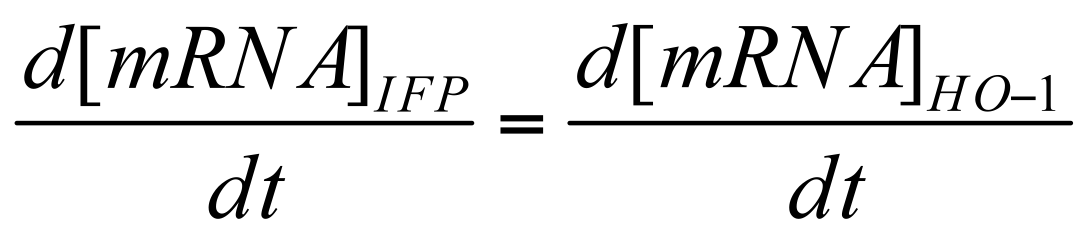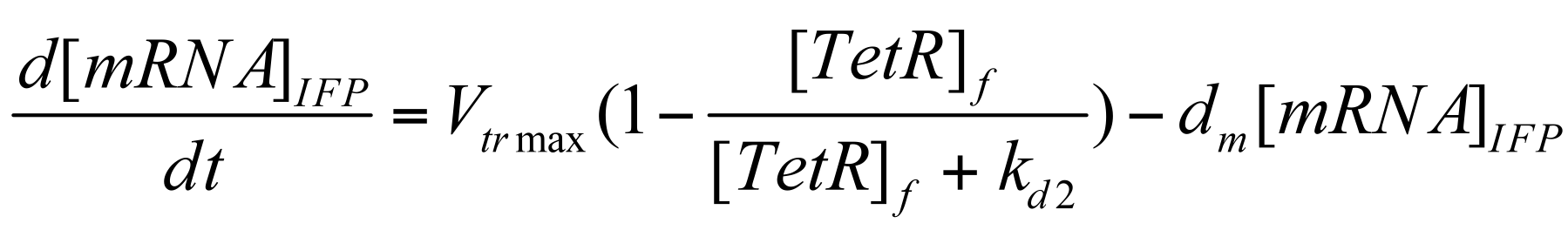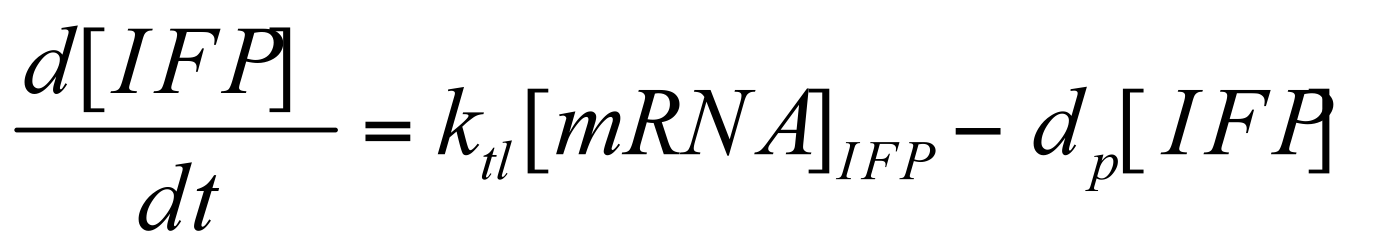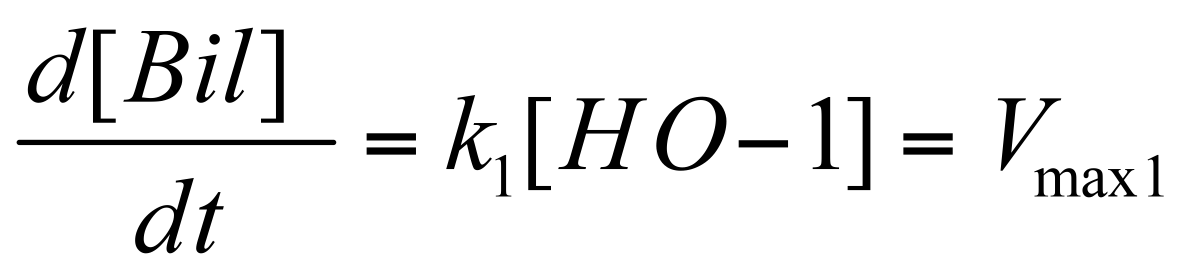Team:NTU-Singapore/Project/Prototype/Image
From 2009.igem.org
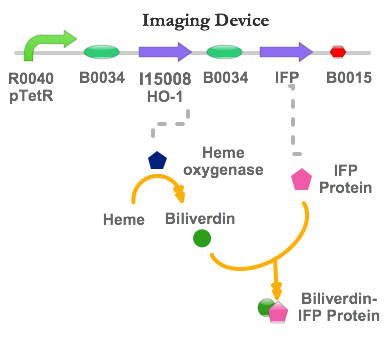
Imaging Device
The Imaging device is also one of our featured parts.
The device consists of mainly pTet regulated expression of IFP & HO-1, which in turn generate a unique fluorescence signal in the infrared spectrum!
We are extremely pleased to report that we have successfully converted this novel reporter protein into a standard Biobrick! Let us now see how exactly the device works.
Abstract
As part of the imaging capability we want for our system, we have eschewed traditional reporter proteins like GFP or YFP and have identified a novel reporter protein, infrared fluorescent protein (IFP). A recent work by Xiaokun et al. (Tsien Lab, UCSD) has shown successful mammalian expression of infrared fluorescent protein engineered from a bacterial phytochrome. Xiaokun et al. have also expressed the infrared fluorescent protein in E. coli strain TOP10.
