Team:Calgary/Lab
From 2009.igem.org
UNIVERSITY OF CALGARY

|
THE LAB PROJECT
The wetlab aspect of our project aims to engineer the Vibrio harveyi AI-2 signalling system in Escherichia coli. This signalling system is in the realm of Quorum Sensing, a process where microorganisms use molecules to monitor their own population density and communicate with different species. Quorum Sensing allows groups of bacteria to organize their behaviour by coordinating their gene expression. Examples include virulence induction, swarming, biofilm formation and genetic competence.
The AI-2 Signalling System
V. harveyi communicate with the quorum sensing signalling molecule AI-2. AI-2 is bound to the protein LuxP in the periplasm, and the LuxP-AI-2 complex interacts with another membrane-bound sensor histidine kinase, LuxQ. At low cell density, with low amounts of autoinducers, LuxQ sensor acts as a kinase, autophosphorylates and transfers the phosphate to the cytoplasmic protein LuxU. LuxU transfers the phosphate to the DNA binding response regulator protein LuxO. Phosphorylated LuxO with a transcription factor σ54, activates the transcription of genes encoding five regulatory small RNAs (sRNAs) termed Qrr1-5. The sRNAs bind to and destabilize the mRNA encoding the transcriptional activator, LuxR4, which is needed to activate the transcription of the luciferase operon, luxCDABE. Therefore, when there is a low cell density, the Lux R mRNA is degraded, thus the bacteria do not express bioluminescence. At higher cell density, when AI-2 concentration reaches the threshold, LuxQ sensor switches from a kinase to a phosphatase and removes the phosphate from Lux O via Lux U. Unphosphorylated LuxO cannot induce the expression of the sRNA, which in turn allows translation of LuxR mRNA, the production of Lux R and the expression of bioluminescence.
We have engineered the V. harveyi AI-2 signalling system in E. coli and coupled this with the degradation AHL and thus biofilms. Appropriate testing circuits have also been constructed in order to ensure this system is fully functional (Figure 1). 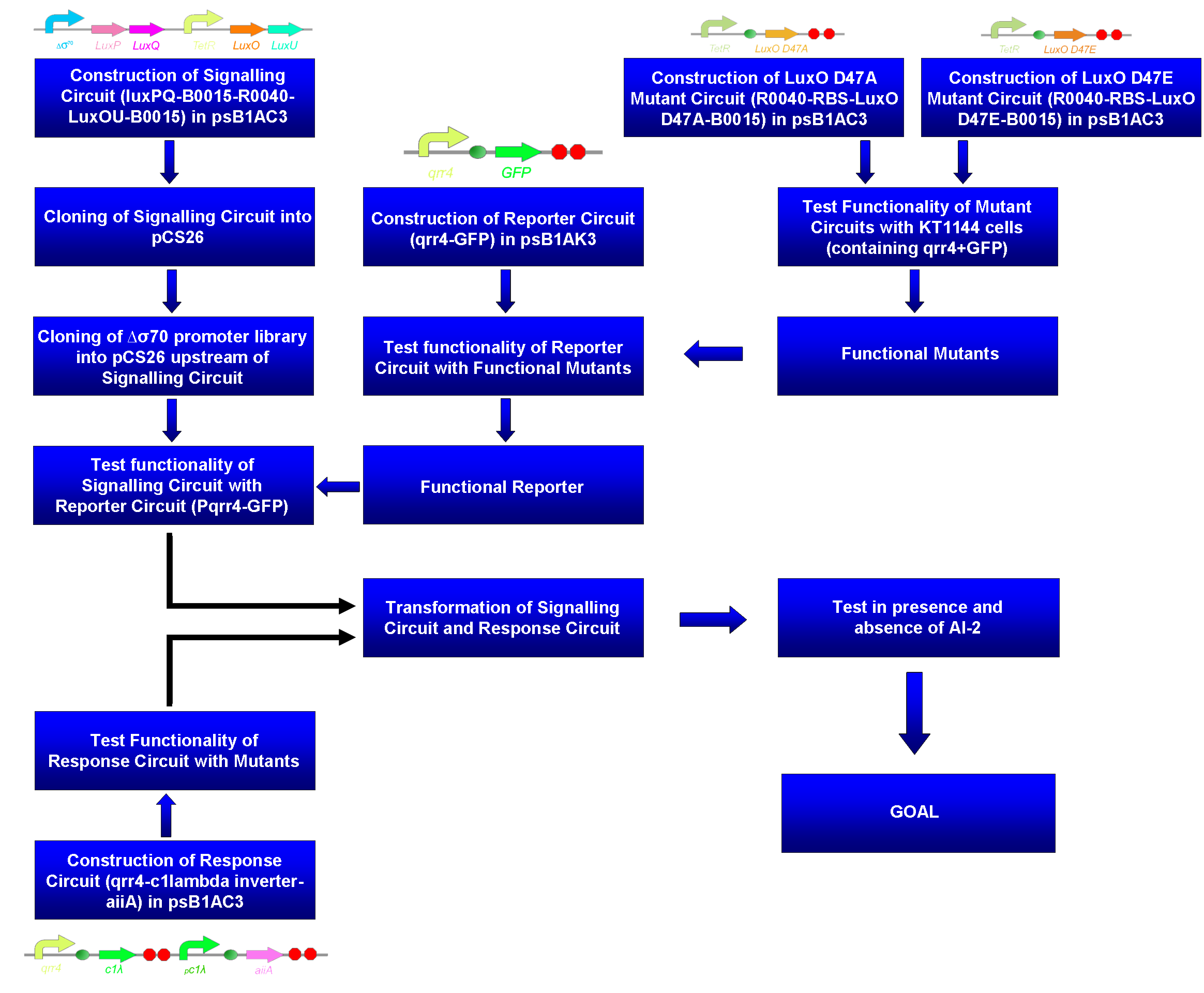
Figure 1. Flow chart of circuits required to ensure coupling of functional signalling and response circuits for AI-2 signalling system in E. coli. We have engineered the signalling circuit that encodes proteins LuxPQ and LuxOU, but in order to ensure that this circuit is functional, we require a reporter circuit. The reporter circuit itself must be functional, something that will be tested with mutant LuxO proteins that will either mimic the phosphorylated and active (LuxO D47E) or unphosphorylated and inactive (LuxO D47A) forms of LuxO. Moreover, once the signalling circuit is functional, we seek to couple this QS system with a desired response to demonstrate how AI-2 signalling can be used. This response circuit is constructed so that in the presence of AI-2, our bacteria produce aiiA, and AHL-degrading enzyme. This will target biofilm maintenance as bacteria such as Pseudomonas aeruginosa rely on this Quorum Sensing molecule for biofilm formation. This response circuit must also be tested with the LuxO proteins. This circuit can then be coupled with the signalling circuit. As a part of iGEM 2008 project, we have cloned, sequenced and standardized genes involved in the AI-2 signalling system in V. harveyi (LuxPQ, BBa_K131015; LuxOU, BBa_K131016; Pqrr4, BBa_K131017; LuxO D47E, BBa_K131022; LuxO D47A, BBa_K131023)6,7.
