| April
|
| M | T | W | T | F | S | S
|
|
|
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/1_April_2009&action=edit 1]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/2_April_2009&action=edit 2]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/3_April_2009&action=edit 3]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/4_April_2009&action=edit 4]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/5_April_2009&action=edit 5]
|
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/6_April_2009&action=edit 6]
| [http://2009.igem.org/Wisconsin-Madison/7_April_2009 7]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/8_April_2009&action=edit 8]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/9_April_2009&action=edit 9]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/10_April_2009&action=edit 10]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/11_April_2009&action=edit 11]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/12_April_2009&action=edit 12]
|
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/13_April_2009&action=edit 13]
| [http://2009.igem.org/Wisconsin-Madison/14_April_2009 14]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/15_April_2009&action=edit 15]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/16_April_2009&action=edit 16]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/17_April_2009&action=edit 17]
| [http://2009.igem.org/Wisconsin-Madison/18_April_2009 18]
| [http://2009.igem.org/Wisconsin-Madison/19_April_2009 19]
|
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/20_April_2009&action=edit 20]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/21_April_2009&action=edit 21]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/22_April_2009&action=edit 22]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/23_April_2009&action=edit 23]
| [http://2009.igem.org/Wisconsin-Madison/24_April_2009 24]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/25_April_2009&action=edit 25]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/26_April_2009&action=edit 26]
|
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/27_April_2009&action=edit 27]
| [http://2009.igem.org/Wisconsin-Madison/28_April_2009 28]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/29_April_2009&action=edit 29]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/30_April_2009&action=edit 30]
|
|
| May
|
| M | T | W | T | F | S | S
|
|
|
|
|
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/1_May_2009&action=edit 1]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/2_May_2009&action=edit 2]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/3_May_2009&action=edit 3]
|
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/4_May_2009&action=edit 4]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/5_May_2009&action=edit 5]
| [http://2009.igem.org/Wisconsin-Madison/6_May_2009 6]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/7_May_2009&action=edit 7]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/8_May_2009&action=edit 8]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/9_May_2009&action=edit 9]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/10_May_2009&action=edit 10]
|
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/11_May_2009&action=edit 11]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/12_May_2009&action=edit 12]
| [http://2009.igem.org/Wisconsin-Madison/13_May_2009 13]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/14_May_2009&action=edit 14]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/15_May_2009&action=edit 15]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/16_May_2009&action=edit 16]
| [http://2009.igem.org/Wisconsin-Madison/17_May_2009 17]
|
| [http://2009.igem.org/Wisconsin-Madison/18_May_2009 18]
| [http://2009.igem.org/Wisconsin-Madison/19_May_2009 19]
| [http://2009.igem.org/Wisconsin-Madison/20_May_2009 20]
| [http://2009.igem.org/Wisconsin-Madison/21_May_2009 21]
| [http://2009.igem.org/Wisconsin-Madison/22_May_2009 22]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/23_May_2009&action=edit 23]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/24_May_2009&action=edit 24]
