Team:British Columbia/pBAD
From 2009.igem.org
(→Overview) |
(→Overview) |
||
| Line 3: | Line 3: | ||
==Overview== | ==Overview== | ||
In order to make an analog biosensor, we need our traffic light to produce distinct, unique responses to a range of concentrations of an input. However, the Registry is lacking in variable strength inducible promoters. We designed two variants of the <partinfo>I13453</partinfo> pBAD promoter, one weaker and one stronger than wild type, based on AraC binding experiments performed by [[Team:British_Columbia/Bibliography|Niland et al.]] | In order to make an analog biosensor, we need our traffic light to produce distinct, unique responses to a range of concentrations of an input. However, the Registry is lacking in variable strength inducible promoters. We designed two variants of the <partinfo>I13453</partinfo> pBAD promoter, one weaker and one stronger than wild type, based on AraC binding experiments performed by [[Team:British_Columbia/Bibliography|Niland et al.]] | ||
| + | |||
| + | ===pBAD Mutagenesis=== | ||
[[Image:E_coli_Traffic_Light_Mutated_Promoters.png|left|thumb||500px|A diagram of the changed nucleotide sequences of arabinose-inducible promoters. Green nucleotides of the wildtype show all sequences that were mutated from the wild type to form strong and weak.]] | [[Image:E_coli_Traffic_Light_Mutated_Promoters.png|left|thumb||500px|A diagram of the changed nucleotide sequences of arabinose-inducible promoters. Green nucleotides of the wildtype show all sequences that were mutated from the wild type to form strong and weak.]] | ||
| Line 15: | Line 17: | ||
Above is a sequence alignment of our promoter variants with the wild type pBAD, truncated for space. For the full sequences, see our parts in the registry at <partinfo>K206000</partinfo> (strong) and <partinfo>K206001</partinfo> (weak). | Above is a sequence alignment of our promoter variants with the wild type pBAD, truncated for space. For the full sequences, see our parts in the registry at <partinfo>K206000</partinfo> (strong) and <partinfo>K206001</partinfo> (weak). | ||
| + | |||
| + | ===Results=== | ||
<html>We assembled each promoter with the RFP reporter part <partinfo>I13507</partinfo> in order to test the relative activity of the mutated promoters</html> | <html>We assembled each promoter with the RFP reporter part <partinfo>I13507</partinfo> in order to test the relative activity of the mutated promoters</html> | ||
| Line 21: | Line 25: | ||
[[Image:Toxicity.jpg]] | [[Image:Toxicity.jpg]] | ||
[[Image:Trans_fcn.jpg]] | [[Image:Trans_fcn.jpg]] | ||
| + | |||
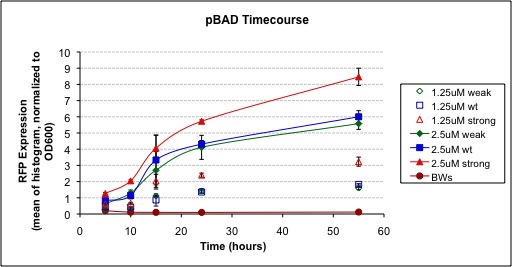
| + | We have been able to successfully show that at low arabinose concentrations, the activity of the Strong pBAD promoter and Weak pBAD promoter following arabinose induction is, as expected, greater and lesser respectively then the Wild Type pBAD promoter. By examining the development of a RFP reporter, it is observed that the Strong pBAD promoter has both a faster rate of development and reaches a higher maximum value compared to the Wild Type sequence. Similarly, the Weak pBAD promoter develops slower and to a lower maximum intensity. Additionally, we have shown that Strong pBAD and Weak pBAD are more and less responsive respectively to lower concentrations of arabinose then the Wild Type promoter. Therefore, they could be suitable to be used in conjunction as a bio-sensor. | ||
Revision as of 00:31, 22 October 2009
Home Team Traffic Light Sensor Lock&Key Jammer [http://partsregistry.org/cgi/partsdb/pgroup.cgi?pgroup=iGEM2009&group=British_Columbia Parts] Safety Sponsors Notebook Bibliography
Contents |
Arabinose sensor: the pBAD promoter
Overview
In order to make an analog biosensor, we need our traffic light to produce distinct, unique responses to a range of concentrations of an input. However, the Registry is lacking in variable strength inducible promoters. We designed two variants of the pBAD promoter, one weaker and one stronger than wild type, based on AraC binding experiments performed by Niland et al.
pBAD Mutagenesis
Above is a sequence alignment of our promoter variants with the wild type pBAD, truncated for space. For the full sequences, see our parts in the registry at (strong) and (weak).
Results
We assembled each promoter with the RFP reporter part
We have been able to successfully show that at low arabinose concentrations, the activity of the Strong pBAD promoter and Weak pBAD promoter following arabinose induction is, as expected, greater and lesser respectively then the Wild Type pBAD promoter. By examining the development of a RFP reporter, it is observed that the Strong pBAD promoter has both a faster rate of development and reaches a higher maximum value compared to the Wild Type sequence. Similarly, the Weak pBAD promoter develops slower and to a lower maximum intensity. Additionally, we have shown that Strong pBAD and Weak pBAD are more and less responsive respectively to lower concentrations of arabinose then the Wild Type promoter. Therefore, they could be suitable to be used in conjunction as a bio-sensor.
 "
"