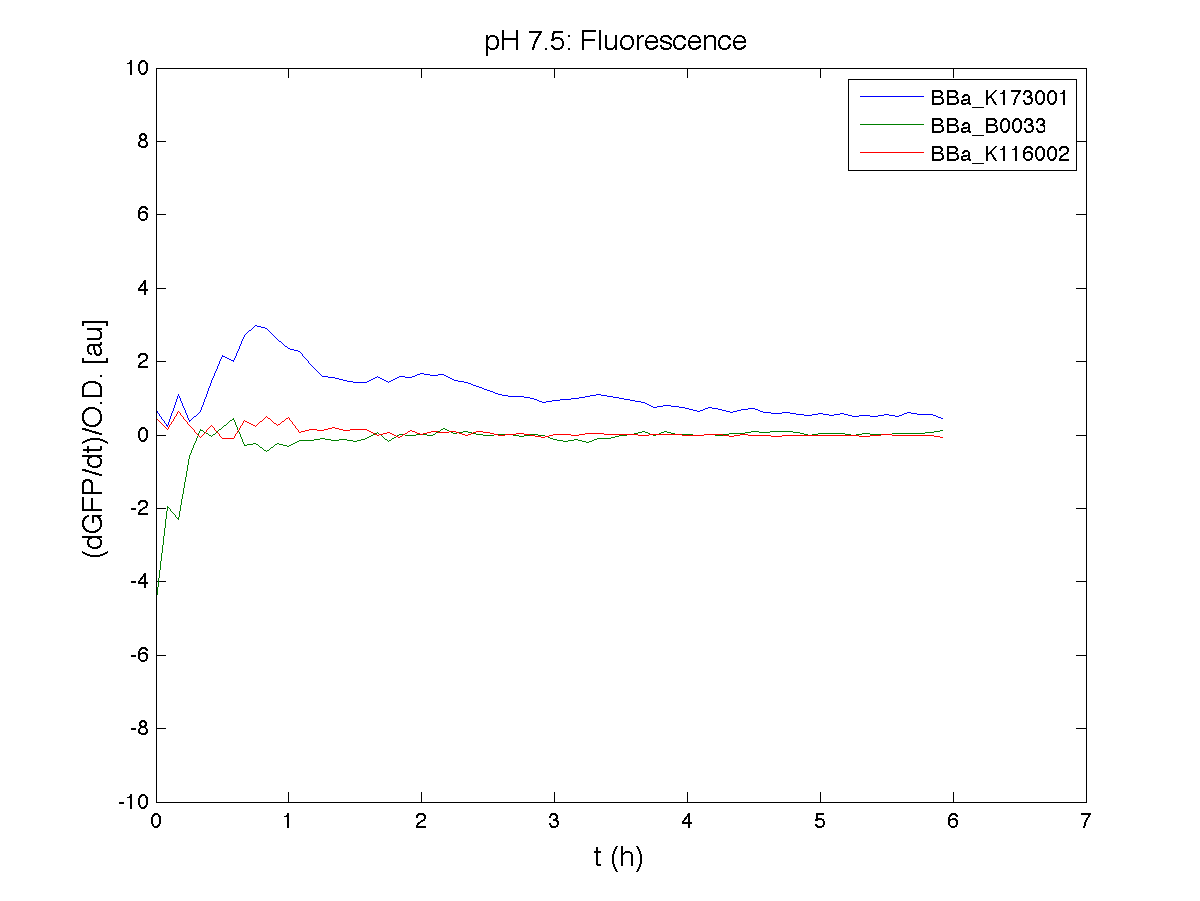
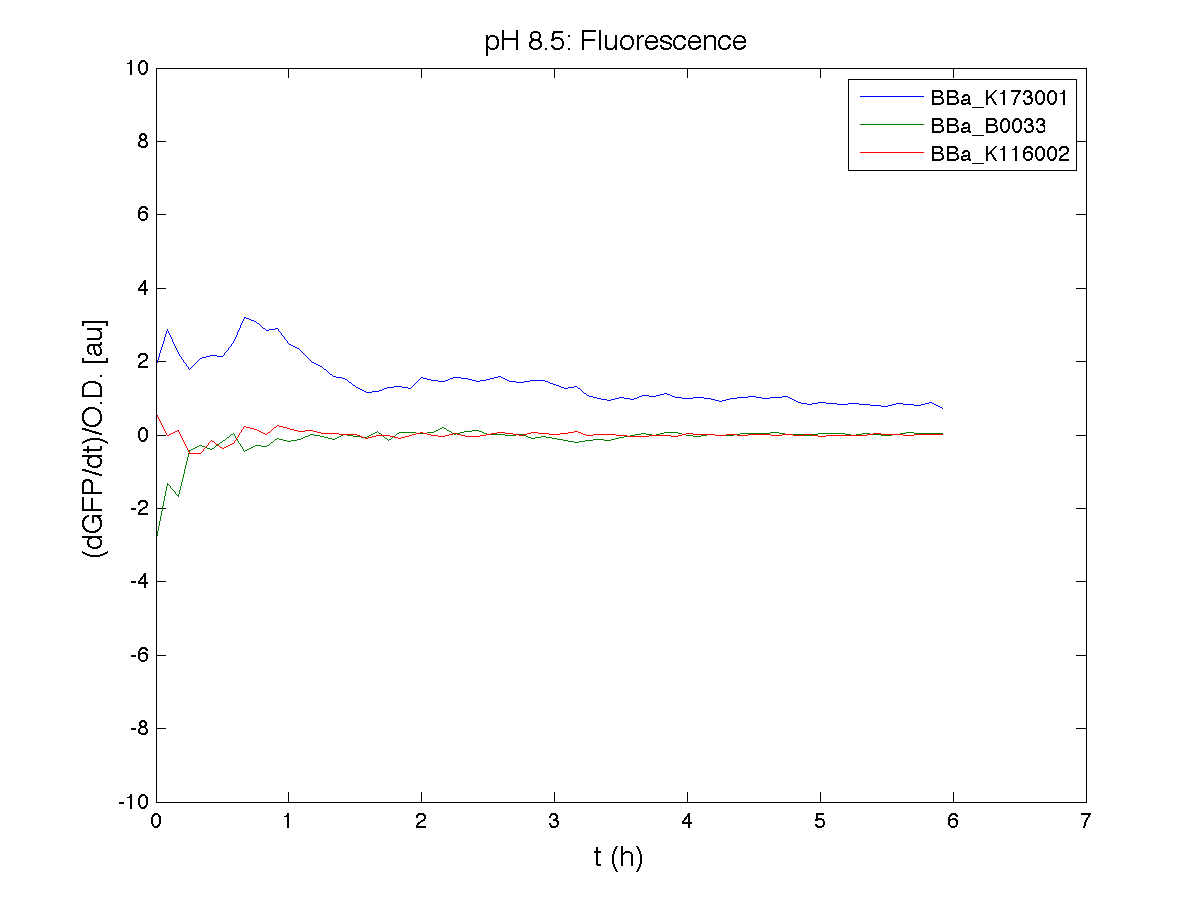
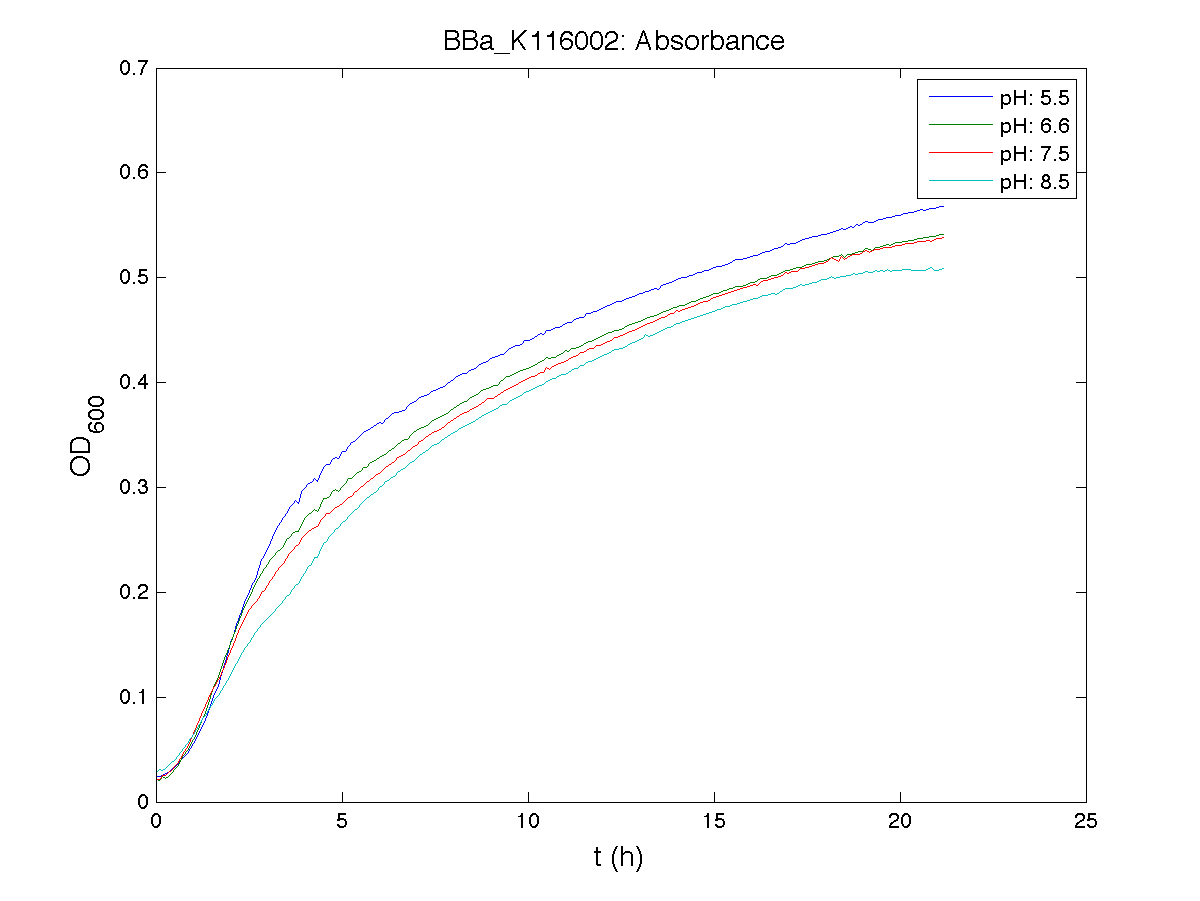
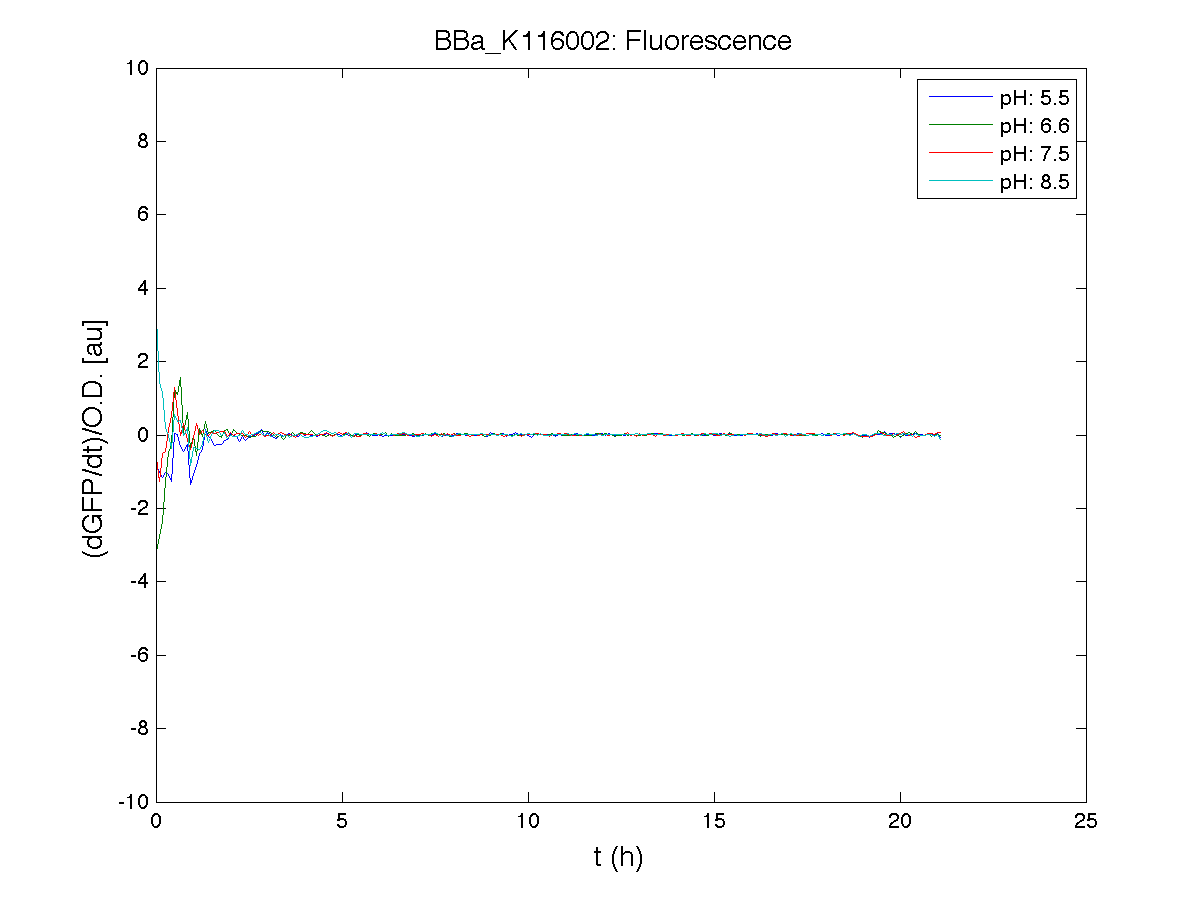
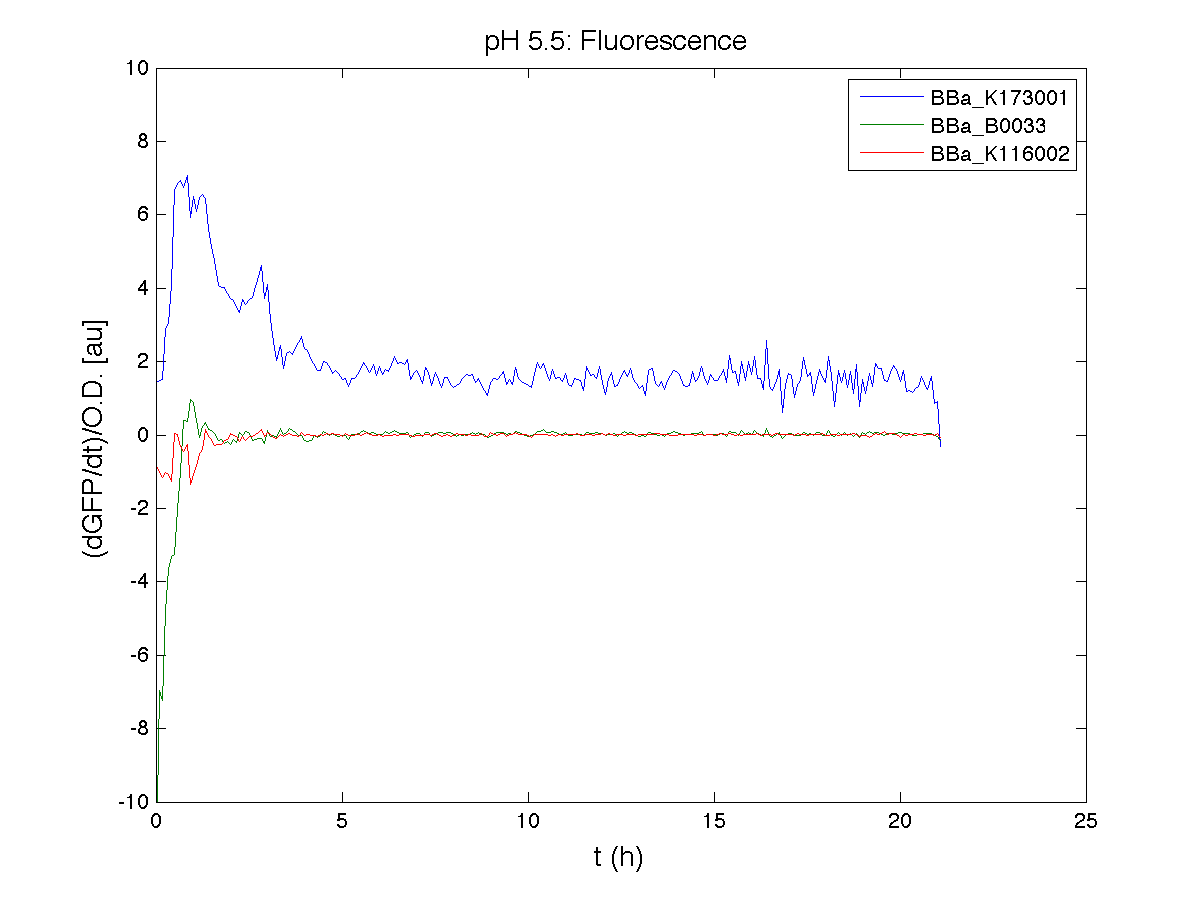
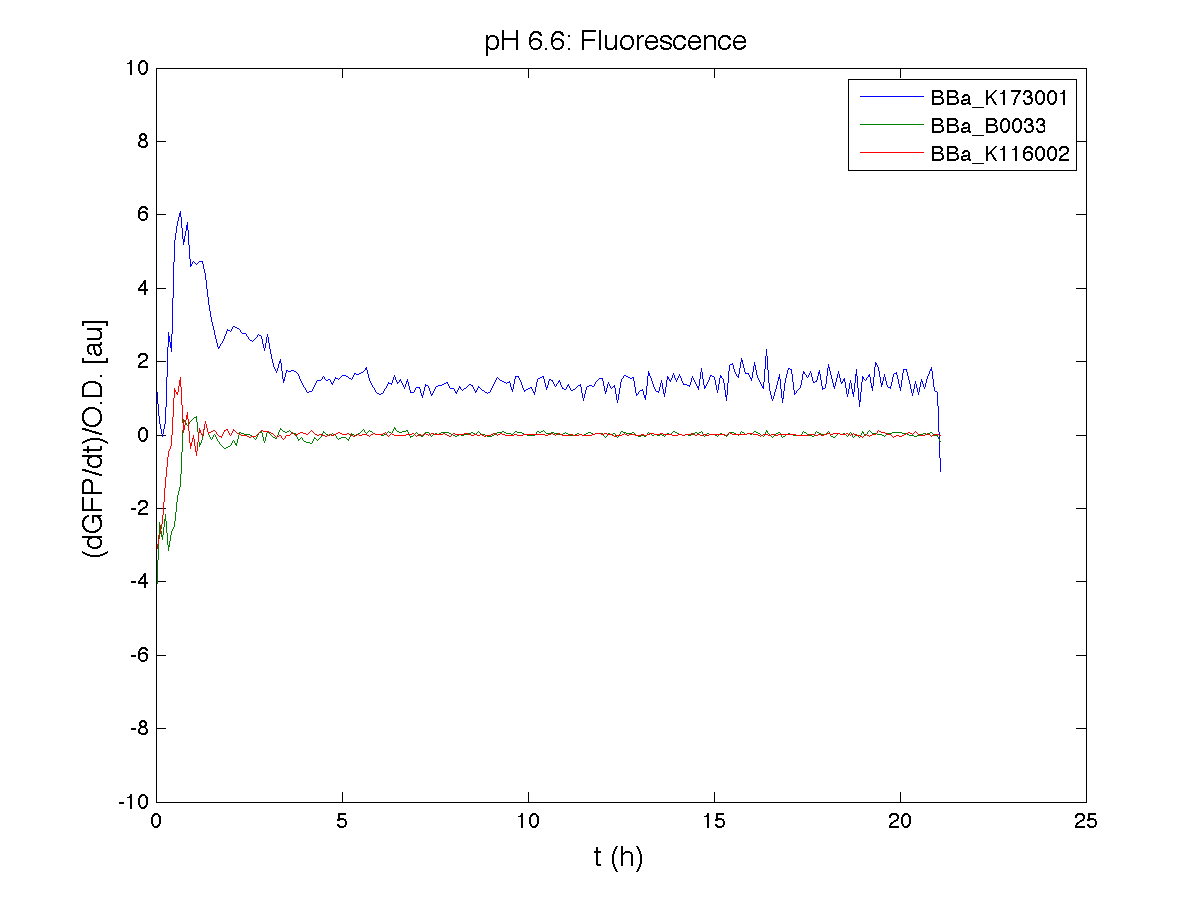
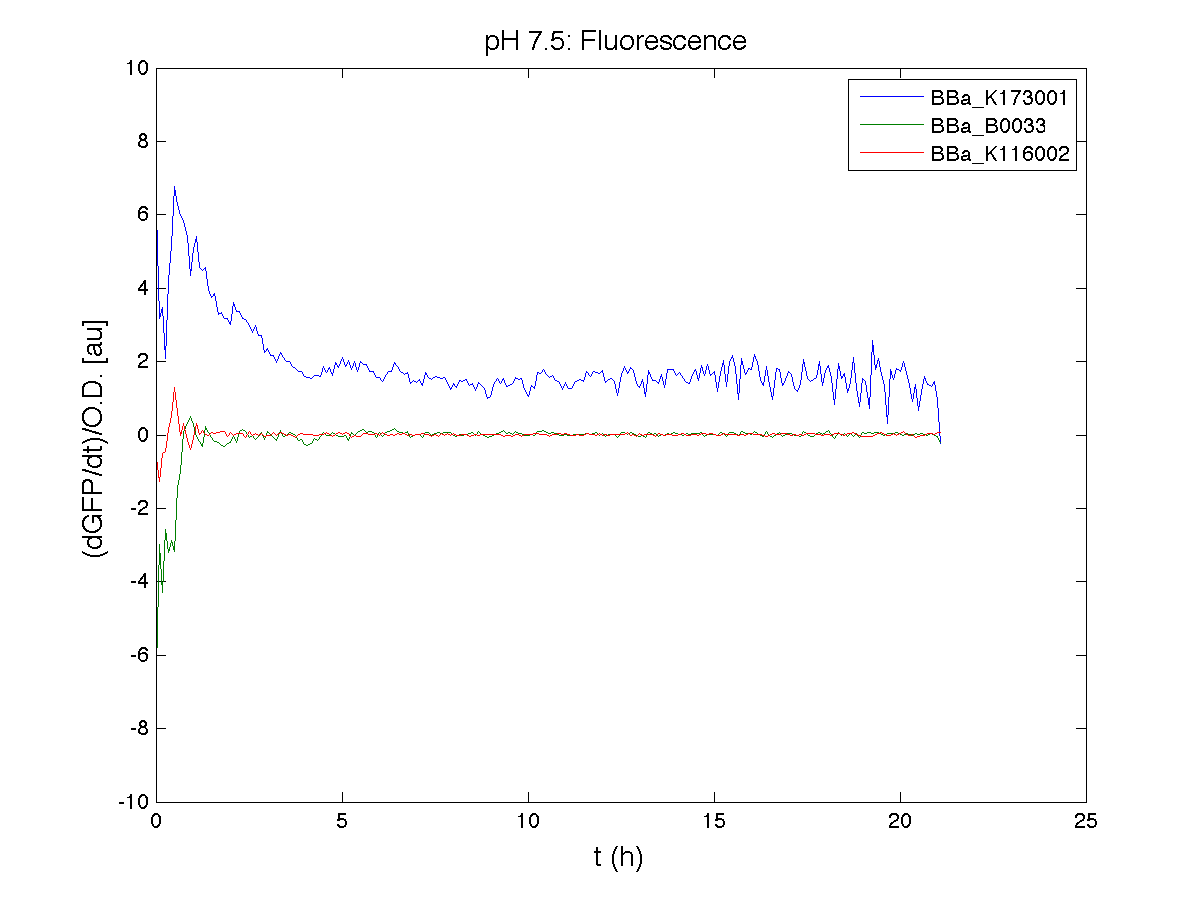
BBa_K116001 - nhaA promoterBBa_K116001 from iGEM 2008 NYMU-Taipei We received this BioBrick from iGEM in September but the bacterial strain that contained the plasmid wasn't declared. So we decided to