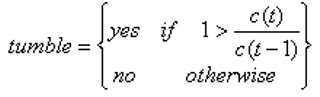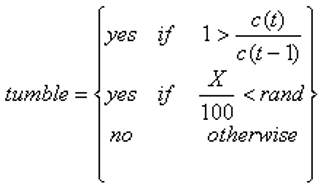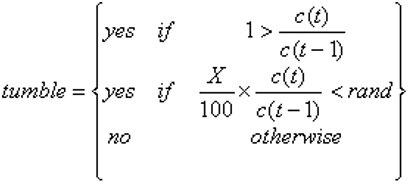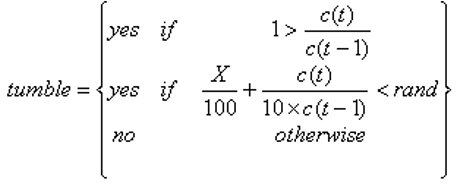Team:Aberdeen Scotland/chemotaxis
From 2009.igem.org
University of Aberdeen - Pico Plumber
Contents |
Chemotaxis Model
The above embedded video is of bacteria chemotaxing towards a source of aspartate in the form of a crack from -300 to 300 in the view along the pipe axis. The units are in microns.
Introduction
When bacteria move in reaction to a chemical this is chemotaxis. A model of this is important to us, as the Pico-Plumber will consist of a colony of bacteria dispersed in water that swim towards potential leaks marked by chemicals. The aim of our chemotaxis model is to simulate how bacteria respond to a source of chemoattractant diffusing through a virtual pipe from a breach in the pipe inner wall. Using this simulation we can ascertain roughly how long it takes bacteria to swim towards a leak in the pipe, and from this, the required density of bacteria in the pipe to seal a hole of known diameter in a given time. A model of thousands of bacteria, each independently moving, is also useful in modelling how populations of interdependent gene regulatory networks behave. It is our aim to integrate a chemotaxis model and multiple gene network simulation together in order to check for any emergent behaviour and to see if the system will work.
E.coli is a motile strain of bacteria, which is to say it can swim. It is able to do so by rotating its flagellum, which is a rotating tentacle like structure on the outside of the cell. Chemotaxis is the movement up concentration gradients of chemical attractants (usually a food source) and away from poisons. E.coli is too small to be able to detect any chemical gradient between the two ends of itself, and so must head in any random direction and then compare the new concentration at the new point to the previous one. Its motion is described by ‘runs’ and ‘tumbles’. If the concentration of food molecules has increased after a run in a certain direction, the bacteria will be unlikely to tumble (which is to change direction) - and if it has decreased, then the likelihood of trying a new direction is high. This motion occurs by altering the direction of flagellar rotation. When the bacterium is on a run it rotates its flagellum counter-clockwise, and when it is tumbling, it rotates the flagellum clockwise. Bacteria always respond to a chemoattractant gradient within a range of values, so that there is a minimum and maximum concentration between which bacteria chemotax.
Designing a Model
Chemotaxis in Escherichia Coli is very well studied and there is a multitude of papers about it - each with different data and parameters. In order to construct a useful model we need reliable - and if possible - accurate data. The paper ‘Motility of Escherichia coli cells in clusters formed by chemotactic aggregation’ [2] is where most of the data used in the model comes from. Another older paper, ‘Chemotaxis in Bacteria’ by Julius Adler [1], examined how the speed of bacteria during each ‘run’ varies randomly around an average value. The paper did not offer a mathematical approximation of this random variation; it simply reported their experimental results and discussed them. Therefore to mimic this variation a random number at each ‘tumble’ is added to a minimal speed such that the average value of this random speed is the average speed of the bacteria. For example, the average swimming speed according to our available data is 27 micrometres per second [3], which in the program is represented by a speed of 25 micrometres per second plus a uniformly distributed random number between zero and four. Modelling E. Coli’s behaviour as it swims up a chemotactic gradient is of course limited by inaccurate and unreliable data. The tumbling frequency for example, according to some papers in the seventies, is reported to be as high as several Hertz, whereas more recent papers have one Hertz being the maximum tumbling frequency. One paper, ‘Temporal comparisons in bacterial chemotaxis’ [4], informs of the fact that bacteria actually look as far back in time when swimming as 4 seconds, but usually take the previous second as their basis on deciding whether or not concentration has increased or not. It is for this reason we have chosen one Hertz to be the maximum rate at which bacteria tumble when moving down a chemoattractant gradient.