|
| [http://2009.igem.org/Wisconsin-Madison/25_May_2009 25]
| [http://2009.igem.org/Wisconsin-Madison/26_May_2009 26]
| [http://2009.igem.org/Wisconsin-Madison/27_May_2009 27]
| [http://2009.igem.org/Wisconsin-Madison/28_May_2009 28]
| [http://2009.igem.org/Wisconsin-Madison/29_May_2009 29]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/30_May_2009&action=edit 30]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/31_May_2009&action=edit 31]
|
|
| June
|
| M | T | W | T | F | S | S
|
| [http://2009.igem.org/Wisconsin-Madison/1_June_2009 1]
| [http://2009.igem.org/Wisconsin-Madison/2_June_2009 2]
| [http://2009.igem.org/Wisconsin-Madison/3_June_2009 3]
| [http://2009.igem.org/Wisconsin-Madison/4_June_2009 4]
| [http://2009.igem.org/Wisconsin-Madison/5_June_2009 5]
| [http://2009.igem.org/Wisconsin-Madison/6_June_2009 6]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/7_June_2009&action=edit 7]
|
| [http://2009.igem.org/Wisconsin-Madison/8_June_2009 8]
| [http://2009.igem.org/Wisconsin-Madison/9_June_2009 9]
| [http://2009.igem.org/Wisconsin-Madison/10_June_2009 10]
| [http://2009.igem.org/Wisconsin-Madison/11_June_2009 11]
| [http://2009.igem.org/Wisconsin-Madison/12_June_2009 12]
| [http://2009.igem.org/Wisconsin-Madison/13_June_2009 13]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/14_June_2009&action=edit 14]
|
| [http://2009.igem.org/Wisconsin-Madison/15_June_2009 15]
| [http://2009.igem.org/Wisconsin-Madison/16_June_2009 16]
| [http://2009.igem.org/Wisconsin-Madison/17_June_2009 17]
| [http://2009.igem.org/Wisconsin-Madison/18_June_2009 18]
| [http://2009.igem.org/Wisconsin-Madison/19_June_2009 19]
| [http://2009.igem.org/Wisconsin-Madison/20_June_2009 20]
| [http://2009.igem.org/Wisconsin-Madison/21_June_2009 21]
|
| [http://2009.igem.org/Wisconsin-Madison/22_June_2009 22]
| [http://2009.igem.org/Wisconsin-Madison/23_June_2009 23]
| [http://2009.igem.org/Wisconsin-Madison/24_June_2009 24]
| [http://2009.igem.org/Wisconsin-Madison/25_June_2009 25]
| [http://2009.igem.org/Wisconsin-Madison/26_June_2009 26]
| [http://2009.igem.org/Wisconsin-Madison/27_June_2009 27]
| [http://2009.igem.org/Wisconsin-Madison/28_June_2009 28]
|
| [http://2009.igem.org/Wisconsin-Madison/29_June_2009 29]
| [http://2009.igem.org/Wisconsin-Madison/30_June_2009 30]
|
|
| July
|
| M | T | W | T | F | S | S
|
|
|
| [http://2009.igem.org/Wisconsin-Madison/1_July_2009 1]
| [http://2009.igem.org/Wisconsin-Madison/2_July_2009 2]
| [http://2009.igem.org/Wisconsin-Madison/3_July_2009 3]
| [http://2009.igem.org/Wisconsin-Madison/4_July_2009 4]
| [http://2009.igem.org/Wisconsin-Madison/5_July_2009 5]
|
| [http://2009.igem.org/Wisconsin-Madison/6_July_2009 6]
| [http://2009.igem.org/Wisconsin-Madison/7_July_2009 7]
| [http://2009.igem.org/Wisconsin-Madison/8_July_2009 8]
| [http://2009.igem.org/Wisconsin-Madison/9_July_2009 9]
| [http://2009.igem.org/Wisconsin-Madison/10_July_2009 10]
| [http://2009.igem.org/Wisconsin-Madison/11_July_2009 11]
| [http://2009.igem.org/Wisconsin-Madison/12_July_2009 12]
|
| [http://2009.igem.org/Wisconsin-Madison/13_July_2009 13]
| [http://2009.igem.org/Wisconsin-Madison/14_July_2009 14]
| [http://2009.igem.org/Wisconsin-Madison/15_July_2009 15]
| [http://2009.igem.org/Wisconsin-Madison/16_July_2009 16]
| [http://2009.igem.org/Wisconsin-Madison/17_July_2009 17]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/18_July_2009&action=edit 18]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/19_July_2009&action=edit 19]
|
| [http://2009.igem.org/Wisconsin-Madison/20_July_2009 20]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/21_July_2009&action=edit 21]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/22_July_2009&action=edit 22]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/23_July_2009&action=edit 23]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/24_July_2009&action=edit 24]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/25_July_2009&action=edit 25]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/26_July_2009&action=edit 26]