IFP is preferred for our system over other fluorescent proteins such as GFP or YFP because it has unique excitation and emission maxima, which fit so well with the wavelength range of typical in vivo optical imaging in deep tissues of animals. At wavelength range between 650 and 900 nm, the absorbance by haemoglobin, water and lipids, as well as light-scattering are minimal.
Therefore, IFP is particularly suitable for our ideal system as we desire to locate the atherosclerotic plaque site non-invasively.
Background
Our pLaqUe Out! system must be capable of highlighting the atherosclerotic plaque site upon detection by the preceding sensing mechanism of nitric oxide sensing promoter (pNO). The first thing that came to our mind was the variety of known fluorescent proteins (FPs) such as GFP, RFP, YFP, CFP. However, since our ultimate environment for our pLaqUe Out! system is in vivo, we envisioned an ideal system that can express a novel reporter protein with properties suitable for in vivo optical imaging. IFP was chosen as a potential candidate.
The gene encoding IFP used in our expression system is essentially the gene for IFP1.4. Xiaokun et al. obtained IFP1.4 by saturation, random mutagenesis, and DNA shuffling of a gene encoding IFP1.0; and it was shown to be about four times brighter than IFP1.0.
Overall, IFP has an excitation and emission maxima of 684 nm and 708 nm, respectively. The excitation and emission maxima for other fluorescent proteins have not exceeded 598 and 655 nm, respectively.
Mechanism
Infrared fluorescent protein is a monomeric protein engineered from a bacterial phytochrome of the species Deinococcus radiodurans.
This bacteriophytochrome incorporates biliverdin IXα (BV) as the chromophore. Biliverdin IXα is in fact a natural product of heme catabolism by the enzyme heme oxygenase-1 (HO-1). BV is essential because in a mouse experiment conducted by Xiaokun et al., BV has been shown to increase the infrared fluorescence to a maximal fivefold 1 hour after BV injection in the mouse with exogenously added BV.
In a phone call made to Dr. Xiaokun Shu, we have also been told that BV helps to stabilize the infrared fluorescent protein. Hence, it is essential that we incorporate HO-1 into our system to generate biliverdin IXα.
Construct Design
As you recall, our ideal system is designed to be in a T-helper cell chassis. However, for infrared fluorescent protein production purposes, we have used E. coli strain K12 as our model system. In order to realize the production system, we have incorporated two parts into the device: the genes encoding infrared fluorescent protein (IFP) and heme oxygenase-1 (HO-1).
The Imaging Device is designed to function only in low [NO] conditions (i.e. in atherosclerotic plaque sites):
After TetR repression stops at plaque site with low [NO], both HO-1 & IFP are produced. The HO-1 catalyzes the conversion of haem to biliverdin, which then binds to IFP to allow generation of infrared signal. Once infrared signalling has begun, we can begin imaging for the plaque site using conventional imaging modality, and establish the location of the plaque site.
Of course, at this stage we also foresee numerous other medical/non-medical uses for the IFP protein!
Characteristic Equations
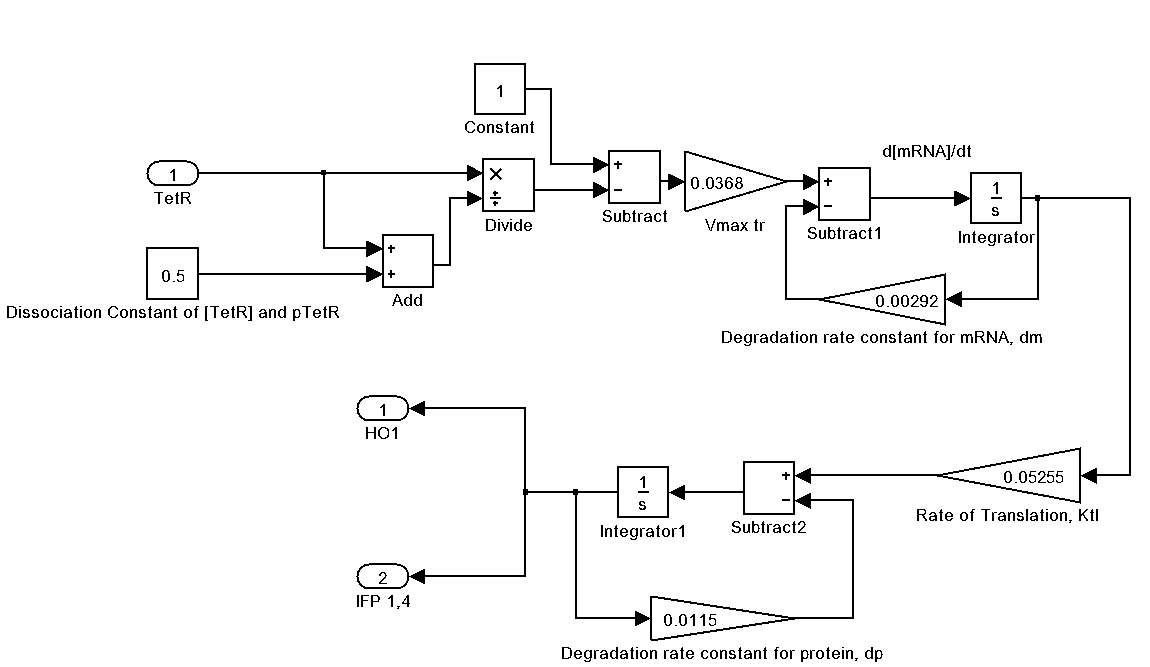
Transcription of IFP / HO-1
First, we can define the stoichiometric ratio of IFP to HO-1 transcribed.
