Team:KULeuven/Modelling/Vanillin Production
From 2009.igem.org
Contents |
Vanillin Production
overview
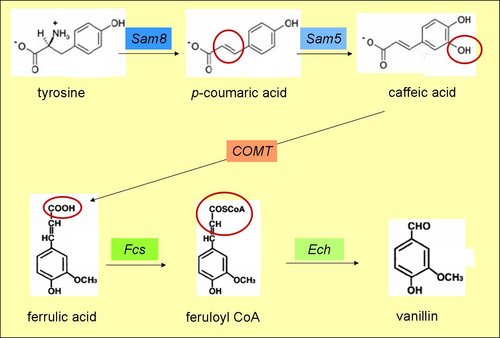
The vanillin synthesis consists of a five step process starting from tyrosine, which is used quite regulary in the biosynthesis of vanillin in E. Coli.
The behaviour of this subsystem should be a production unit of vanillin which can be regulated by the instantaneous amount of RIBOKEY present in the cell.
E. coli controls its own tyrosine production which is an non essential amino acid. However,should this be insufficient, we can always add extra tyrosine. The production process will be tested in two steps. First, from tyrosine to ferulic acid; then from ferrulic acid to vanillin.
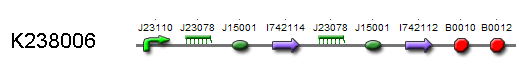
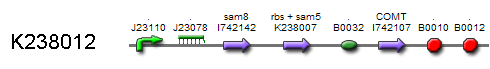
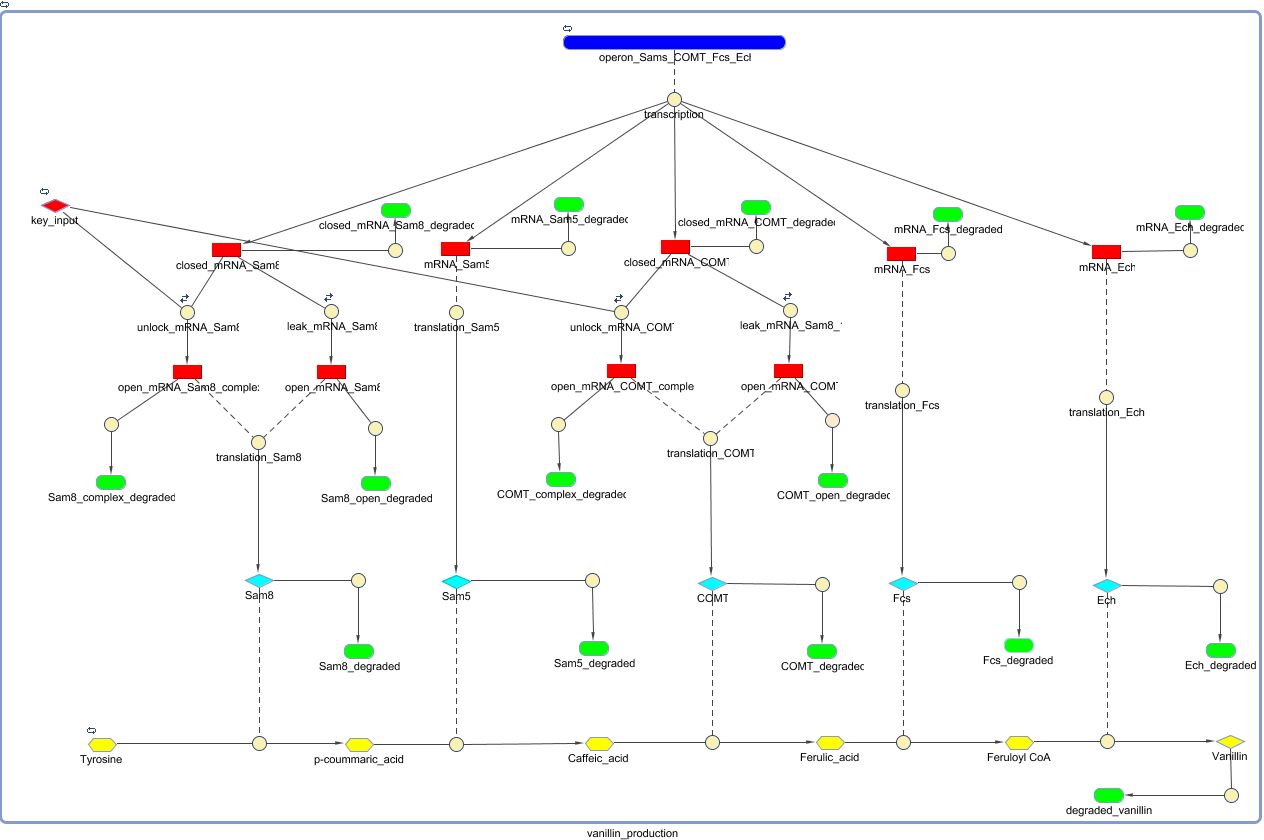
By locking both the transcription of the Sam 8 and COMT enzyme we prevent vanillin synthesis without the presence of RIBOKEY. Because we want the production of vanillin proportional to the amount of input key, it is essential that the regulated enzymes that catalyse the production of (intracellular) vanillin degrade as fast as possible. As proteins