Team:Washington/Notebook/IMAC protocol
From 2009.igem.org
BioRad Micro Column Protein Prep Protocol
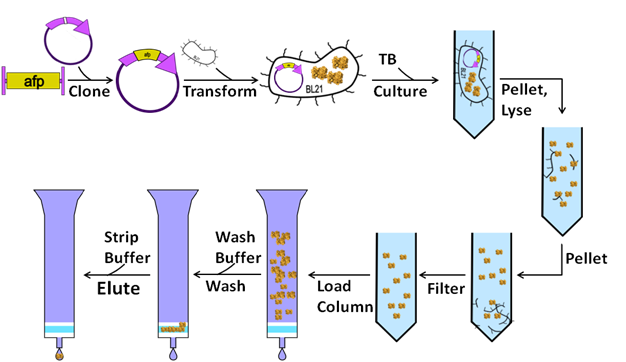
- Day 0: Clone gene of interest into vector of interest using standard cloning techniques.
- Day 1: Overnights
- Pick a single colony from plate and inoculate 1mL fresh media, with proper antibiotic, in 15mL culture tube
- Place lid on tube as not to form an air tight seal
- Shake at 37C for 16-24 hours at 200+ rpm
- Day 2: Expression
- Inoculate 50mL media with 500uL over night, in 250mL flask
- Shake 250mL flask at 37C at 200+ rpm
- Monitor OD600 of culture until OD600 ~0.4
- Use 200uL of culture and 800uL water in 1mL cuvette
- Read OD600 and multiply by 5 t get original OD600
- Should take ~2-3 hours
- INDUCE culture once culture gets to OD600 ~0.4 with IPTG so that the final concentration of IPTG is 0.5mM (ie 25uL 1M IPG for 50mL culture)
- Place in shaker 24-36hr at 18C.
- Day 3: Purify (~3.5 hours)
- Pellet Cells (0.5hr)
- Spin down cells at 4000rpm for 20 min in 50mL falcon tubes, discard supernatant
- Cells can be stored in -20C until ready for purification
- Lyse (0.5-1hr)
- Add 1mL of lysis buffer, re-suspend, transfer to a 2mL eppindorf tube
- Continue to incubate at room temperature for 20min, mixing with plate mixer
- If worried about protein stability then incubate in cold room for 1hr on rocker
- Pellet Insoluble Fraction (0.5-1hr)
- Spin the lysis for 30-60min at 15000rpm (spin longer if supernatant is not clear, but 1hr should be plenty!)
- Transfer supernatant to a fresh tube (~1800uL)
- Filter (0.1hr)
- Run supernatant through a 0.4micron filter to remove any remaning particules that would clog columns.
- Bind Protein (0.5hr)
- Add 100uL of NiNTA Agarose 50% slurry to each column (this is the Ni-beads)
- Add 600uL of diH2O to each column
- Spin 500rpm for 10s, discard flow through
- Add 500uL of wash buffer, spin, discard flow through
- Add 500uL of supernatant, spin, discard flow through
- Repeat previous step until all supernatant has passed over column (~4x)
- Wash Protein (0.5hr)
- Add 500uL of wash buffer, spin, discard flow through
- Elute Protein (0.2hr)
- CHANGE TO FRESH COLLECTION TUBE
- Add 200uL of elution buffer pipette up and down to mix the beads
- Incubate for 2min the spin at 500rpm for 1min
- THE FLOW THROUGH IS YOUR PURIFIED PROTEIN!
- Pellet Cells (0.5hr)
- Recover the beads for re-use (~45min)
- Extract beads using 200uL diH2O and transfer to 15mL falcon tube
- Store columns in dry place for later use after thoroughly washed out with water. It is fine to just run tap water over the columns after beads have been removed
- Spin down 800rpm for 5min, then decant supernatant
- Fill falcon tube with 100mM EDTA, and re-suspend beads
- Spin down 800rpm for 5min, then decant supernatant
- Fill falcon tube with diH2O, and re-suspend beads
- Spin down 800rpm for 5min, then decant supernatant
- Fill falcon tube with 100mM NiSO4, and re-suspend beads
- Spin down 800rpm for 5min, then decant supernatant
- Fill falcon tube with diH2O, and re-suspend beads
- Spin down 800rpm for 5min, then decant supernatant
- Add 20% ethanol until a 50% slurry has been reached.
- Store at 4C
- Extract beads using 200uL diH2O and transfer to 15mL falcon tube
- Buffer Examples (Alter to fit your protein's ideal buffer)
- Lysis Buffer
- 1.5x BugBuster (Solubilizes cell wall)
- 1mg/ml lysozyme (Helps break cell wall)
- 0.1mg/ml DNAseI (chews up DNA)
- 1mM PMSF (Protease Inhibitor)
- 1x PBS
- 30mM Imidizole
- Lysis Buffer
- Wash Buffer
- 1x PBS
- 30mM Imidizole
- BUGBUSTER Lysis Buffer
- 1x PBS
- 1mM PMSF
- 2x Bug Buster
- 2mg/mL lysozyme
- 0.2mg/ml DNAse
- Elution Buffer
- 1x PBS
- 250mM Imidizole
- Wash Buffer
 "
"