Next, we establish the deterministic repression-regulated transcription rate of IFP & HO-1.
Where Vmax is max transcription rate & Dm is the degradation rate of mRNA.
Again, bracketed term generates the fraction of pTet not bound with TetR i.e. actively promoting pTet sites.
Translation of IFP / HO-1
Deterministic first order ODE is used again to represent the translation of IFP / HO-1 protein. Same eqn with appropriate term and constants changed.
Where Ktl is the translation rate of mRNA & Dp is the degradation rate of IFP / H0-1 protein.
Infrared signal production
Before the infrared signal can be produced, HO-1 must first convert haem to biliverdin.
And then finally we can determine an equation for rate of infrared signal, (assuming that intensity of infrared signal is directly proportional to the amount of bound-IFP-biliverdin in the system).
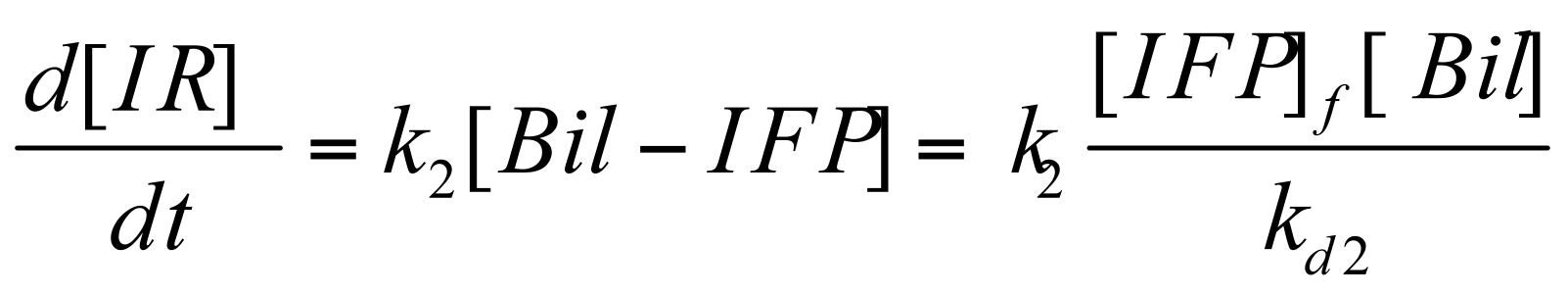 based on the ratio of bound / activated to total [IFP] available:
based on the ratio of bound / activated to total [IFP] available: 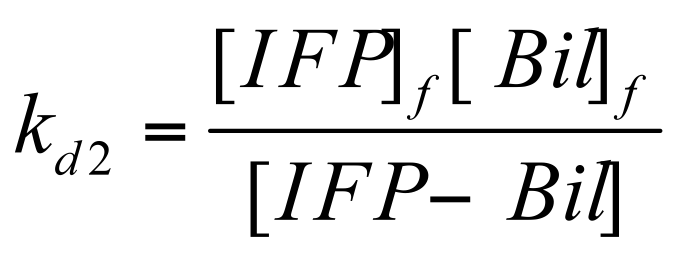
Modelling & Simulation
We make the following assumptions:
- Biological systems of transcription and translation are assumed to be linear and time-invariant.
- Concentration of the haem is considered to be non-limiting.
- There is no time lag between expression & activity.
- Constant Degradation rates for mRNA as well as protein.
- Intensity of infrared signal is directly proportional to [IFP-BV] complex.
The following is our Imaging device represented as a Simulink system. Please click on it for a larger view.
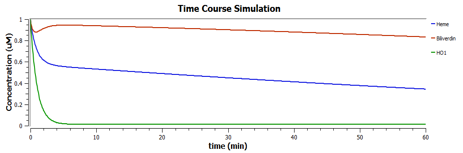
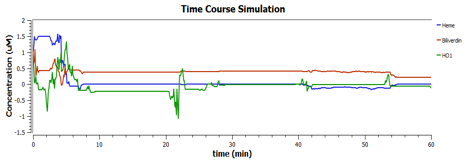
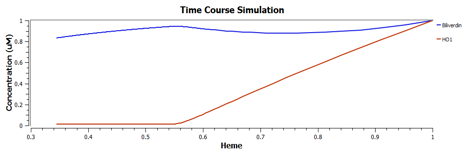
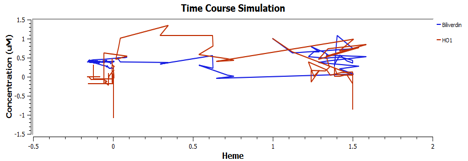
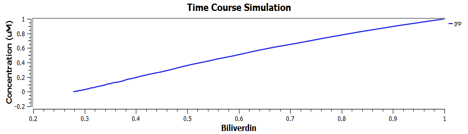
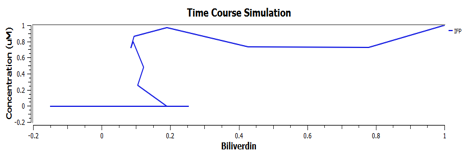
Using TinkerCell, a systems biology software, we can do deterministic (left) & stochastic (right) modelling side by side. Click on the images for a larger view.