are normally very stable in the intracellular environment the SAM8 and COMT where labelled with a LVA-tag, which insures relatively fast half times. Due to the non zero half life time of the regulating enzymes, the production depends on the integrated amount of RIBOKEY over several half lives.
Biological model
Sequences used to test the vanillin production, the pathway from ferulic acid to vanillin in E. Coli is better investigated than the first. [7]
Mathematical model
| Name | Value | Comments | Reference |
|---|---|---|---|
| Degradation Rates | |||
| dmRNA | 2.3105E-3 s-1 | [1] | |
| dSam8 | 2.8881E-4 s-1 | Fast degradation through to LVA tag | [2] |
| dSam5 | 1.9254E-5 s-1 | [3] | |
| dCOMT | 2.8881E-4 s-1 | Fast degradation through to LVA tag | [2] |
| dFcs | 1.9254E-5 s-1 | [3] | |
| dEch | 1.9254E-5 s-1 | [3] | |
| dVanillin | 8.6643E-6 s-1 | Equivalent degradation dilution rate of vanillin | [4] |
| Transcription Rates | |||
| ktranscription | 0.00848 s-1 | estimate | [6] |
| ktranslation | 0.167 s-1 | estimate | [6] |
| Key Lock Parameters | |||
| kunlock | 0.00237 s-1 | Rate of unlocking the RIBOLOCK through key | [5] |
| klock | 0.00416 s-1 | Rate of locking of unlocked RIBOLOCK-Key complex. | [5] |
