Team:Aberdeen Scotland/parameters/invest 4
From 2009.igem.org
Nick Smart (Talk | contribs) |
Nick Smart (Talk | contribs) (→Possible Solution) |
||
| (50 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
{{:Team:Aberdeen_Scotland/css}} | {{:Team:Aberdeen_Scotland/css}} | ||
{{:Team:Aberdeen_Scotland/header2}} | {{:Team:Aberdeen_Scotland/header2}} | ||
| + | = Quorum Sensing = | ||
| - | + | One of the triggering mechanisms of our Pico plumber in order to start glue production, is quorum sensing. Quorum sensing is a response of bacteria to their population density. In the natural system of the marine bacterium Vibro fischeri a small molecule, termed autoinducer, is constantly produced at a low level. This autoinducer molecule (HSL) can freely diffuse in and out the E.Coli membrane. The HSL molecule then diffuses into the surroundings. Other bacteria in the surrounding environment can sense each other via diffusion of HSL through their cell membrane. In the absence of a high density of cells HSL rapidly diffuses into the environment and the HSL concentration is too low to trigger quorum sensing. | |
| + | Quorum sensing is based on a lux-type regulated transcriptional system controlling the production of the autoinducer. In Vibro fischeri, LuxR is encoded on the left side of the operon whereas LuxI is produced on the right side. LuxI together wuth an enzyme always present in the cell, called SAM, together generate in an enzymatic reaction the autoinducer, HSL. The autoinducer HSL together with LuxR form a transcriptional activator for the lux operon, also known as lux box. This complex enables a stronger production of LuxI and LuxR and can been seen as an amplifying loop for LuxI production [1-3]. | ||
| - | + | {{:Team:Aberdeen_Scotland/break}} | |
| - | + | == The issue == | |
| + | In the planning stage of the project, we designed the quorum sensing circuit to have a constant medium production of LuxI in order to activate a lux box on the plasmid where the glue production takes place, in the presence of sufficient cell density: | ||
| + | [[Image:Qs_invest_1.jpg|center|400px]] | ||
| + | [[Image:Qs_invest_2.jpg|center|400px]] | ||