|
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/27_July_2009&action=edit 27]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/28_July_2009&action=edit 28]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/29_July_2009&action=edit 29]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/30_July_2009&action=edit 30]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/31_July_2009&action=edit 31]
|
|
| August
|
| M | T | W | T | F | S | S
|
|
|
|
|
|
| [http://2009.igem.org/Wisconsin-Madison/1_August_2009 1]
| [http://2009.igem.org/Wisconsin-Madison/2_August_2009 2]
|
| [http://2009.igem.org/Wisconsin-Madison/3_August_2009 3]
| [http://2009.igem.org/Wisconsin-Madison/4_August_2009 4]
| [http://2009.igem.org/Wisconsin-Madison/5_August_2009 5]
| [http://2009.igem.org/Wisconsin-Madison/6_August_2009 6]
| [http://2009.igem.org/Wisconsin-Madison/7_August_2009 7]
| [http://2009.igem.org/Wisconsin-Madison/8_August_2009 8]
| [http://2009.igem.org/Wisconsin-Madison/9_August_2009 9]
|
| [http://2009.igem.org/Wisconsin-Madison/10_August_2009 10]
| [http://2009.igem.org/Wisconsin-Madison/11_August_2009 11]
| [http://2009.igem.org/Wisconsin-Madison/12_August_2009 12]
| [http://2009.igem.org/Wisconsin-Madison/13_August_2009 13]
| [http://2009.igem.org/Wisconsin-Madison/14_August_2009 14]
| [http://2009.igem.org/Wisconsin-Madison/15_August_2009 15]
| [http://2009.igem.org/Wisconsin-Madison/16_August_2009 16]
|
| [http://2009.igem.org/Wisconsin-Madison/17_August_2009 17]
| [http://2009.igem.org/Wisconsin-Madison/18_August_2009 18]
| [http://2009.igem.org/Wisconsin-Madison/19_August_2009 19]
| [http://2009.igem.org/Wisconsin-Madison/20_August_2009 20]
| [http://2009.igem.org/Wisconsin-Madison/21_August_2009 21]
| [http://2009.igem.org/Wisconsin-Madison/22_August_2009 22]
| [http://2009.igem.org/Wisconsin-Madison/23_August_2009 23]
|
| [http://2009.igem.org/Wisconsin-Madison/24_August_2009 24]
| [http://2009.igem.org/Wisconsin-Madison/25_August_2009 25]
| [http://2009.igem.org/Wisconsin-Madison/26_August_2009 26]
| [http://2009.igem.org/Wisconsin-Madison/27_August_2009 27]
| [http://2009.igem.org/Wisconsin-Madison/28_August_2009 28]
| [http://2009.igem.org/Wisconsin-Madison/29_August_2009 29]
| [http://2009.igem.org/Wisconsin-Madison/30_August_2009 30]
|
| [http://2009.igem.org/Wisconsin-Madison/31_August_2009 31]
|
|
| September
|
| M | T | W | T | F | S | S
|
|
| [http://2009.igem.org/Wisconsin-Madison/1_September_2009 1]
| [http://2009.igem.org/Wisconsin-Madison/2_September_2009 2]
| [http://2009.igem.org/Wisconsin-Madison/3_September_2009 3]
| [http://2009.igem.org/Wisconsin-Madison/4_September_2009 4]
| [http://2009.igem.org/Wisconsin-Madison/5_September_2009 5]
| [http://2009.igem.org/Wisconsin-Madison/6_September_2009 6]
|
| [http://2009.igem.org/Wisconsin-Madison/7_September_2009 7]
| [http://2009.igem.org/Wisconsin-Madison/8_September_2009 8]
| [http://2009.igem.org/Wisconsin-Madison/9_September_2009 9]
| [http://2009.igem.org/Wisconsin-Madison/10_September_2009 10]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/11_September_2009&action=edit 11]
| [http://2009.igem.org/Wisconsin-Madison/12_September_2009 12]
| [http://2009.igem.org/Wisconsin-Madison/13_September_2009 13]
|
| [http://2009.igem.org/Wisconsin-Madison/14_September_2009 14]
| [http://2009.igem.org/Wisconsin-Madison/15_September_2009 15]
| [http://2009.igem.org/Wisconsin-Madison/16_September_2009 16]
| [http://2009.igem.org/Wisconsin-Madison/17_September_2009 17]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/18_September_2009&action=edit 18]
| [http://2009.igem.org/Wisconsin-Madison/19_September_2009 19]
| [http://2009.igem.org/Wisconsin-Madison/20_September_2009 20]
|
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/21_September_2009&action=edit 21]
| [http://2009.igem.org/Wisconsin-Madison/22_September_2009 22]
| [http://2009.igem.org/Wisconsin-Madison/23_September_2009 23]
| [http://2009.igem.org/Wisconsin-Madison/24_September_2009 24]
| [http://2009.igem.org/Wisconsin-Madison/25_September_2009 25]