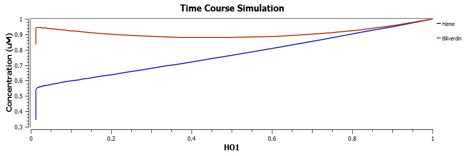
The following are the graphs of HO-1 behaviours in the system.
We find that
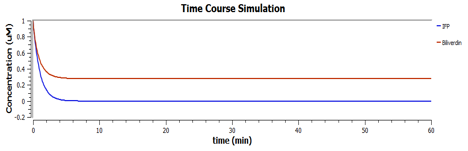
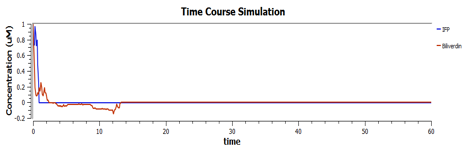
These are the behaviours of IFP in the system modelling.
Device Construction
Researchers at Tsien’s Lab graciously provided us the gene sequence of IFP1.4 optimized for E. coli, and we had the gene encoding IFP1.4 synthesized by GeneArt in accordance with the standard stated by the Registry of Standard Biological Parts.
Part: BBa_I15008 from the Registry of Standard Biological Parts contains the HO-1 gene sequence of cyanobacteria, Synechocystis, and therefore is extracted directly for our needs.
In order to achieve the final construct as described in the device construction, the following Biobricks have been successfully constructed:
- IFP
- RBS-HO1
- IFP-T
- RBS-IFP
- RBS-HO1-T
- RBS-IFP-T
- RBS-HO1-RBS-IFP-T
- pTet-RBS-HO1-T
- J23119-RBS-HO1-T
- J23119-RBS-IFP-T
- J23119-RBS-IFP-T-J23119-RBS-HO1-T
- J23119-R-IFP-T in KAN vector
Our parts listed above have been verified through the correct DNA fragments sizes by running both PCR and double digestion products on agarose gel electrophoresis. positive!
IFP Verification
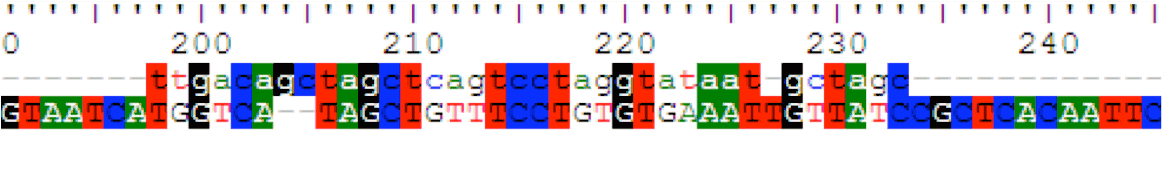
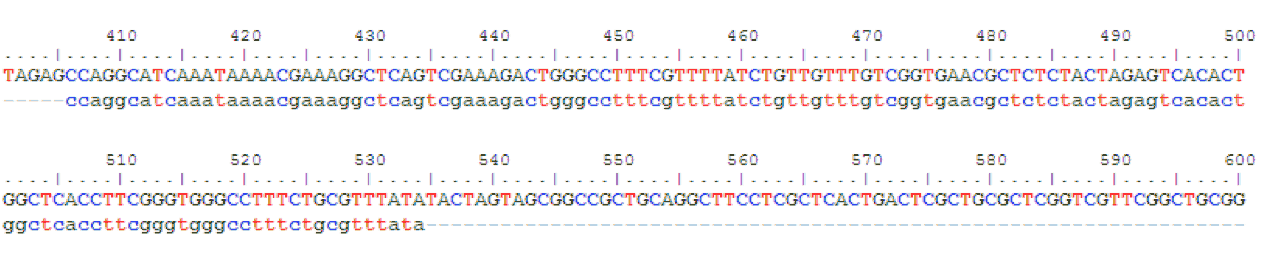
We had sent the part J23119-RBS-IFP-T that we have constructed in the final construct for verification. It came back indicating 100% match! This means that our entire construct has perfect identity to that of reported sequence, and sure enough, it WORKS!
The summary of the sequencing results is as follows:
J23119 Sequencing
The first 10 base pairs of the promoter cannot be verified due to sequencing limitation.
RBS 5 (Part: BBa_B0034)
100% verified!
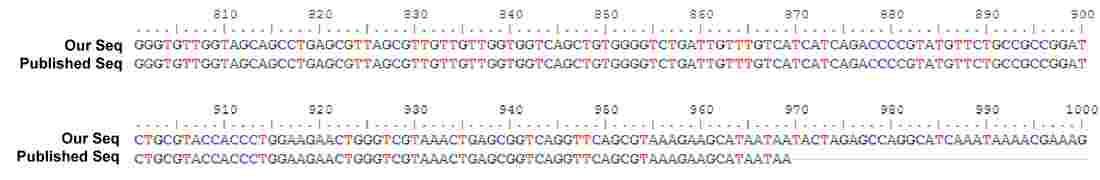
Sequence alignment of our IFP gene (top) with literature sequence (bottom).
Terminator (Part: BBa_B0015)
100% sequence identity.
Device Verification
We performed verification experiments in the laboratory to determine the successful production of infrared fluorescent protein (IFP) and/or heme oxygenase-1 (HO-1), and subsequently the functionality of our imaging device. A test construct (J23119-RBS-IFP-T-J23119-RBS-HO1-T) was constructed for this purpose.