Although for some specific strains of E.coli the motion has been very well studied, the actual strain used for our project may behave differently, as they vary in motility. An example of this would be that some strains of E.coli swim as fast as 50 micrometres per second. Another example would also be that some mutant strains of E.coli tumble very infrequently and can check concentrations as infrequently as every 30 seconds. The only way to ensure accuracy and reliability in simulating how our bacteria behave is to actually experiment with our own strain and compare this to the model. The model can then be altered to fit the data, or we could attempt a more in-depth analysis to try and find the tumbling frequency behaviour and swimming speed of our own strain.
In order to design a simulation, some simplifications have to be made. In fact, our model has more simplifications than some models in literature. This chemotaxis model, for instance, neglects the fact that E.coli do not travel in straight lines but often take curved paths due to unequal firing of the flagella. Moreover there almost always is a slight curvature due to Brownian motion in the fluid. In a static fluid there are usually minute convection currents due to local temperature variations. These currents can transport bacteria against their own will, as we have seen for ourselves under the microscope. The model also does not consider the fact that E.coli change in size, and regarding the collisions the bacteria are simply modelled as hard spheres of 2 micrometres diameter that cannot overlap. The collisions simply result in a change of direction and there is no conservation of momentum or kinetic energy, or modelling of the physics of the collision. The tendency of bacteria to congregate into small groups by quorum sensing is also ignored.
Modelling Behaviour
How the tumbling frequency varies as the bacteria move up and down a chemical gradient is hard to define and model exactly. One paper suggests that the frequency of tumbling is roughly one Hertz, and decreases to almost zero when moving up a chemotactic gradient. To attempt to model this we have greatly simplified the behaviour. Our simplifications however, are not as extensive as some models which simply have a probability of 0% of tumbling when moving up a chemotactic gradient.
Firstly, as an introduction to how the model works, we must understand how the bacteria decide to tumble when moving away from a source of chemoattractant. At every second, which is every third iteration, the bacteria compare the current concentration at that point to the concentration at the previous point, and if it has decreased then the bacteria tumbles:
In the previous described motion the bacteria will seek out the source of chemoattractant very well. However bacteria do still tumble when moving up a concentration gradient, and so this must be accounted for. The most simple way of describing this is giving the bacteria a probability of X% (with X < 100) of not tumbling when moving up a gradient and 100% when moving down a gradient described by the statement:
However, it is known that the behaviour varies depending on the difference in concentration between the current and the previous locations. If the ratio of these concentrations is greater than one, then the probability of not tumbling is roughly between 60% and 80%, expressed by either the following possible statements:
In order to experiment and to look at how various functions cause varying behaviour, we used many different functions like above for the probability of not tumbling. To illustrate these different behaviours the embedded video below shows bacteria that are given one hundred seconds to travel 8000 micrometers towards a source of chemoattractant - although the speed with which each bacterium moves is multiplied by six for the sake of making it easier to see them move:
In the above video, the colours correspond to the probability of not tumbling as follows (where p(t) = c(t)/c(t-1) and a probability of 1 = 100% and 0 = 0%): Green = 0.6, Cyan = (0.6+(p(t)/10)), Red = 0.6*p(t), Black = 0.6*p(t) + (p1(t)/10), Blue = 0.6*(p(t)^3).
As it can be seen in the video, the speed at which the bacteria travel is very similar. The red and green colonies, however, do not congregate as tightly around the source of food as the others do.
All the data and probabilities of tumbling when moving up a chemotactic gradient have been chosen without scientific grounding except educated guess-work after reading through the literature. If actual data could be obtained about our strain of bacteria, the parameters could be tuned to fit experimental findings. Therefore, another useful comparison is to look at how the bacteria move depending on the basic probability of not tumbling, chosen originally as 60% in our model. In the embedded video below the following colours have the respective probabilities of not tumbling: Green = 40%, Cyan = 50%, Red = 60%, Black = 75%, Blue = 90%.
As it can be seen, the blue colony finds the source far better than the others. The green and cyan colonies, however, find it hard to even locate the source in the given time. The red colony, which corresponds to our choice, does show some bacteria unable to find the source quickly. In our experience of watching E.coli under the microscope, however, some bacteria do take more time to find the source than others. Exactly which probability to use here is not an easy choice, although through experiments in the lab we hope to be able to decide upon a value with confidence.