Part 1: Engineer functional AI-2 signalling system in E. coli
We will be engineering the complete AI-2 Signalling Circuit following Biobrick cloning methods. In order to accomplish this goal we need to:
1. Engineer variable σ70 promoter (Δσ70) 2. Clone the engineered Δσ70 promoter in front of the luxPQ construct. 3. Test robustness of the signalling system. The major obstacle of this part of the project is ensuring that we have appropriate expression levels of the LuxP and LuxQ proteins. The creation of the engineered Δσ70 promoter driving the expression of LuxPQ will provide us with adequate levels of LuxP and LuxQ for efficient AI-2 signalling. In addition, this promoter will allow us to examine the robustness of AI-2 signalling with respect to LuxP and LuxQ levels.
Part 2: Engineer a reporter circuit to test AI-2 signalling system
An appropriate reporter circuit is designed to test to test the proper functioning of the Signalling Circuit. To this end, a reporter circuit regulated by AI-2 signalling was devised. In order to engineer the Reporter Circuit we will:
1. Perform site directed mutagenesis of luxCDABE (Luciferase genes) in order to remove XbaI restriction enzyme recognition sequence. 2. Create a reporter construct comprising of luxCDABE controlled by Pqrr4 promoter.
Part 3: Engineer two testing circuits that are used to test behaviour of the reporter circuit.
Before the Reporter Circuit may be used to test the proper functioning of the Signalling Circuit, the Reporter Circuit itself must be functionally verified. We will be using mutant forms of LuxO protein in order to test the behaviour of the Reporter Circuit. The LuxOD47A mutant protein mimics the inactive form of LuxO, and the LuxOD47E mutant protein mimics the phosphorylated active form of LuxO8. In order to express mutant proteins, a constitutively-active promoter and a ribosome-binding site (available from the iGEM Registry) will be cloned in front of LuxOD47E and LuxOD47A constructs using the BioBrick cloning method. Testing will be performed in the KT1144 E. coli strain containing the cosmid-encoded qrr4-gfp fusion construct9.
Part 4: Construct an AI-2 inducible Response Circuit to be used for the wetlab application.
Once the Signalling circuit is tested, the addition of a response circuit will allow for our engineered E. coli to perform a target activity in the presence of AI-2. Different types of circuits have been considered, but this circuit will comprise of the cloned Pqrr4 promoter followed by a Registry available inverter and ending with the gene(s) of interest. The use of the Pqrr4 promoter and the inverter will allow for the induction of target activity following AI-2 exposure.
|
 "
"