| [http://2009.igem.org/Wisconsin-Madison/26_September_2009 26]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/27_September_2009&action=edit 27]
|
| [http://2009.igem.org/Wisconsin-Madison/28_September_2009 28]
| [http://2009.igem.org/Wisconsin-Madison/29_September_2009 29]
| [http://2009.igem.org/Wisconsin-Madison/30_September_2009 30]
|
|
| October
|
| M | T | W | T | F | S | S
|
|
|
|
| [http://2009.igem.org/Wisconsin-Madison/1_October_2009 1]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/2_October_2009&action=edit 2]
| [http://2009.igem.org/Wisconsin-Madison/3_October_2009 3]
| [http://2009.igem.org/Wisconsin-Madison/4_October_2009 4]
|
| [http://2009.igem.org/Wisconsin-Madison/5_October_2009 5]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/6_October_2009&action=edit 6]
| [http://2009.igem.org/Wisconsin-Madison/7_October_2009 7]
| [http://2009.igem.org/Wisconsin-Madison/8_October_2009 8]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/9_October_2009&action=edit 9]
| [http://2009.igem.org/Wisconsin-Madison/10_October_2009 10]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/11_October_2009&action=edit 11]
|
| [http://2009.igem.org/Wisconsin-Madison/12_October_2009 12]
| [http://2009.igem.org/Wisconsin-Madison/13_October_2009 13]
| [http://2009.igem.org/Wisconsin-Madison/14_October_2009 14]
| [http://2009.igem.org/Wisconsin-Madison/15_October_2009 15]
| [http://2009.igem.org/Wisconsin-Madison/16_October_2009 16]
| [http://2009.igem.org/Wisconsin-Madison/17_October_2009 17]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/18_October_2009&action=edit 18]
|
| [http://2009.igem.org/Wisconsin-Madison/19_October_2009 19]
| [http://2009.igem.org/Wisconsin-Madison/20_October_2009 20]
| [http://2009.igem.org/Wisconsin-Madison/21_October_2009 21]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/22_October_2009&action=edit 22]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/23_October_2009&action=edit 23]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/24_October_2009&action=edit 24]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/25_October_2009&action=edit 25]
|
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/26_October_2009&action=edit 26]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/27_October_2009&action=edit 27]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/28_October_2009&action=edit 28]
| [http://2009.igem.org/wiki/index.php?title=Wisconsin-Madison/29_October_2009&action=edit 29]
| [http://2009.igem.org/Wisconsin-Madison/30_October_2009 30]
| [http://2009.igem.org/Wisconsin-Madison/31_October_2009 31]
|
|
June 26, 2009
Ex 17: GFP Regulation by ProU Promoter
Test 2: Results:
Salt testing of GFP, ProU had very indicative results that the promoter is working
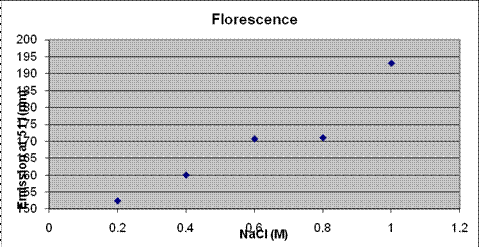

Problems: no GFP only, no ProU+GFP Control 0M, no DH10B: controls put in after test NEED BETTER Experimental Design
Experimental Design: (next test - Tomorrow)
0.0-1.0M in increments of 0.2 concentrations of (4 wells of each concentration):
1. Wild Type E.Coli
2. GFP only
3. ProU + GFP
(Plan to include the SAM synthase (METK) to boost gsSDMT function, and eventually getting rid of the GFP and evaluating solely on growth)
4. ProU + GFP + gsSDMT
5. ProU + GFP + gsSDMT + MetK
6. Salt Free LB – start (2 wells of each concentration)
7. Water (2 wells of each concentration)
1. Grow four different cultures till OD600=0.4 to 0.6
2. Induce cultures with various concentrations of salt in the plate reader
3. Let the plate reader shake and incubate overnight
4. Take OD and excitation/emission value at 501/511nm during the entire time course
Inoculate: ProU+GFP, GFP only, DH10B
Ex 16: Choline, ProU, NudF, YhfR, ProU + GFP
Choline BB Plating is Decent: 3 white colonies in the lower left-hand corner
(will redo along with other samples just to be safe since the pink quality is expressed with time)
Streak: Independent lanes of three white colonies
Redo Cloning: YhfR, NudF, ProU, Choline
Important: Take concentration before and after digestion.