sequence it as check and transform it into E.coli TOP10. We wanted to perform some experiments to better understand how it works and if can be successfully used. We performed several experiments with different LB medium and we got almost the same results. We used:
Here we show just two experiments to explicate our work. You can download the complete list from this link. Experiment Na+ 0MMotivationIn our opinion the working principle of the antiporter Na+/H+ channel described in [Rachel Karpel et al., Etana Padan et al., N. Dover et al.] makes the nhaA promoter a Na+ sensor and only under certain conditions (presence of Na+) a pH sensor. Methods
ResultsCommentsAs expected BBa_K116001 didn't produce any GFP. So we can consider it a Na+ sensor and only secondarily a pH sensor. Experiment Na+ 250mMMotivationWe’ll try again to make E.coli producing GFP at the variation of pH. Methods
ResultsCommentsWe didn't expect this. After looking better for a motivation in some articles ([Rachel Karpel et al.]) we think this could be because of the E.coli strain: we use TOP10 while a special strain (delta-pump) without some membrane proteins that regulate E.coli homeostasis is used in other experiments. Final considerationsIn our opinion this sensor (primarily sodium sensor and secondarily pH sensor) needs very particular conditions to work (first of all a specific bacterial strain) we couldn’t reproduce, so we consider it almost unusable. BBa_K112808 - Enterobacteria phage T4 Lysis DeviceBBa_R0011 - Plac hybrid promoterGrowth conditionsFor every culture we tested:
Data analysisGrowth curvesThe presented growth curves have all been processed as OD600_culture-OD600_broth for each time sample. OD600_broth is the medium in the same conditions as in the culture (e.g. induced with the same inducer concentration as in the culture). Doubling timeThe natural logarithm of the growth curves (processed according to the above section) was computed and the linear phase (corresponding to the bacterial exponential growth phase) was isolated by visual inspection. Then the linear regression was performed in order to estimate the slope of the line m. Finally the doubling time was estimated as d=ln(2)/m [minutes]. In the case of multiple growth curves for a strain, the mean value of the processed curves was computed for each time sample and then this procedure was performed. Relative Promoter Units (RPUs)The RPUs are standard units proposed by Kelly J. et al., 2008, in which the transcriptional strength of a promoter can be measured using a reference standard, just like the ground in electric circuits. RPUs have been computed as: in which:
RPU measurement has the following advantages:
The hypotheses on which RPU theory is based can be found in Kelly J. et al., 2008, as well as all the mathematical steps. From our point of view, the main hypotheses to satisfy are the following:
Materials
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"