Team:UAB-Barcelona/Modeling2
From 2009.igem.org
Contents |
PHOSPHATE BIOSENSOR MODEL
|
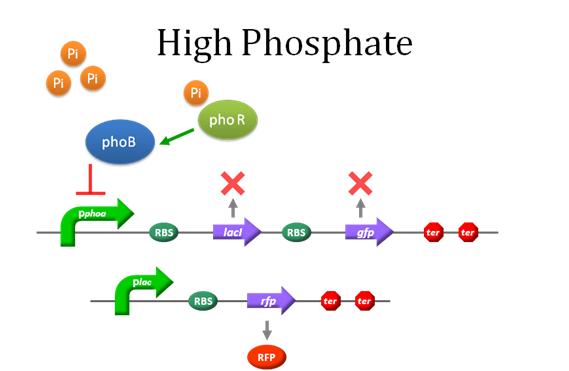
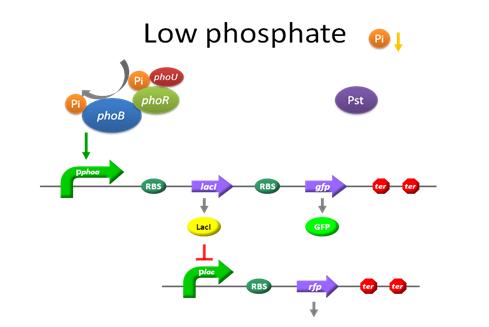
To go into this model in depth is highly recommended to read the first article listed on the model References(A Dynamic Model of the Escherichia coli Phosphate-Starvation Response). Most of the equation and parameters are taken from there. View Fig.1 of the article mentioned before to see a clear diagram of the system. |
|---|
INTRODUCTION
|
A mathematical model for the detection of phosphate by a recombinant Escherichia Coli is proposed. The model includes phosphate transport to the cell, detection of the phosphate concentration at the cell surface, and the signal transduction cascade ultimately leading to the induction of various Pho-controlled genes. Our model consist of a system of twenty nonlinear ordinary differential equations describing the temporal evolution of the key variables involved in the regulation of the pathway mentioned before.
|
|---|
PARAMETERS
VARIABLES IN THE MODEL
INITIAL STATES
EQUATIONS
REFERENCES
|
[1] A Dynamic Model of the Escherichia coli Phosphate-Starvation Response, S. J. Van Dien, J.D. Keasling, J. theor. Biol. (1998) 190, 37-49.
|
|---|
 "
"