| - | + | We are using the lux operon and LacO to give us AND–logic gate to start glue production (here denoted as gene X and gene Y). It is expected that if IPTG is present and there is no HSL within the cell, the promoter starts to produce the glue at a low level. In the presence of HSL and IPTG the promoter starts fabrication of the glue at its maximum level. | |
| + | A possible source of error in our design is that our system might trigger glue production and cell lysis on its own. Having in mind that we have a constitutive medium strength promoter, we need a lower concentration of LuxI. To achieve this LuxI was tagged and a Shine-Dalgarno sequence was used to reuce ribosome affinity to 60%. | ||
| - | + | {{:Team:Aberdeen_Scotland/break}} | |
| + | == The Model == | ||
| + | We ran our simulation using the IPTG and HSL concentration outside the cell as inputs. We changed both IPTG and HSL concentration at different times by a switch from low to high to see how the inputs affected the simulation. In our later simulations we simulated both the IPTG and HSL inputs more realistically. We did this applying the sochastic simulation (tau-leap method). | ||
| + | [[Image:Qs_invest_3.jpg|center|700px]] | ||
| - | + | The graph shows the concentration of molecules against the iteration number (which is equivalent to time multiplied by a time-step constant of h=0.5). GFP is a place holder for the glue, P stands for the complex of HSL and LuxR, and finally, IPTG and HSL are the input signals for our cell. IPTG is released at 15000 iterations, whereas HSL diffuses from the outside at 25000 iterations. We want a system to have AND-logic gate, so that both high IPTG and HSL concentration is required for system response. However, it is clearly visible that maximal production of GFP already starts shortly after IPTG gets into the cell. | |
| + | We can conclude from this that the lux-box is activated from within the cell. TThe high production of LuxI and hence, HSl, leads to a high concentration of the HSl-LuxR complex. Moreover, the HSL-LuxR complex is ver stable. This complex cannot diffuse out of the cell as HSL can, so that its concentration can build up very much and hence, activate the lux-box. | ||