| kopen | 7.5 s-1 | Rate of unlocking RIBOLOCK when no key is present (LEAK). | [5] |
| kclose | 500 s-1 | Rate of locking of leaked RIBOLOCK. | [5] |
| Sam8 catalysis | |||
| kcat | 0.015 s-1 | [8] | |
| Km | 15.5 microMolarity | [8] | |
| Sam5 catalysis | |||
| kcat | 4.2385 s-1 | [9] | |
| Km | 432.0 microMolarity | [9] | |
| COMT catalysis | |||
| kcat | 0.03319 s-1 | [10] | |
| Km | 43.0 microMolarity | [10] | |
| Fcs catalysis | |||
| kcat | 0.074068 s-1 | [11] | |
| Km | 43.0 microMolarity | [11] | |
| Ech catalysis | |||
| kcat | 0.9578 s-1 | [12] | |
| Km | 2.4 microMolarity | [12] | |
| Tyrosine parameters | |||
| ntyrosine | 300000 s-1 | Because tyrosine is an amino acid we consider its level regulated by the cell | [13] |
Simulation
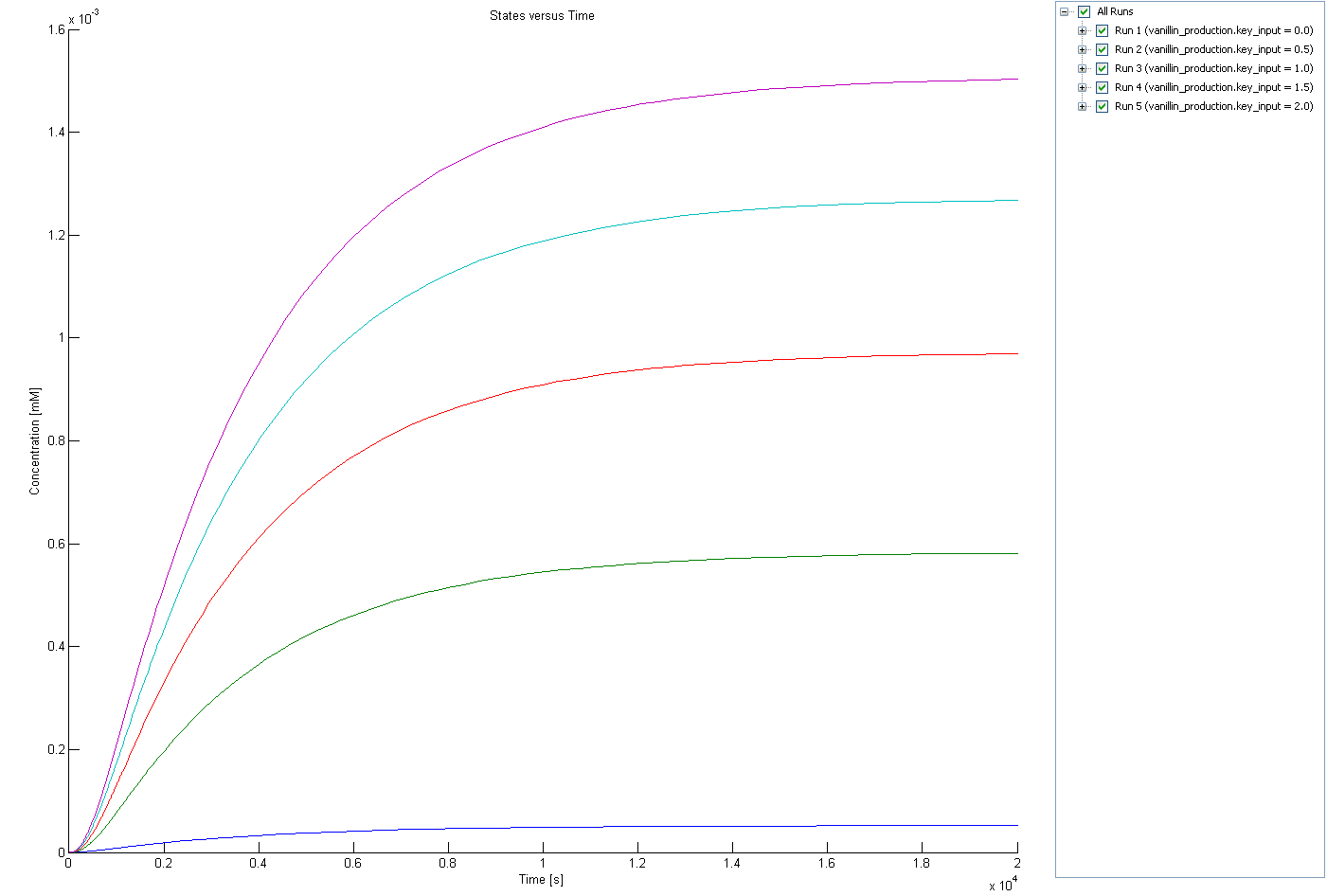
Because COMT and SAM8 are regulated by the amount of mRNA key its worthy to investigate the change of COMT and SAM8 production in function of the input amount of mRNA key. Because Sam8 and COMT are located behind the same lock they are both translated at the same rate, therefore only the concentration of COMT is shown, the translation rate of both RNA strands is considered equal.
