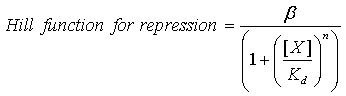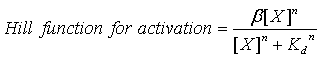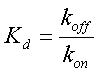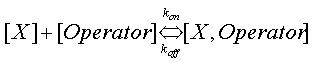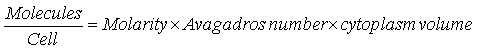Team:Aberdeen Scotland/parameters/invest 1
From 2009.igem.org
Nick Smart (Talk | contribs) |
|||
| (7 intermediate revisions not shown) | |||
| Line 4: | Line 4: | ||
= Overview = | = Overview = | ||
| - | The following section shows how we used our different modeling tools to analyse our original system, find its flaws, change it, and then optimise it to increase glue output, lysis timing and robustness. | + | The following section shows how we used our different modeling tools to analyse our original system, find its flaws, change it, and then optimise it to increase glue output, lysis timing and robustness. This process led to the eventual change of the quorum sensing system but with any investigation, it is important to begin with the basics- in this case, the "dissociation constants" |
{{:Team:Aberdeen_Scotland/break}} | {{:Team:Aberdeen_Scotland/break}} | ||
| - | + | = Dissociation Constants = | |
| - | + | == Introduction == | |
| - | Our model to describe the | + | Our model to describe the transcriptional dynamics of the constructs uses Hill kinetics; we have three repression Hill functions of the form: |
[[Image:Dissociation_Constants_Eq_1.gif|center]] | [[Image:Dissociation_Constants_Eq_1.gif|center]] | ||
| Line 24: | Line 24: | ||
[[Image:Dissociation_Constants_Eq_3.gif|center]] | [[Image:Dissociation_Constants_Eq_3.gif|center]] | ||
| - | where β is the maximal transcription rate, [X] is the concentration of protein X and K<sub>d</sub> is the dissociation constant for molecule X to the promoter in question. Similarly, [S] is the concentration of the inducer S and K<sub>s</sub> is the dissociation constant for the inducer to the repressor X. The | + | where β is the maximal transcription rate, [X] is the concentration of protein X and K<sub>d</sub> is the dissociation constant for molecule X to the promoter in question. Similarly, [S] is the concentration of the inducer S and K<sub>s</sub> is the dissociation constant for the inducer to the repressor X. The dissociation constant K<sub>d</sub> is defined as follows: |
[[Image:Dissociation_Constants_Eq_4.gif|center]] | [[Image:Dissociation_Constants_Eq_4.gif|center]] | ||
| Line 36: | Line 36: | ||
{{:Team:Aberdeen_Scotland/break}} | {{:Team:Aberdeen_Scotland/break}} | ||
| - | + | == Discontinuity == | |
The units of K<sub>d</sub> are usually given in M, the molarity, or moles-per-litre. Our model works with the exact number of molecules, so that we convert the K<sub>d</sub> values into molecules-per-cell. This is achieved as follows: | The units of K<sub>d</sub> are usually given in M, the molarity, or moles-per-litre. Our model works with the exact number of molecules, so that we convert the K<sub>d</sub> values into molecules-per-cell. This is achieved as follows: | ||
| Line 48: | Line 48: | ||
Here, we face a major problem, since most dissociation constants found in the literature equate to a value of molecules per cell that is less than 1. Clearly, in a cell with 10 plasmids and therefore 10 operators, 1 molecule could not repress all of them! | Here, we face a major problem, since most dissociation constants found in the literature equate to a value of molecules per cell that is less than 1. Clearly, in a cell with 10 plasmids and therefore 10 operators, 1 molecule could not repress all of them! | ||
| - | Below is a table of conflicting information we found. This is an extract from the ETHZ Wiki [6] , where a new | + | Below is a table of conflicting information we found. This is an extract from the ETHZ Wiki [6] , where a new column with the values of K<sub>d</sub> in units of molecule-per-cell has been added. |
<html> | <html> | ||
| Line 151: | Line 151: | ||
{{:Team:Aberdeen_Scotland/break}} | {{:Team:Aberdeen_Scotland/break}} | ||
| - | = | + | = Solution = |
Upon further investigation we have concluded that the majority of K<sub>d</sub> values found in papers were unrealistically low for the following reasons: | Upon further investigation we have concluded that the majority of K<sub>d</sub> values found in papers were unrealistically low for the following reasons: | ||