The first approach is by running agarose gel electrophoresis of the double digested plasmids and PCR products to verify the correct DNA fragment size as well as the presence of HO-1 and IFP genes in the test construct.
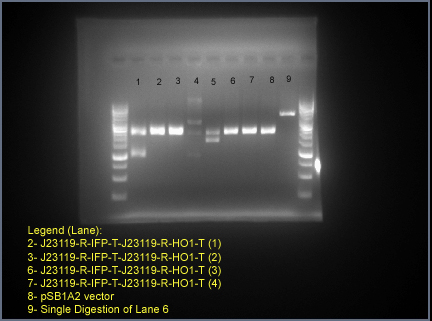
Gel electrophoresis to verify our final construct: J23119-R-IFP-T-J23119-R-HO1-T.
Lane 2, 3, 6 and 7 correspond to four different colonies picked from the ligation of J23119-R-HO1-T (insert) into J23119-R-IFP-T (vector). Single band was observed as the size of the vector and the complete construct (J23119-R-IFP-T-J23119-R-HO1-T) are similar (approximately 2400 bp). Lane 8 is the size of pSB1A2 vector in which the final construct resides. Lane 9 corresponds to the single digestion of Lane 6 to show that the lane 2, 3, 6, and 7 are not the band corresponding to single digest. The DNA gel ladder used was Fermentas GeneRuler 100bp plus.
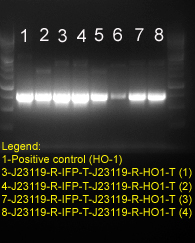
Gel electrophoresis to verify HO1 gene in the final construct.
Lane 1 is the HO-1 positive control. Lane 3, 4, 7 and 8 correspond to four different colonies picked from the ligation of J23119-R-HO1-T (insert) into J23119-R-IFP-T (vector). The result confirmed the presence of HO-1 gene in our construct: J23119-R-IFP-T-J23119-R-HO1-T.
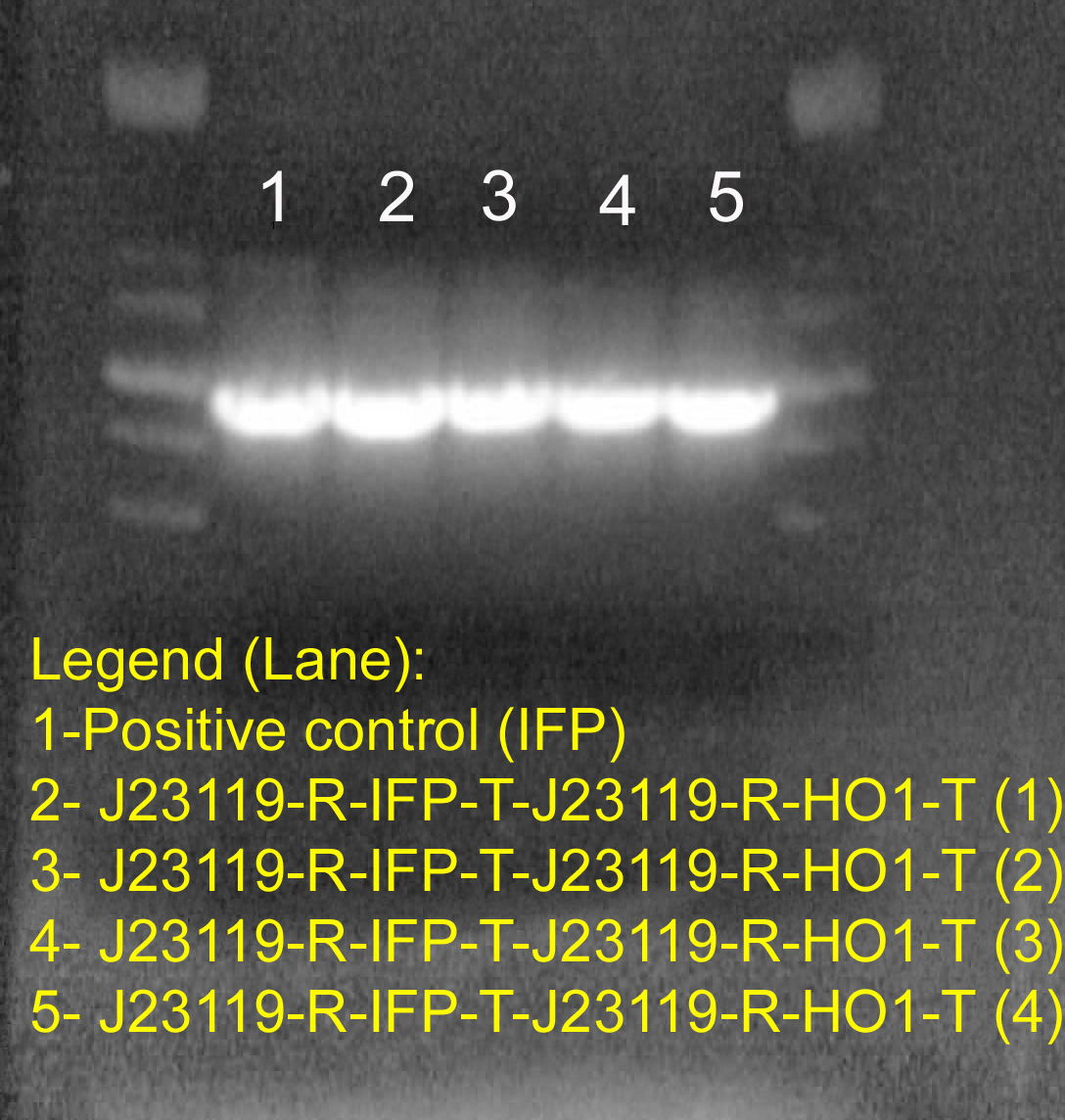
Gel electrophoresis to verify IFP gene in the final construct.