A. PCR clean up: Taq PCR Product - Promega Wizard Genomic DNA Purification Kit
B. Spec 1:
NudF (560) 680 ug/mL
YhfR (560) 665 ug/mL
Choline (2k) 716 ug/mL
ProU (300) 542 ug/mL
BB (2600) 98 ug/mL
C. Digestion: DNA should be at 1ug/50uL concentration
ProU
YhfR
NudF
Choline
BB P1
BB P2
*EcoR1 Buffer and Protein
D. Purification: according to Promega Wizard Genomic DNA Purification Kit
E. Spec 2: original - speedvac
NudF (560) 2 ug/mL - 30 ug/mL
YhfR (560) 4 ug/mL - 27 ug/mL
Choline (2k) 1 ug/mL - 63 ug/mL
ProU (300) 1 ug/mL - 104 ug/mL
BB P1(2600) 4 ug/mL - 97 ug/mL
BB P2(2600) 2 ug/mL - 100 ug/mL
F. Ligation:
BB P2 + ProU
BB P1 + YhfR
BB P1 + NudF
BB P1 + Choline
BB P2
BB P1
F. Transformation: of YhfR, NudF, ProU, Choline into BL21 (DE3) (extremely high competency)
Ex 15: Inducing various Modified (Triple) E.Coli Strain Cultures 3
Transformed Confirmed Triple DNA into following strains and plated:
1. BL21 (DE3) == Lawn
2. K-12 MG1655 AaraBAD == 6 colonies
3. K-12 MG1655 (wildtype) == 4 colonies
Transformed Confirmed Double (MevT, NudF) DNA into following strains and plated (incase triple was unsuccessful):
1. BL21 (DE3) === Lawn
2. K-12 MG1655 AaraBAD == ~300 colonies
3. K-12 MG1655 (wildtype) == ~300 colonies
Important: BL21 are the most competent cells we have been able to obtain - should use for transformations in cloning instead of DH10B
Inoculate: 250ml culture – 250 uL Amp, 250 uL Tet, 183.75 uL Cm – with triple transformed plasmid
1. BL21 (DE3)
2. MG1655 Wildtype
3. MG1655 Delta Arabad
Ex 10: Made medium and grew cyanos 7002
Bubbling of C02 has substantially increased the growth rate of cyanobacteria
Ex 18:

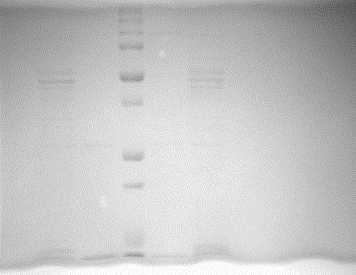
WHAT DOES THIS MEAN???
EX 10: Cyanobacteria
cyano freezer stock test and attempt
- tested natural transformation on PCC 7002 and PCC 7942 (1mL of culture and DNA from one mini prep each, plated .5mL of 7942 and made 1mL liquid culture, made 2mL liquid culture of 7002) - NP
- inoculated stock cultures of Synechococcus sp PCC 7002 (10:1 in media A) with aeration - NP
- made two types of freezer stocks of PCC 7942, both 25% glycerol, the first 600uL culture to 400uL of 60% glycerol, the second 50mL culture centrifuged at 4000rpm for 10 mins and resuspended in 1mL then 666uL glycerol - NP
- tested freezer stock made on June 22 - NP
|
 "
"