| - | + | {{:Team:Aberdeen_Scotland/break}} | |
| - | + | = Possible Solution = | |
| - | + | To try to overcome this problem, we have redesigned the system to have LuxI and LuxR on the same promoter but with a lux box in front of both. For this model, we no longer assume the trivial process of repression of LacI being lifted and Quorum Sensing (QS) being turned on. The LacI repression is now a repression / induction hill function with a K<sub>d</sub> of 1200 molecules per cell for IPTG forming the LacI-IPTG complex. We assume IPTG leaks into the cell from outside. We only set the outside concentration of IPTG. | |
| + | For quorum sensing all simply set an elevated exterior HSL concentration which is free to diffuse in. We now include a feedback loop into the QS mechanism. To do this we attach a lux box onto the plasmid producing LuxR and LuxI, as follows: | ||
| - | |||
| - | |||
| - | + | [[Image:Input Model Graph 1.jpg|center|500px]] | |
| - | + | However, the production strength of this lux box is smaller than that of the lux box for producing X, Y and cI. The strengths are as follows: | |
| + | Production max for X, Y and cI = 0.44 PoPS (Polymerases-per-second) | ||
| - | + | Production min for X Y and cI = 0.013 PoPS | |
| - | + | Production max for QS feedback = 0.002 PoPS | |
| - | + | Production min for QS feedback = 0.00015 PoPS | |
| + | (check our "parameters" section for references) | ||
| - | + | An important new parameter is K<sub>P</sub>. This is the dissociation constant for “P” activating the lux boxes, where P is the LuxR-HSL complex. In the next section we show how the most important results obtained with this model. | |
| - | + | ||
| - | + | {{:Team:Aberdeen_Scotland/break}} | |
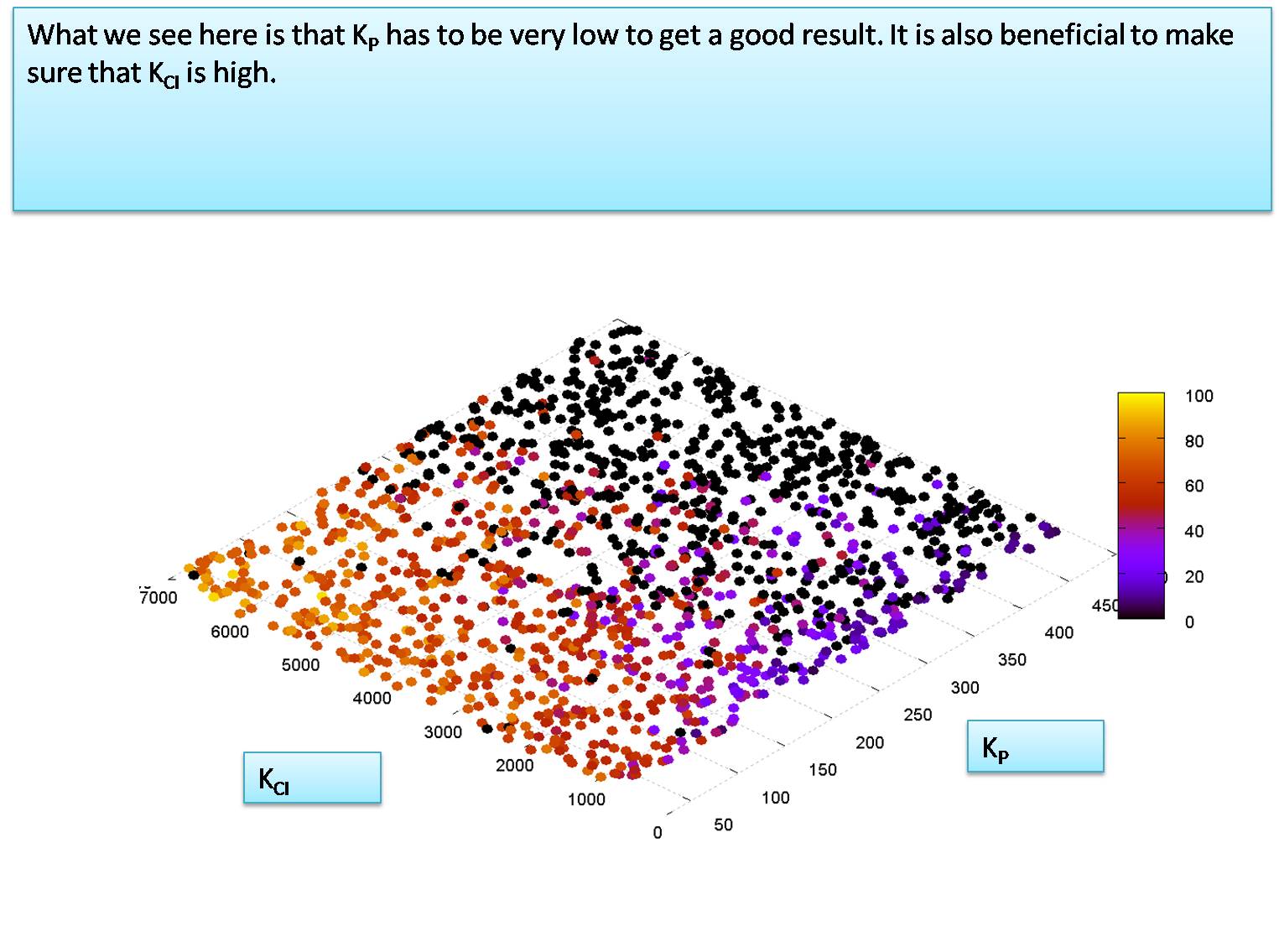
| - | + | =Level of K<sub>P</sub> required for system to function = | |
| + | here we use a Monte Carlo simulation to determine the value of K<sub>P</sub> in molecules per cell that is required for the system to function (note that the IPTG and HSL input levels added are both 10000 molecules) | ||