Lane 1 is the HO-1 positive control. Lane 1 is the IFP positive control. Lane 3,4,7 and 8 correspond to four different colonies picked from the ligation of J23119-R-HO1-T (insert) into J23119-R-IFP-T (vector). The result confirmed the presence of IFP gene in our construct: J23119-R-IFP-T-J23119-R-HO1-T.
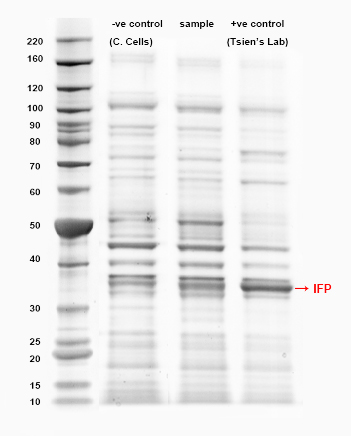
SDS-PAGE to determine the presence of IFP protein
The production of IFP in our imaging device was confirmed by SDS-PAGE on a NuPAGE® Novex 4-12% Bis-Tris gel. Protein ladder used is BenchMark™ Protein Ladder (Invitrogen Cat. No. 10747-012).
SDS-PAGE to test for the presence of IFP protein.
The first lane is the negative control, followed by our construct: J23119-R-IFP-T-J23119-R-HO1-T and the third lane is the positive control, using the sample from Tsien’s Lab. The band corresponding to the size of IFP is pointed as in the figure (for positive control). The result suggests that IFP is indeed present in our sample. The vector for the positive control is pBAD which is an inducible over-expression vector. Hence this may explain the difference in the intensities of both bands corresponding to our construct and the positive control.
To further verify that IFP is produced, Optical Imaging Experiments with the appropriate optical instruments for the detection of the infrared fluorescence emission of our constructs are also conducted repeatedly. The excitation and emission maxima of IFP are 684 and 708 nm, respectively; thus, our experimental setup must be equipped with the appropriate optical components that are of this wavelength range. Optical Imaging Experiments are extensively discussed in the following section. Our positive control for all the detection of infrared fluorescence emission is cell culture containing plasmids which ware generously provided by Tsien’s Lab.
Optical Experiment Setup
As most of commercial optical experiment setups are not equipped with excitation and emission filters of wavelengths that are in our desired range, we resorted to a customized optical experimental setup. With this in mind, we had collaborated with Prof. Lee Kijoon on the fluorescence imaging.
Ocean Optics USB-4000 Portable Spectrometer
A 660 nm Melles Griot 56ICS254/HS plus 56IMA022 laser is used as the light source. The 660 nm laser source fits almost perfectly with the ideal excitation maximum at 684 nm.
We shine the 660 nm laser onto our four-hour re-inoculated cell culture. The Ocean Optics USB-4000 Portable Spectrometer detects any signal emitted by the cell culture with an optical fiber connected to the spectrometer. The optical fiber is pointed and directed towards the cell culture for signal receiving.
In order to block the excitation light from entering the spectrometer, we included Semrock FF665-Di01-25-D dichroic mirror placed at 45-degree angle of incidence. The emission spectrum for our construct is generated with Ocean Optics SpectraSuite software.
Ocean Optics USB-4000 Portable Spectrometer
Experimental setup for infrared emission spectrum measurement.
In our attempts to optimize the experimental setup, we conducted repeated experiments with the positive control (sample of Tsien’s Lab) to generate the maximum signal intensity.
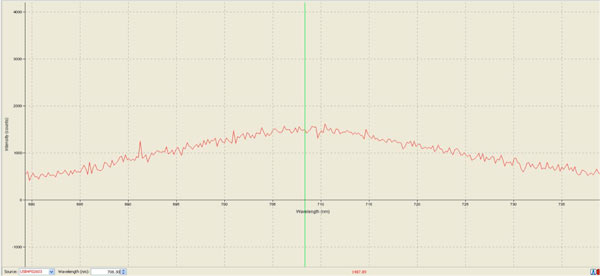
Our optical experimental setup proved to be working well when we managed to optimize our readings to a maximum peak of intensity (counts) of 1500 for a 16-hour cell culture incubated at 37°C (fluoresence emission spectrum attached as below):
Fluorescence emission spectrum of 16-hour Tsien Lab cell culture
Note excitation near the peak excitation wavelength at 660 nm. This fluorescence emission spectrum was obtained with electric dark correction.
This strongly suggests that this optical experiment setup with the Ocean Optics USB-4000 Portable Spectrometer is capable of detecting signals from our cells containing the infrared signal reporter.
The successfully constructed device: J23119-RBS-IFP-T-J23119-RBS-HO1-T (J23119:B0034:K256008:B0015:J23119:B0034:I15008:B0015) was tested for infrared signal activity with this similar setup. For simplicity, this device will be called 119-IFP-119-HO1 in subsequent text.