| - | 1. Most K<sub>d</sub> values are measured <i>in-vitro</i>, which yields a low measurement since the conditions of the | + | 1. Most K<sub>d</sub> values are measured <i>in-vitro</i>, which yields a low measurement since the conditions of the reaction - most notably the salt concentration and pressure - are completely different than in an <i>E.coli</i> cell. The salt concentration affects the reaction significantly since it lowers the electrostatic affinity of the protein to the promoter. We know from Thermodynamics that pressure and temperature will change reaction kinetics and hence, the <i>in-vitro</i> experiments will have different reaction rate and hence, different K<sub>d</sub> values than would be found in the cell. |
| - | 2. We have found measurements of K<sub>d</sub> values which have been done in conditions which try to replicate <i>in vivo</i> conditions. These K<sub>d</sub> values are better, but also | + | 2. We have found measurements of K<sub>d</sub> values which have been done in conditions which try to replicate <i>in vivo</i> conditions. These K<sub>d</sub> values are better, but also unfeasibly low, since they do not take into account non-specific DNA binding and cell pressure. |
| - | 3. In our model we describe concentrations in terms of molecules-per-cell, instead of moles-per-litre. Upon converting the K<sub>d</sub> values from moles-per-lire to molecules-per-cell we found that many of the K<sub>d</sub> values were less than 1 molecule per cell. This implies that less than one protein (LacI, TetR, etc.) is required to half the overall production. This is physically | + | 3. In our model we describe concentrations in terms of molecules-per-cell, instead of moles-per-litre. Upon converting the K<sub>d</sub> values from moles-per-lire to molecules-per-cell we found that many of the K<sub>d</sub> values were less than 1 molecule per cell. This implies that less than one protein (LacI, TetR, etc.) is required to half the overall production. This is physically misrepresentative for a number of reasons, including the fact that the probability that one protein molecule will collide with a single promoter in the cell at the correct angle is close to zero. |
| - | We consulted with Prof. Peter McGlynn of the Institute of Medical Sciences in Aberdeen, who agreed with our analysis that the K<sub>d</sub> values were | + | We consulted with Prof. Peter McGlynn of the Institute of Medical Sciences in Aberdeen, who agreed with our analysis that the K<sub>d</sub> values were unfeasibly low and introduced the idea of non-specific DNA binding to us. He showed us a PhD thesis from one of his students, Dr. Bryony Payne, from 2006. In this thesis a far more direct and accurate measurement of the number of LacI tetramers present in the cell was made. It stated that on average 340 tetramers of LacI were present in an e. coli cell. Since under normal conditions (when glucose is available and no lactose is present) the LacO operon is repressed. We can assume that 340 tetramers will fully repress the lacO operon. From this value, we estimated the K<sub>d</sub> value for the LacO operon to be 700 molecules-per-cell. |
{{:Team:Aberdeen_Scotland/break}} | {{:Team:Aberdeen_Scotland/break}} | ||
| - | + | == Our new estimations for K<sub>d</sub>== | |
| - | According to Dr. Payne's 2006 paper; 340 LacI tetramers completely repress a promoter. Hence, roughly 170 tetramers will give half repression. Assuming the tetramers are stable, this gives a value of K<sub>LacI</sub> - K<sub>d</sub> for LacI to LacO - of | + | According to Dr. Payne's 2006 paper; 340 LacI tetramers completely repress a promoter. Hence, roughly 170 tetramers will give half repression. Assuming the tetramers are stable, this gives a value of K<sub>LacI</sub> - K<sub>d</sub> for LacI to LacO - of 4×170 or K<sub>LacI</sub> ~ 700. |
For the LacI-IPTG complex formation, we estimated K<sub>IPTG</sub> ~1200 using [7] and our value of K<sub>LacI</sub> above. | For the LacI-IPTG complex formation, we estimated K<sub>IPTG</sub> ~1200 using [7] and our value of K<sub>LacI</sub> above. | ||
TetR to tetO have a lower affinity to each other than LacI to LacO. However, the <i>in-vitro</i> values suggest that TetR still binds with a strong affinity to TetR. Thus the K<sub>TetR</sub> value was roughly estimated to be up to 10 times K<sub>LacI</sub>. | TetR to tetO have a lower affinity to each other than LacI to LacO. However, the <i>in-vitro</i> values suggest that TetR still binds with a strong affinity to TetR. Thus the K<sub>TetR</sub> value was roughly estimated to be up to 10 times K<sub>LacI</sub>. | ||