| - | + | [[Image:Input Model Graph 10.jpg|centre|700px]] | |
| + | [[Image:Input Model Graph 11.jpg|centre|700px]] | ||
| - | + | {{:Team:Aberdeen_Scotland/break}} | |
| - | + | ||
| - | + | ||
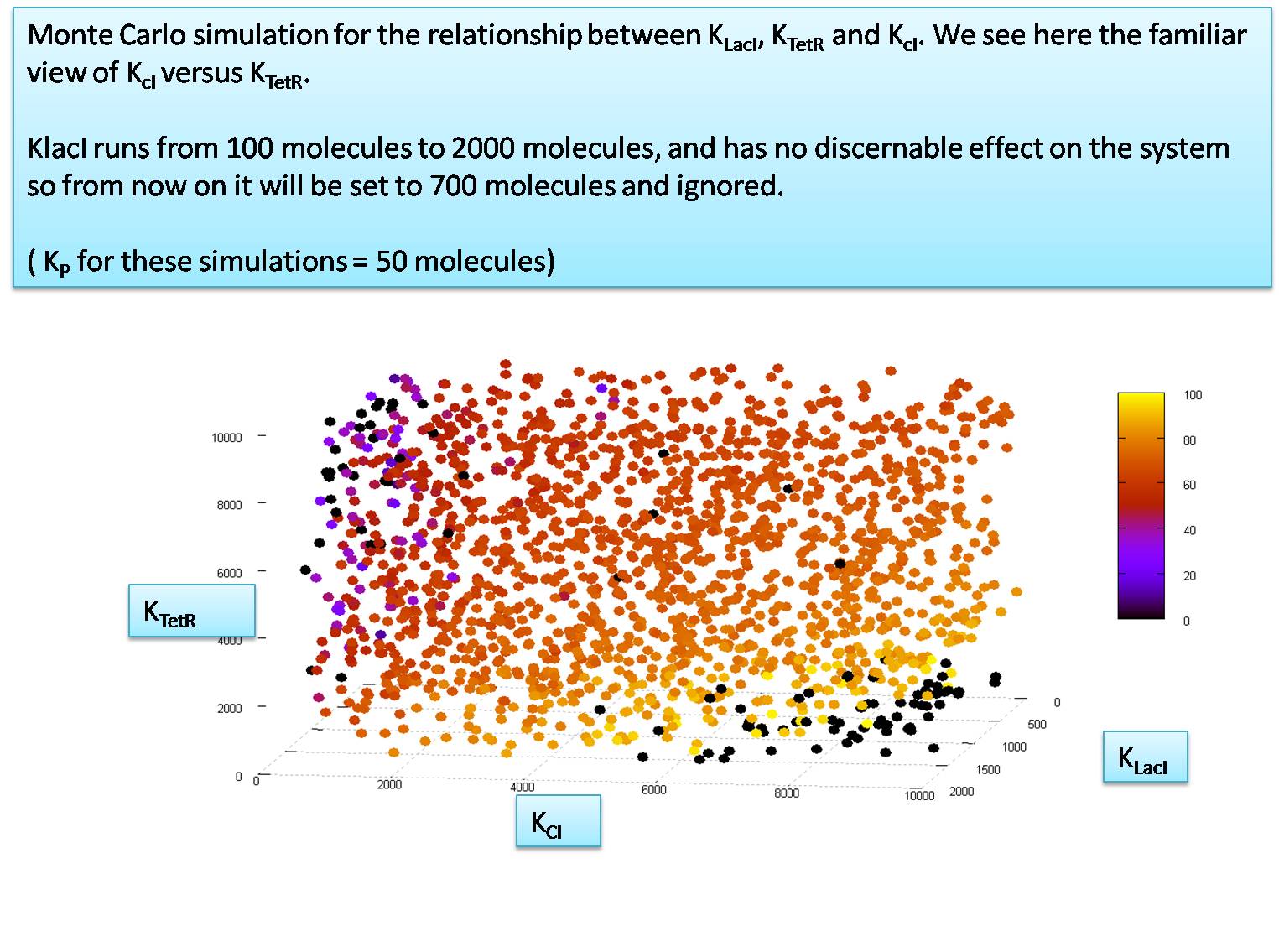
| - | K<sub>P</sub> = | + | =Relationship between K<sub>TetR</sub>, K<sub>CI</sub> and K<sub>LacI</sub> when K<sub>P</sub> is 50 = |
| + | [[Image:Input Model Graph 4.jpg|centre|700px]] | ||
| - | |||
{{:Team:Aberdeen_Scotland/break}} | {{:Team:Aberdeen_Scotland/break}} | ||
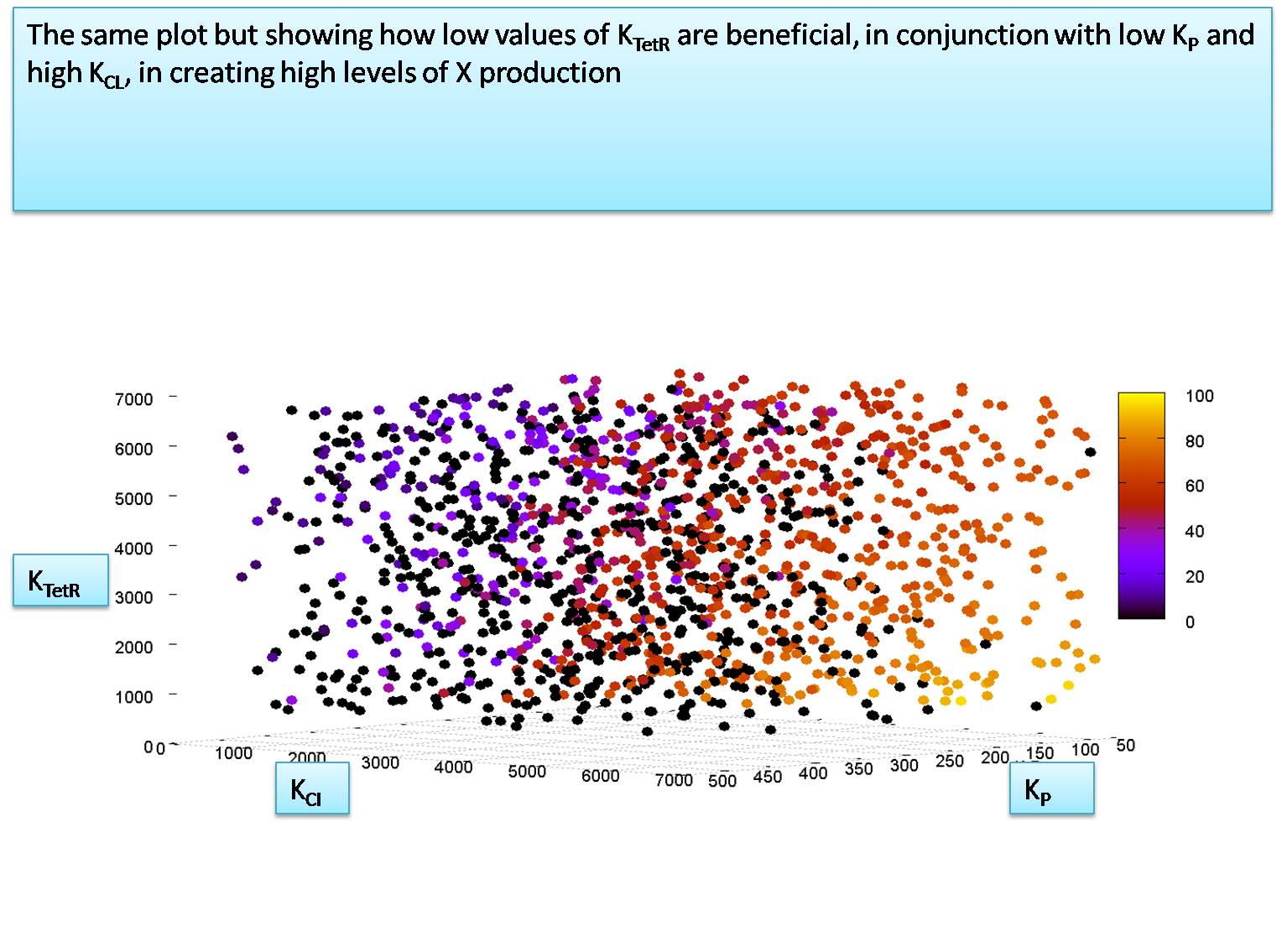
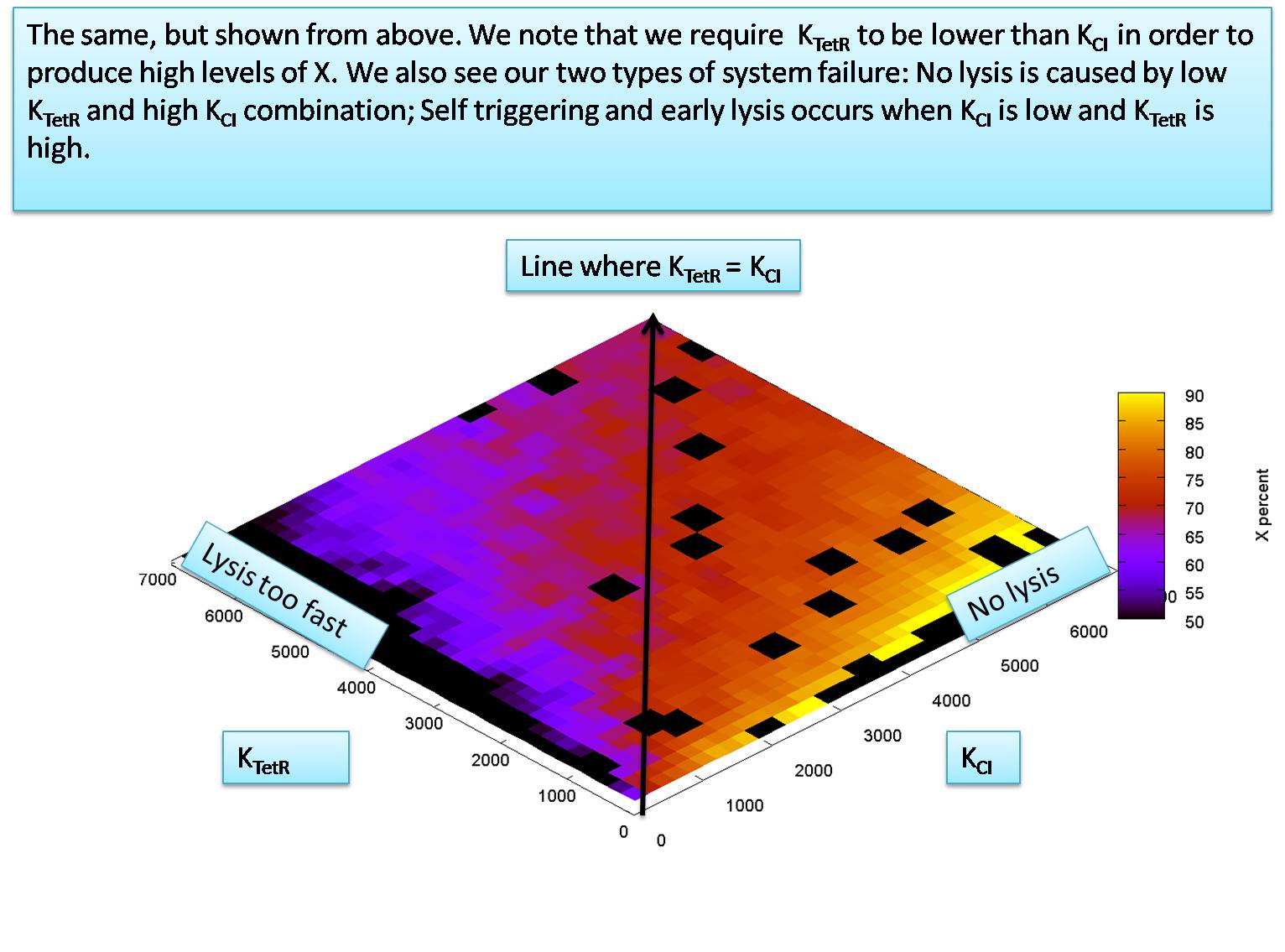
| + | =Relationship between K<sub>TetR</sub> and K<sub>CI</sub> when K<sub>P</sub> is 50 and K<sub>LacI</sub> is 700 = | ||
| - | |||
| - | |||
| + | We notice that K<sub>P</sub> must be around 50 molecules to let the system function. We will now explore the relationship between K<sub>TetR</sub> and K<sub>CI</sub> at the value of K<sub>P</sub>=50 molecules per cell. | ||
| + | for this simulation we set the HSL and IPTG input levels to 50,000 molecules. In our next section we will try to calculate the minimum levels of HSL and IPTG required to activate the system correctly. | ||
| - | |||
| - | |||
| + | [[Image:Input Model Graph 8.jpg|centre|700px]] | ||
| + | [[Image:Input Model Graph 9.jpg|centre|700px]] | ||
| + | The graph shows a great deal of stochastic fluctuation, which shows this system is not very robust. Increasing K<sub>P</sub> above 50 greatly increases this problem, as does decreasing HSL outside below 50,000. | ||
| - | + | Our estimation for K<sub>TetR</sub> and K<sub>CI</sub> suggest that K<sub>TetR</sub> is less than K<sub>CI</sub>. We assume the worst case value for these two dissociation constants and set them equal to each other, at K<sub>TetR</sub> = K<sub>CI</sub> = 7000 molecules per cell. | |
| + | {{:Team:Aberdeen_Scotland/break}} | ||
| - | + | = Required input levels of IPTG and HSL = | |
| - | |||
| - | [[Image:Input Model Graph | + | [[Image:Input Model Graph 2.jpg|centre|700px]] |
| - | |||
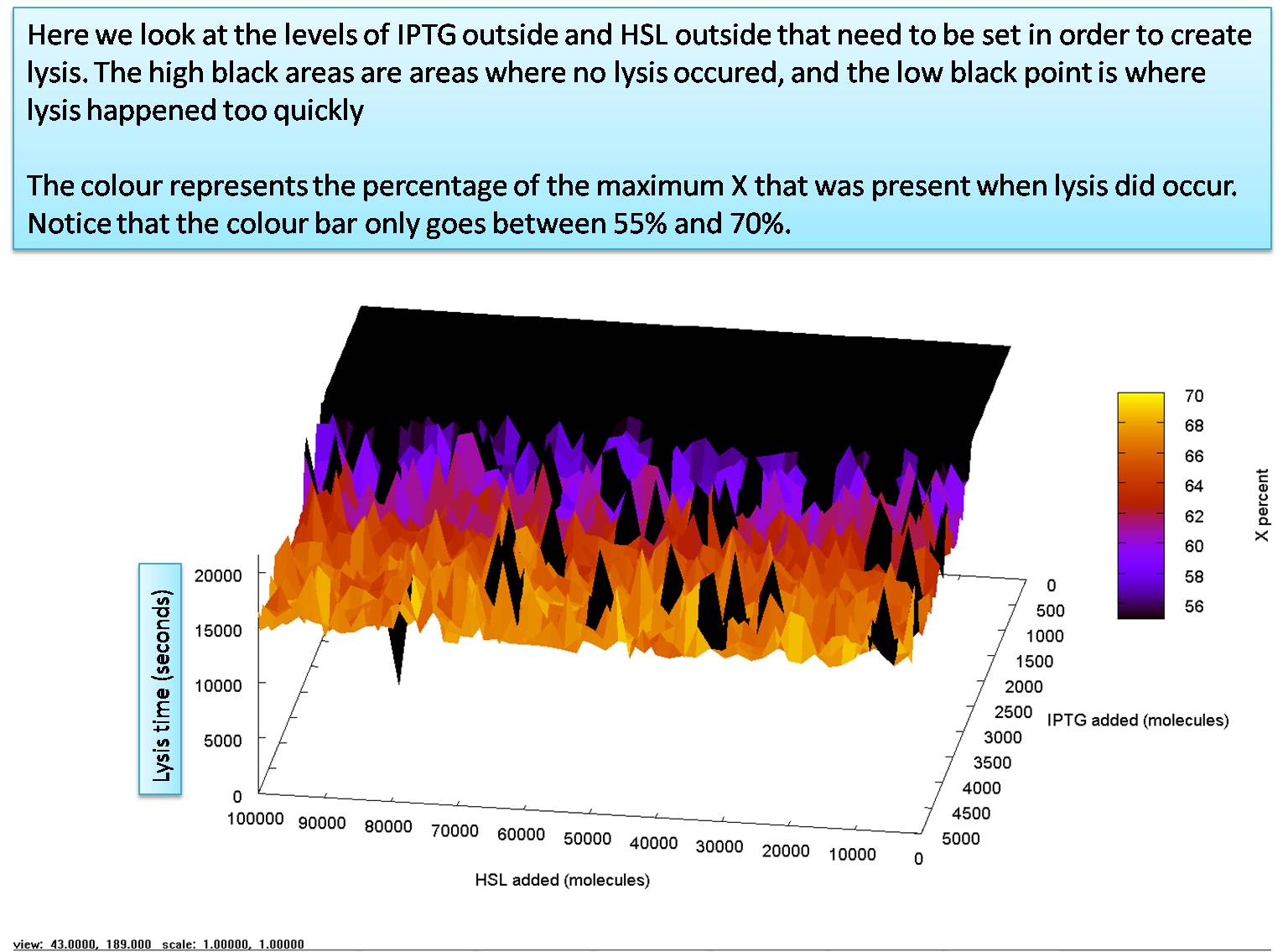