The time scale on which COMT and Sam8 reaches equilibrium conditions is about 2 magnitudes faster than the time scale on which vanillin reaches equilibrium. Therefore only the steady state levels of COMT and Sam8 are relevant for further investigation regulatory role of both enzymes in the production of vanillin.
Because we expect only to encounter small amount of free RIBOKEY (because of the fast decay times of untranslated RNA), the steady state levels of COMT and Sam8 are relatively linearly controlled by the RIBOKEY.
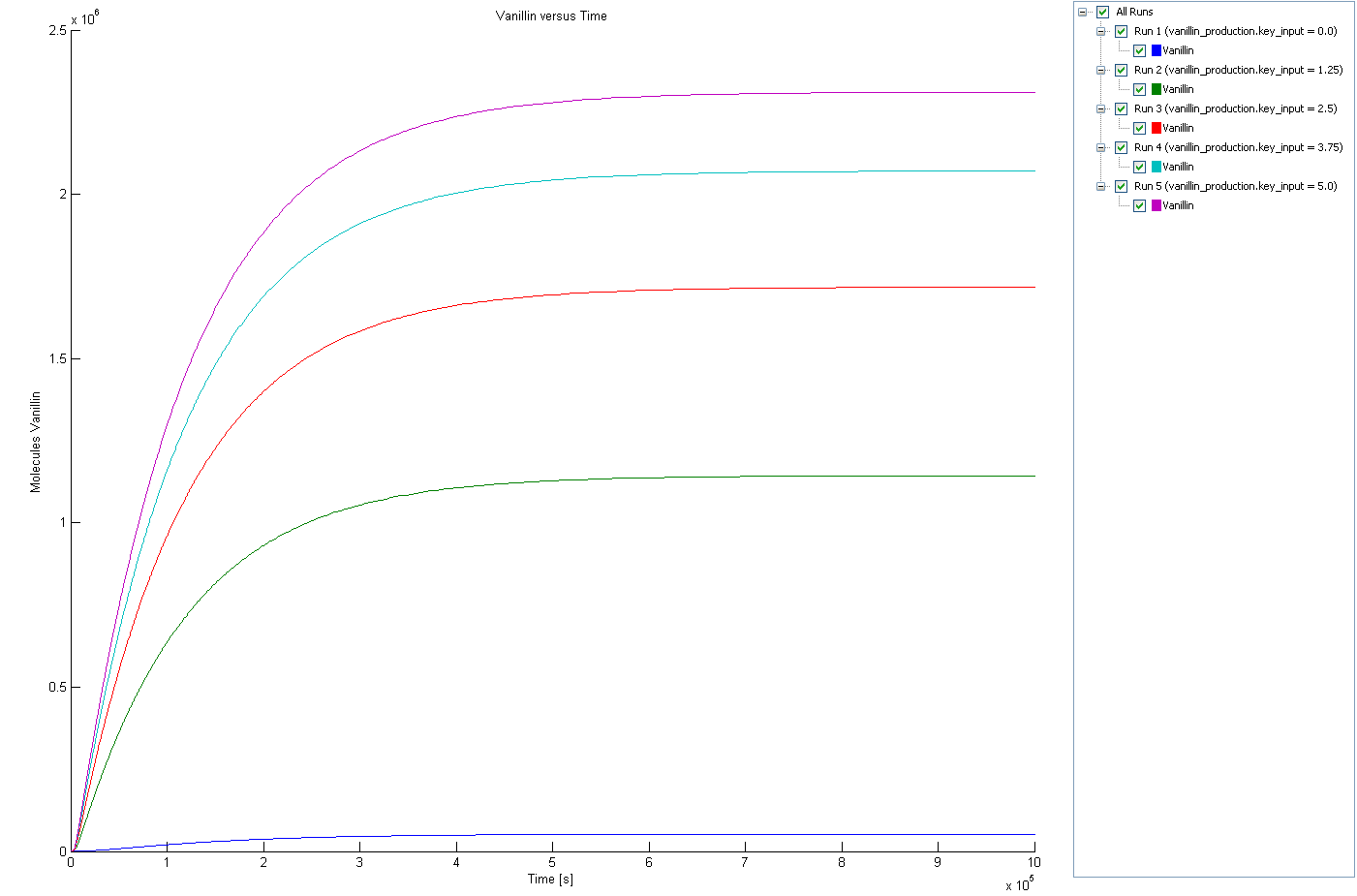
.Due to the complicated non linear (Michaelis-Menten kinetics) pathway from tyrosine to vanillin, the steady state level of vanillin well not be linearly depend on the concentration of RIBOKEY, also it takes a relatively large amount of time (hours) to reach steady state conditions.