| - | The <i>in-vitro</i> values for cI to its operon seem to suggest that the <i>in-vivo</i> K<sub>cI</sub> value is of the same order of magnitude, but | + | The <i>in-vitro</i> values for cI to its operon seem to suggest that the <i>in-vivo</i> K<sub>cI</sub> value is of the same order of magnitude, but possibly smaller, than K<sub>TetR</sub>. In <i>V.fischeri</i>, it is unlikely that HSL enters the cell before the amplifying loop starts. Assuming this, the complex HSL-LuxR (P) has to have a high affinity for the lux box same as LacI to lacO. |
Now we have: | Now we have: | ||
| Line 189: | Line 189: | ||
<tr> | <tr> | ||
<td> | <td> | ||
| - | <a href="https://2009.igem.org/Team:Aberdeen_Scotland/ | + | <a href="https://2009.igem.org/Team:Aberdeen_Scotland/chemotaxis"><img src="https://static.igem.org/mediawiki/2009/e/ed/Aberdeen_Left_arrow.png"> Back to Chemotaxis</a> |
</td> | </td> | ||
<td align="right"> | <td align="right"> | ||
| Line 199: | Line 199: | ||
{{:Team:Aberdeen_Scotland/break}} | {{:Team:Aberdeen_Scotland/break}} | ||
| - | + | = References = | |
[1] Mitchel Lewis (2005) The Lac repressor. C. R. Biologies 328 (2005) 521–548 | [1] Mitchel Lewis (2005) The Lac repressor. C. R. Biologies 328 (2005) 521–548 | ||
| Line 213: | Line 213: | ||
[5] Volume 272, Number 11, Issue of March 14, 1997 pp. 6936-6942, The Role of the Variable Region in Tet Repressor for Inducibility by Tetracycline, Christian Berens , Dirk Schnappinger and Wolfgang Hillen | [5] Volume 272, Number 11, Issue of March 14, 1997 pp. 6936-6942, The Role of the Variable Region in Tet Repressor for Inducibility by Tetracycline, Christian Berens , Dirk Schnappinger and Wolfgang Hillen | ||
| - | [6] | + | [6] https://2007.igem.org/title=ETHZ/Parameters |
[7] Detailed map of a cis-regulatory input function – Y. Setty*,†, A. E. Mayo*,†, M. G. Surette‡, and U. Alon*,†,§ | [7] Detailed map of a cis-regulatory input function – Y. Setty*,†, A. E. Mayo*,†, M. G. Surette‡, and U. Alon*,†,§ | ||
Latest revision as of 17:27, 21 October 2009
University of Aberdeen - Pico Plumber
Contents |
Overview
The following section shows how we used our different modeling tools to analyse our original system, find its flaws, change it, and then optimise it to increase glue output, lysis timing and robustness. This process led to the eventual change of the quorum sensing system but with any investigation, it is important to begin with the basics- in this case, the "dissociation constants"
Dissociation Constants
Introduction
Our model to describe the transcriptional dynamics of the constructs uses Hill kinetics; we have three repression Hill functions of the form:
Moreovre, the model has one activation Hill function of the following form:
and one repression / induction Hill function of the following form
where β is the maximal transcription rate, [X] is the concentration of protein X and Kd is the dissociation constant for molecule X to the promoter in question. Similarly, [S] is the concentration of the inducer S and Ks is the dissociation constant for the inducer to the repressor X. The dissociation constant Kd is defined as follows:
where koff and kon are the on and off rates in the following reaction
The dissociation constant Kd has more biologically meaningful definition; it is the concentration of X at which the promoter will be free 50% of the time.
Discontinuity
The units of Kd are usually given in M, the molarity, or moles-per-litre. Our model works with the exact number of molecules, so that we convert the Kd values into molecules-per-cell. This is achieved as follows:
Where the volume of the cytoplasm of the cell is 6.7×10-16 litres
This conversion constant of Avogadro’s number multiplied by the cytoplasm volume is ~ 402000000 (402 million).
Here, we face a major problem, since most dissociation constants found in the literature equate to a value of molecules per cell that is less than 1. Clearly, in a cell with 10 plasmids and therefore 10 operators, 1 molecule could not repress all of them!
Below is a table of conflicting information we found. This is an extract from the ETHZ Wiki [6] , where a new column with the values of Kd in units of molecule-per-cell has been added.