| + | We start by going from 5,000 molecules to 100,000 molecules for HSL and from 10 molecules to 5,000 molecules for IPTG. we see that the sysem requires only around 2000 molecules of IPTG outside, although it benefits slighty when it is exposed to more. Note that the graphs color bar only extends from 55% of X production to 70% of X production. There appears to be no disernable effect in increasing the HSL outside, although when it is above 70,000 it seems to reduce the stochastic effects that stop the system lysing. | ||
| + | We now look at a graph showing HSL at values below 5000 molecules. | ||
| + | |||
| + | [[Image:Input Model Graph 3.jpg|centre|700px]] | ||
{{:Team:Aberdeen_Scotland/break}} | {{:Team:Aberdeen_Scotland/break}} | ||
| - | = | + | = Optimal Operation = |
| + | Here are the following assumptions that need to make to get the optimal system performance | ||
| - | + | K<sub>P</sub> has to be ~50 molecules per cell | |
| - | + | K<sub>TetR</sub> has to be ~3000 molecules per cell | |
| - | + | K<sub>CI</sub> has to be ~ 5000 molecules per cell | |
| - | + | the input level of HSL has to be ~10,000 molecules | |
| - | + | the input level of IPTG has to be ~5,000 molecules | |
| + | |||
| + | [[Image:Input Model Graph 12.jpg|centre|700px]] | ||
| + | [[Image:Input Model Graph 13.jpg|centre|700px]] | ||
{{:Team:Aberdeen_Scotland/break}} | {{:Team:Aberdeen_Scotland/break}} | ||
| - | + | = Actual Operation = | |
| - | + | The graphs above show how the system would function with our carefully calculated optimum parameter set. However, we cannot guarantee that the actual parameters will be close to these estimates. while the input levels of HSL and IPTG are reasonable, our values of K<sub>CI</sub> and K<sub>TetR</sub> may well be almost equal to each other, and around 7000 molecules per cell. While changing these to 7000 would reduce the level of the glue production at lysis the killing blow for this model is that K<sub>P</sub> is almost definitely larger than 50 molecules per cell. | |
| - | + | The following graph shows how the system functions at the values of K<sub>d</sub> we believe to be most likely: | |
| - | [[Image:Input Model Graph | + | [[Image:Input Model Graph 14.jpg|centre|700px]] |