2mL of the overnight culture of 119-IFP-119-HO1 was pipetted into the fluorimetric four-clear-side cuvette. The cuvette was placed in the cuvette holder for detection of infrared signal. A positive control which was the sample from Tsien’s Lab and a negative control which was competent cells (E. coli strain K12) were included. However no fluorescence emission signal was generated from 119-IFP-119-HO1.
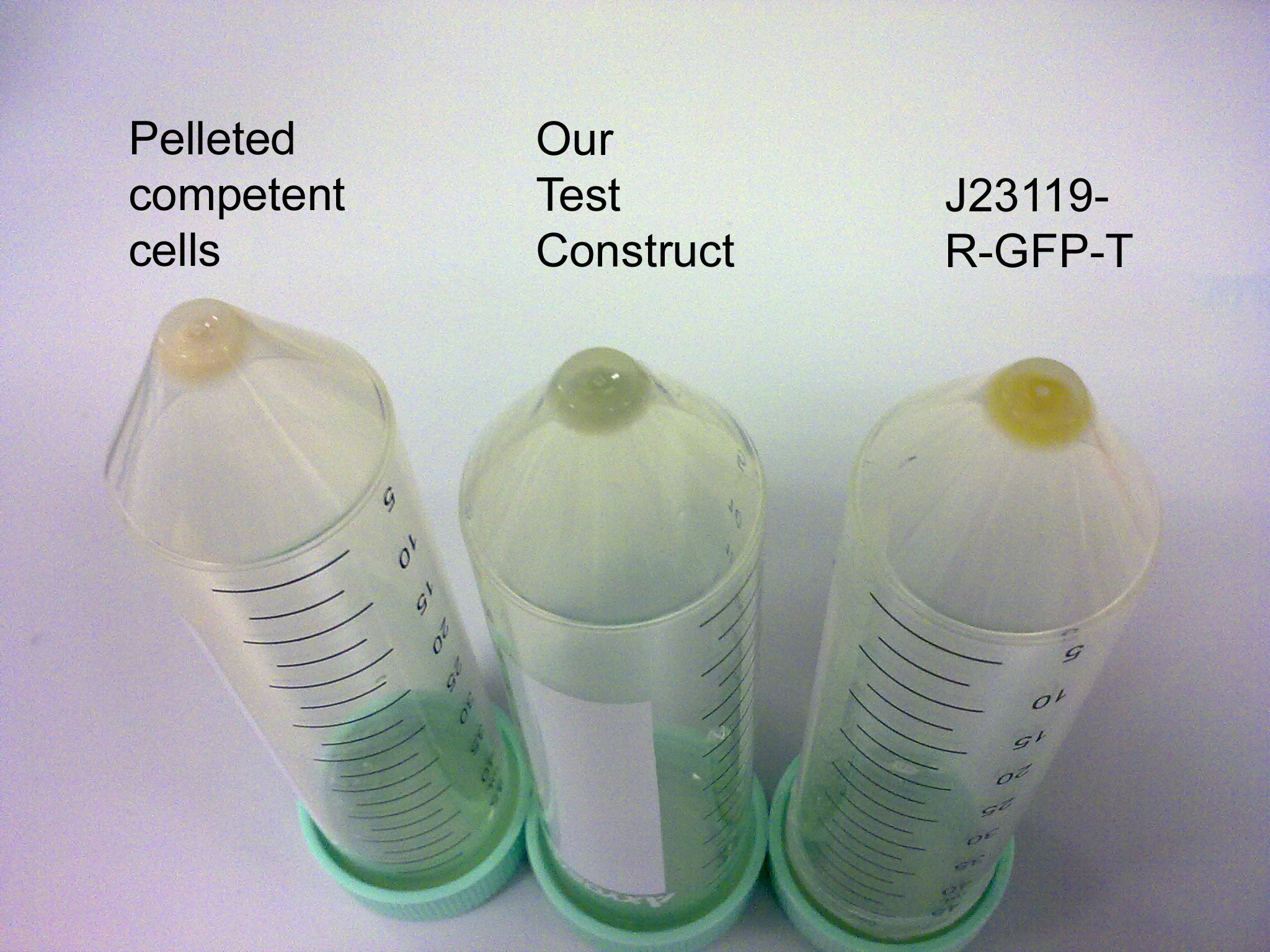
Possibilities are the concentration of the cells might be too low to generate the fluorescence emission peak. The same overnight culture of 20mL of 119-IFP-119-HO1 was centrifuged at 4000rpm for 10 minutes and the pelleted cells were resuspended in 2mL of LB for a 10x concentrated volume of cells. Anticipating a greenish pelleted cells for this device due to the heme-oxygenase 1 that will generate the biliverdin (a green pigment), the pelleted cells of 119-IFP-119-HO1 were compared and contrast by observation with pelleted competent cells (E. coli strain K12) and pelleted GFP-containing cells. Indeed, the pelleted cells of 119-IFP-119-HO1 was greenish in colour!
Cell Pellet Comparison
119-IFP-119-HO1 was greenish in colour, different from greenish of GFP.
The resuspended 2mL cell culture was pipetted into a fluorimetric four-clear-side cuvette and placed into the cuvette holder. Detection of signal was performed with the optical fiber. Results showed that there was a considerable large peak formed at the desirable emission peak wavelength (emission maximum of 708 nm). Detailed results are provided in Parts Chracterization part.
Device Characterization
Temperature Dependence
Temperature dependence of IFP protein by fluorescence emission measurement were conducted. Cell cultures were incubated for 14 hours at 225 rpm at three different temperatures: 25°C, 30°C and 37°C. Centrifugation and resuspension in 2 mL of LB were done to obtain significant signals.