| Parameter | Value | Molecules per cell | Description |
| KLacI | 0.1 - 1 [pM] OR 800 [nM] | 0.00004-0.0004 molecules OR 322 molecules | LacI repressor dissociation constant |
| KIPTG | 1.3 [µM] | 522 molecules | IPTG-LacI repressor dissociation constant |
| KtetR | 179 [pM] | 0.07 molecules | TetR repressor dissociation constant |
| KcI | 8 [pM] OR 50 [nM] | 0.003 molecules OR 20 molecules | cI repressor dissociation constant |
| KHSL | 0.09 - 1 [µM] | 402 molecules | HSL-LuxR activator dissociation constant |
And here are other parameters that we found in the literature:
| Parameter | Value | Molecules per cell | Description | Reference |
| KLacI | ~1*10 -12 M OR ~1.8*10-12 M | 0.0004 molecules OR 0.00072 molecules | Dissociation constant for LacI to LacO DNA site | [1][2] |
| KIPTG | 1*10-6 M | 402 molecules | Dissociation constant for IPTG to LacI | [3] |
| KtetR | (5.6 ± 2) × 10-9 M OR 1.53*10-8 M | 2.25 molecules OR 6.1506 molecules | Dissociation constant for TetR to TetO | [4][5] |
| KcI | 50 * 10-9 M | 20 molecules | Dissocitation constant for cI to DNA site | [6] |
Solution
Upon further investigation we have concluded that the majority of Kd values found in papers were unrealistically low for the following reasons:
1. Most Kd values are measured in-vitro, which yields a low measurement since the conditions of the reaction - most notably the salt concentration and pressure - are completely different than in an E.coli cell. The salt concentration affects the reaction significantly since it lowers the electrostatic affinity of the protein to the promoter. We know from Thermodynamics that pressure and temperature will change reaction kinetics and hence, the in-vitro experiments will have different reaction rate and hence, different Kd values than would be found in the cell.
2. We have found measurements of Kd values which have been done in conditions which try to replicate in vivo conditions. These Kd values are better, but also unfeasibly low, since they do not take into account non-specific DNA binding and cell pressure.
3. In our model we describe concentrations in terms of molecules-per-cell, instead of moles-per-litre. Upon converting the Kd values from moles-per-lire to molecules-per-cell we found that many of the Kd values were less than 1 molecule per cell. This implies that less than one protein (LacI, TetR, etc.) is required to half the overall production. This is physically misrepresentative for a number of reasons, including the fact that the probability that one protein molecule will collide with a single promoter in the cell at the correct angle is close to zero.
We consulted with Prof. Peter McGlynn of the Institute of Medical Sciences in Aberdeen, who agreed with our analysis that the Kd values were unfeasibly low and introduced the idea of non-specific DNA binding to us. He showed us a PhD thesis from one of his students, Dr. Bryony Payne, from 2006. In this thesis a far more direct and accurate measurement of the number of LacI tetramers present in the cell was made. It stated that on average 340 tetramers of LacI were present in an e. coli cell. Since under normal conditions (when glucose is available and no lactose is present) the LacO operon is repressed. We can assume that 340 tetramers will fully repress the lacO operon. From this value, we estimated the Kd value for the LacO operon to be 700 molecules-per-cell.
Our new estimations for Kd
According to Dr. Payne's 2006 paper; 340 LacI tetramers completely repress a promoter. Hence, roughly 170 tetramers will give half repression. Assuming the tetramers are stable, this gives a value of KLacI - Kd for LacI to LacO - of 4×170 or KLacI ~ 700.
For the LacI-IPTG complex formation, we estimated KIPTG ~1200 using [7] and our value of KLacI above. TetR to tetO have a lower affinity to each other than LacI to LacO. However, the in-vitro values suggest that TetR still binds with a strong affinity to TetR. Thus the KTetR value was roughly estimated to be up to 10 times KLacI. The in-vitro values for cI to its operon seem to suggest that the in-vivo KcI value is of the same order of magnitude, but possibly smaller, than KTetR. In V.fischeri, it is unlikely that HSL enters the cell before the amplifying loop starts. Assuming this, the complex HSL-LuxR (P) has to have a high affinity for the lux box same as LacI to lacO.
Now we have:
KLacI = 700 molecules per cell
KIPTG = 1200 molecules per cell
KP = 700 molecules per cell
KcI = 7000 molecules per cell
KTetR = 7000 molecules per cell
 Back to Chemotaxis Back to Chemotaxis
|
Continue to Sensitvity Simulations 
|
References
[1] Mitchel Lewis (2005) The Lac repressor. C. R. Biologies 328 (2005) 521–548
[2] Falcon C.M and Matthews K.S. (2000) Operator DNA sequence Variation Enhances High Affinity Binding by Hinge Helix Mutants of Lactose Repressor Protein. Biochemistry. 39, 11074-11084
[3] Uri Alon, An introduction to systems Biology, p244
[4] Nucleic Acids Res. 2004; 32(2): 842–847. Two mutations in the tetracycline repressor change the inducer anhydrotetracycline to a corepressor Annette Kamionka, Joanna Bogdanska-Urbaniak, Oliver Scholz, and Wolfgang Hillen*
[5] Volume 272, Number 11, Issue of March 14, 1997 pp. 6936-6942, The Role of the Variable Region in Tet Repressor for Inducibility by Tetracycline, Christian Berens , Dirk Schnappinger and Wolfgang Hillen
[6] https://2007.igem.org/title=ETHZ/Parameters
[7] Detailed map of a cis-regulatory input function – Y. Setty*,†, A. E. Mayo*,†, M. G. Surette‡, and U. Alon*,†,§
 "
"