| - | |||
| - | + | {{:Team:Aberdeen_Scotland/break}} | |
| - | + | = Conclusions = | |
| - | + | As we have seen above, our system is very stochastically unstable, and extremly sensitive to the parameters we have investigated. As our best estimates of these parameters do not fall close to the optimal range required for the quorum sensing system we must redesign it. In the next section we amended our quorum sensing system again, to try to produce the correct response to our inputs. | |
| - | + | <html> | |
| + | <table class="nav"> | ||
| + | <tr> | ||
| + | <td> | ||
| + | <a href="https://2009.igem.org/Team:Aberdeen_Scotland/parameters/invest_3"><img src="https://static.igem.org/mediawiki/2009/e/ed/Aberdeen_Left_arrow.png"> Back to Sensitivity Simulations</a> | ||
| + | </td> | ||
| + | <td align="right"> | ||
| + | <a href="https://2009.igem.org/Team:Aberdeen_Scotland/parameters/invest_5">Continue to the Amended Model <img src="https://static.igem.org/mediawiki/2009/4/4c/Aberdeen_Right_arrow.png"></a> | ||
| + | </td> | ||
| + | </tr> | ||
| + | </table> | ||
| + | </html> | ||
| + | |||
| + | {{:Team:Aberdeen_Scotland/break}} | ||
| - | + | = Bibliography = | |
| + | (1) Goryachev. Systems analysis of a quorum sensing network: Design constraints imposed by the functional requirements, network topology and kinetic constants. BioSystems 2006;83(2-3 SPEC. ISS.):178. | ||
| - | + | (2) James. Luminescence control in the marine bacterium Vibrio fischeri: An analysis of the dynamics of lux regulation. J.Mol.Biol. 2000;296(4):1127. | |
| + | (3) Ward JP. Mathematical modelling of quorum sensing in bacteria. IMA. 2001;18:263-292. | ||
| + | (4) <html><a href="https://2009.igem.org/Team:Aberdeen_Scotland/internal/stochastic">Link to stochastic Equations</a></html> | ||
{{:Team:Aberdeen_Scotland/footer}} | {{:Team:Aberdeen_Scotland/footer}} | ||
Latest revision as of 17:18, 18 August 2009
University of Aberdeen - Pico Plumber
Quorum Sensing
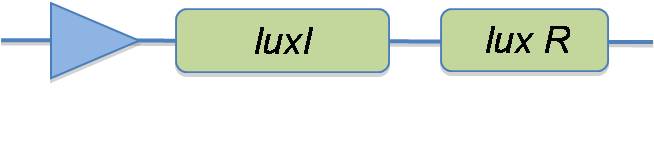