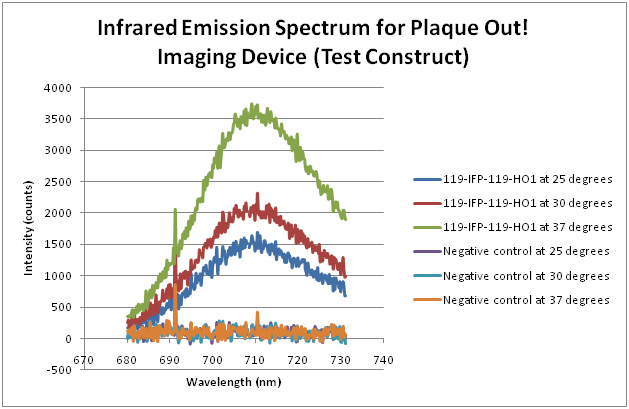
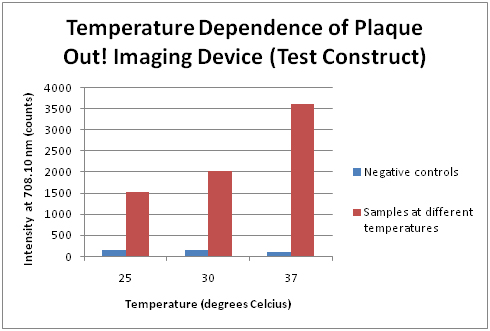
Infrared emission spectrum for Plaque! Out (Test Construct)
Our test construct was subjected to different incubation temperatures at 25, 30 and 37°C and produced infrared emission spectrum at different intensity. The incubation temperature at 37°C yields the highest spectrum as compared to those at 25°C and 30°C. Negative controls are competent cells.
Temperature Dependence of Plaque Out! Imaging Device (Test Construct)
The readings of the intensity (counts) at 708.10 nm for our test construct were plotted with the negative controls at 25, 30 and 37 degrees Celcius. This plot shows that the intensity (counts) of the infrared emission increases with incubation temperature.
Concentration Dependence
To check that our produced IFP is indeed functioning properly, we varied the concentration of the cells and measured the fluorescence intensity. Upon harvest, the cell pellets were resuspended with various amounts of buffers.
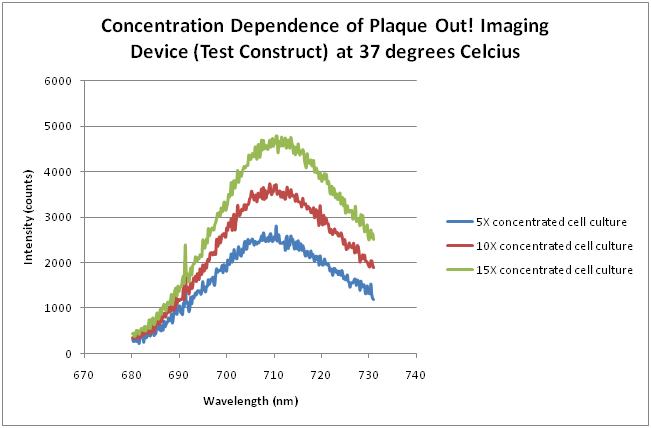
Concentration Dependence of Plaque Out! Imaging Device (Test Construct) at 37 degrees Celcius
Cell pellets from 10, 20 and 30 mL cell cultures grown at 37°C were resuspended with 2 mL of LB medium which gives 5X, 10X and 15X final concentrations, respectively. The intensity at 15X final concentration is approximately double that of the 5X final concentration.
pH Dependence
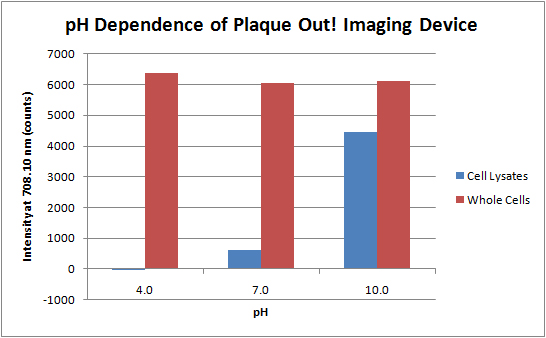
Xiaokun Shu et al. has also shown that the fluorescence of IFP is dependent upon pH. Hence, we characterized the pH dependence of IFP fluorescence. We carried out the experiment in pH 4, 7 and 10. These pH-s are of interest because the normalized fluorescence is stable over the range of pH 5 to 9. The cells were cultured at 37°C and 225 rpm for 14 hours and the cell culture was centrifuged at 4000 rpm for 10 minutes. The cell pellet was resuspended in 2 mL of Phosphate Buffered Saline adjusted to the corresponding pH. Two approaches were carried out. The first approach used whole cell while in the second approach used cell lysate to better expose the IFP to the pH condition. Subsequently, the samples were subjected to the optical experiment setup with Ocean Optics USB4000 Spectrometer as before.
pH Dependence of Plaque Out! Imaging Device
Whole cell extract does not show significant difference between the three experimental pH values. This might be due to the cell membrane which limited diffusion of the PBS to the internal cell environment. Subsequently, we sonicated the cells to obtain cell lysate. However, this procedure might have caused the biliverdin to dissociate from the IFP resulting in significantly lower intensity level.
Literature / References
Please proceed here to view our full list of references.
 "
"