The following figure shows that the steady state level of vanillin concentration is (non linearly) regulated by the amount of mRNA input key. Thought not linearly, the most important fact remains that the concentration of vanillin is efficiently regulated by the amount of RIBOKEY, and the leak of the RIBOLOCK is sufficiently low to obtain relatively low levels of vanillin.
References
[1] J.A. Bernstein et al., “Global analysis of mRNA decay and abundance in Escherichia coli at single-gene resolution using two-color fluorescent DNA microarrays,” Proceedings of the National Academy of Sciences of the United States of America, vol. 99, Jul. 2002, pp. 9697–9702
[2] J.B. Andersen et al., “New Unstable Variants of Green Fluorescent Protein for Studies of Transient Gene Expression in Bacteria,” Applied and Environmental Microbiology, vol. 64, Jun. 1998, pp. 2240–2246
[3] K. Nath et al., “Protein degradation in Escherichia Coli,” The Journal of Biological Chemistry, vol. 246, Nov. 1971, pp. 6956-6967
[4] https://2009.igem.org/Team:KULeuven/Modeling/Vanillin_Receptor
[5] https://2008.igem.org/Team:KULeuven/Model/Filter
[6] S.L. Gotta et al., “rRNA Transcription Rate in Escherichia Coli,” Journal of Bacteriology, vol. 173, Oct. 1991, pp. 6647-6649
[7] E.G. LEE et al., “Directing Vanillin Production From Ferulic Acid by Increased Acetyl-CoA Consumption in Recombinant Escherichia coli,” Biotechnology and Bioengineering, Jul. 2008
[8] http://jb.asm.org/cgi/content/abstract/188/7/2666
[9] B. Hamberger et al., “The 4-coumarate:CoA ligase gene family in Arabidopsis thaliana comprises one rare, sinapate activating and three commonly occurring isoenzymes,” Max Planck Institute for Plant Breeding Research, Dec. 2003
[10] C. Zubieta et al., “Structural Basis for the Modulation of Lignin Monomer Methylation by Caffeic Acid/5-Hydroxyferulic Acid 3/5-O Methyltransferase,” American Society of Plant Biologists, Jun. 2002
[11] J. Overhage et al., “Biochemical and genetic analyses of ferulic acid catabolism in Pseudomoas sp. Strain HR199,” Applied and Environmental Microbiology, Aug. 1999
[12] A. Mitra et al., “4-Hydroxycinnamoyl-CoA Hydratase/lyase (HCHL) An Enzyme of Phenylpropanoid Chain Cleavage from Pseudomonas,” Department of Genetics and Microbiology, Apr. 2002
[13] http://gchelpdesk.ualberta.ca/CCDB/cgi-bin/STAT_NEW.cgi
 "
"