One of the triggering mechanisms of our Pico plumber in order to start glue production, is quorum sensing. Quorum sensing is a response of bacteria to their population density. In the natural system of the marine bacterium Vibro fischeri a small molecule, termed autoinducer, is constantly produced at a low level. This autoinducer molecule (HSL) can freely diffuse in and out the E.Coli membrane. The HSL molecule then diffuses into the surroundings. Other bacteria in the surrounding environment can sense each other via diffusion of HSL through their cell membrane. In the absence of a high density of cells HSL rapidly diffuses into the environment and the HSL concentration is too low to trigger quorum sensing. Quorum sensing is based on a lux-type regulated transcriptional system controlling the production of the autoinducer. In Vibro fischeri, LuxR is encoded on the left side of the operon whereas LuxI is produced on the right side. LuxI together wuth an enzyme always present in the cell, called SAM, together generate in an enzymatic reaction the autoinducer, HSL. The autoinducer HSL together with LuxR form a transcriptional activator for the lux operon, also known as lux box. This complex enables a stronger production of LuxI and LuxR and can been seen as an amplifying loop for LuxI production [1-3].
The issue
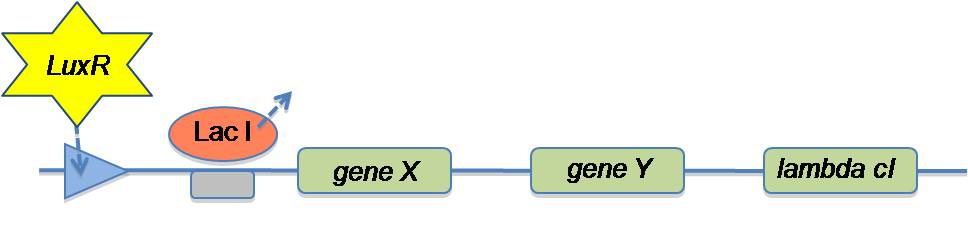
In the planning stage of the project, we designed the quorum sensing circuit to have a constant medium production of LuxI in order to activate a lux box on the plasmid where the glue production takes place, in the presence of sufficient cell density:
We are using the lux operon and LacO to give us AND–logic gate to start glue production (here denoted as gene X and gene Y). It is expected that if IPTG is present and there is no HSL within the cell, the promoter starts to produce the glue at a low level. In the presence of HSL and IPTG the promoter starts fabrication of the glue at its maximum level. A possible source of error in our design is that our system might trigger glue production and cell lysis on its own. Having in mind that we have a constitutive medium strength promoter, we need a lower concentration of LuxI. To achieve this LuxI was tagged and a Shine-Dalgarno sequence was used to reuce ribosome affinity to 60%.