Another parameter is how often the bacteria decide to check the concentration gradient. Last year's University of Bristol team used the figure of bacteria checking their concentration every three seconds, whereas after reading the literature and comparing papers we have decided to use a figure of once per second. This is because the tumbling rate in other papers seemed to be around one Hertz, or sometimes higher, and if they only checked their concentration every three seconds this level of tumbling could never occur. In the embedded video below the following colours correspond to the following times for how often the concentration is checked: Green = 3 seconds, Cyan = 2 seconds, Red = 1 second, Black = 2/3 seconds, Blue = 1/3 seconds.
As the bacteria are attracted to a chemical, in our case aspartate ions - which diffuse from a source into the surrounding water - modelling the diffusion is also important. As the diffusion in all our cases is from what can be considered a point source and equal in all radial directions, the following solution to Fick’s Law can be used:
Where x is the radial distance from the source, t is the time since diffusion began and erfc is the complementary error function, D is the coefficient of diffusion and C(0) is the concentration at the source at time zero. The diffusion coefficient of the aspartate ion, or aspartic acid, was calculated from [5] as the diffusion coefficients of the amino acids correlate strongly to their molecular mass. The concentration at the source is assumed to be the saturation point of aspatic acid, as the boundary layer of water closest to the dissolving solid would be saturated.
In the chemotaxis model this is used to calculate the concentration at any time in any location. Here however, is where another series of simplifications and assumptions are made. The above solution of Fick’s Law is for a static fluid in which there exist no eddy currents. As there will be small temperature fluctuations there will also be convection currents mixing the fluid, and as the bacteria themselves swim around they will be further mixing the fluid. Estimating the eddy diffusion coefficient is very difficult to do accurately. For the purposes of the chemotaxis model in most cases eddy diffusion has been ignored as the fluid is for all intensive purposes static. However; in the case where we were performing experiments, a greater level of turbulence and mixing exists as samples are taken from a channel of water every five minutes.
E.coli is only capable of sensing our signalling chemical, aspartate, at levels above 10-8 M according to ‘Chemotaxis in Bacteria’ [1]. This has been accounted for in the model and can be shown by the video below in which aspartate is diffusing out (without eddy diffusion and with data from [5]) from a source in the middle of the bacteria. The black circle represents the point where the concentration is 10-8 M, and everything inside it is greater than 10-8 M:
Our model, unlike many chemotaxis models, is not agent based, and so collision behaviour of correctly shaped E. Coli cells has not been included. In our 2-d model tumbling of bacteria due to collisions and stopping of overlap are included. However if we implement this effect and count how many bacterial collisions occur, the frequency is surprisingly low. The model which detects collisions and changes the behaviour is extremely computationally intensive for MATLAB when dealing with thousands of bacteria and has little impact on the behaviour. The model is mostly concerned with how long it takes E. Coli to travel through mostly empty space to our target source of chemoattractant, and not how inter-bacterial collisions affect a tight quorum of bacteria.
 Back to PDEs/Next Steps Back to PDEs/Next Steps
|
Continue to Dissociation Constants 
|
References
[1] Julius Adler. “Chemotaxis in Bacteria”. Annu. Rev. Biochem. 1975.44:341-356.
[2] Nikhil Mittal, Elena O. Budrene, Michael P. Brenner, and Alexander van Oudenaarden. “Motility of Escherichia coli cells in clusters formed by chemotactic aggregation”. PNAS. Vol 100. no 23. Nov 11, 2003.
[3] Kayo Maeda, Yasuo Imae, Jun-Ichi Shioi, and Fumio Oosawa. “Effect of Temperature on Motility and Chemotaxis of Escherichia coli”. JOURNAL OF BACTERIOLOGY, Vol. 127, No. 3 Sept. 1976, p. 1039-1046
[4] Jeffrey E. Segall, Steven M. Block, and Howard C. Berg. “Temporal comparisons in bacterial chemotaxis”. Proc. Nati. Acad. Sci. USA Biophysics Vol. 83, pp. 8987-8991, December 1986
[5] Alfred Polson. “On The Diffusion Constants of the Amino-Acids”. Institute of Physical Chemistry, The University of Upsala, Sweden. 20 August 1937.
 "
"