Team:BCCS-Bristol/Bioscaffold/Protein Fusion Problem
From 2009.igem.org
iGEM 2009
Protein Fusion Problems
Each gene that encodes a protein has to have two stop codons at its 3' end (5'-TAATAA-3'). Moreover when two genes are fused together an additional third stop codon is created within the DNA scar (DNA region that connects the two genes). Hence this would prevent the expression of the second partner in the fusion protein.
Existing Solutions
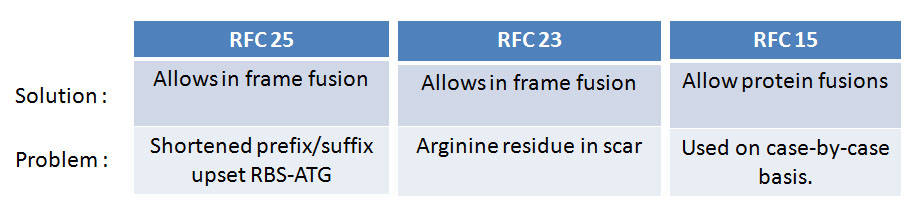
The current iGEM assembly standards do not fully support protein fusions.
Current assembly standards address this issue poorly. Assembly standard 23 removes
a base pair from the prefix and suffix respectively giving a 5'-ACT AGA-3' scar, which
replaces the DNA scar stop codon but instead inserts a problematic Arginine residue that
is highly positively charged and also not preferred by the E.coli' translational machinery.
Assembly standard 25 adds an extra restriction consensus prior to the start codon of the
upstream protein and this would upset the 5-6 base pair spacer rule that should exist between
the RBS and the protein to ensure that translation is optimal. Neither standards
address the problem with the two TAA TAA stop codons present in the biobrick sequence.
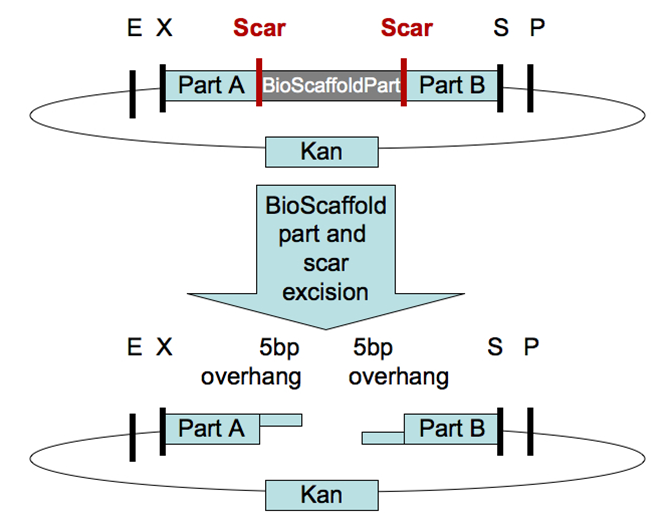
Only assembly standard 15, a concept known as Bioscaffold, tries to address this problem in some depth. The Bioscaffold is a DNA sequence that is ligated between the two protein encoding genes and contains restriction sites of atypical Type II restriction enzymes. The enzymes will cleave outside their recognition sequence hence removing the stop codons of the first protein in the fusion.
However the current scaffolds are not compatible as biobricks or not available in biobrick format and some require further manipulation with PCR primers.
 "
"