The Model
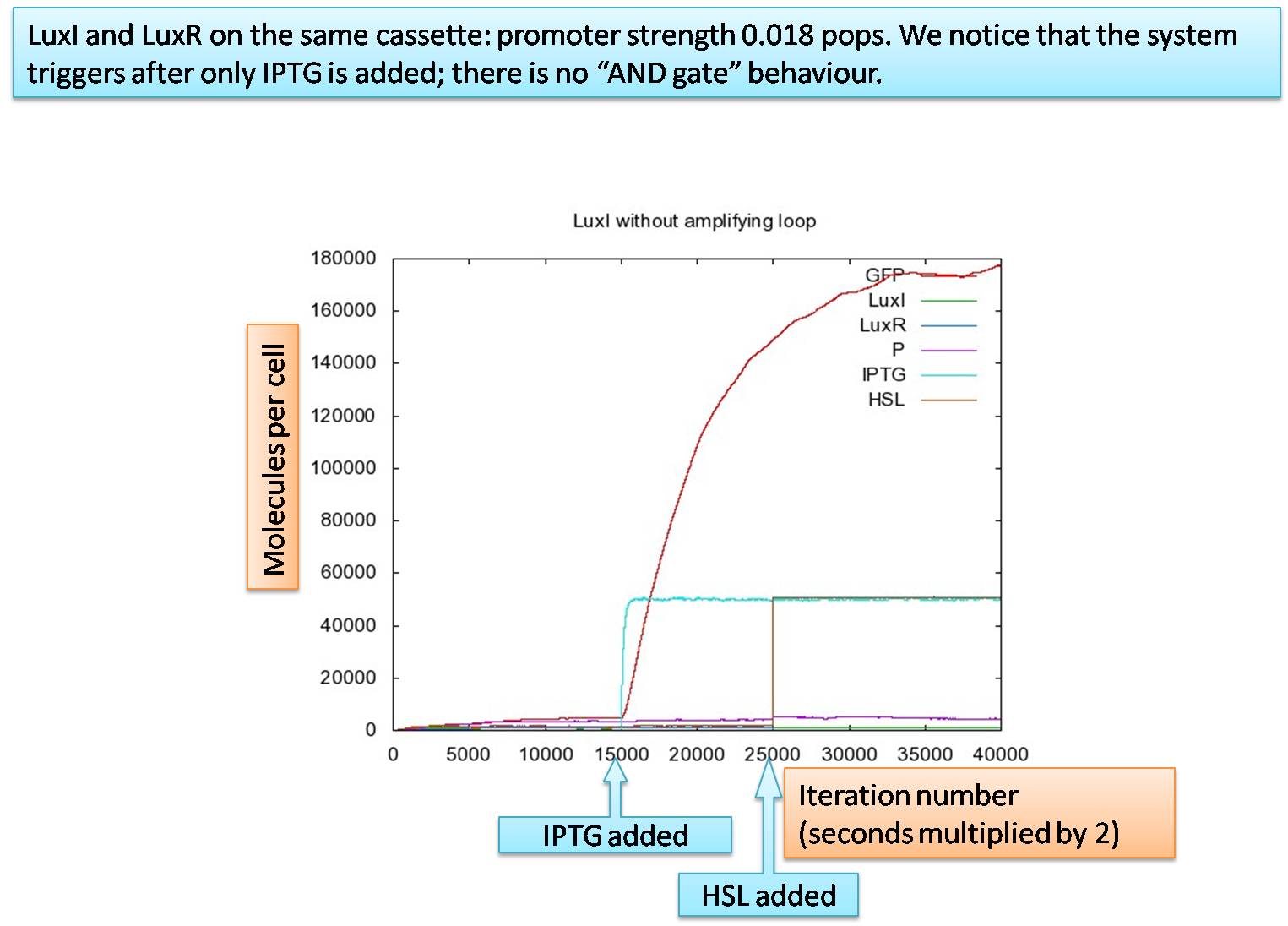
We ran our simulation using the IPTG and HSL concentration outside the cell as inputs. We changed both IPTG and HSL concentration at different times by a switch from low to high to see how the inputs affected the simulation. In our later simulations we simulated both the IPTG and HSL inputs more realistically. We did this applying the sochastic simulation (tau-leap method).
The graph shows the concentration of molecules against the iteration number (which is equivalent to time multiplied by a time-step constant of h=0.5). GFP is a place holder for the glue, P stands for the complex of HSL and LuxR, and finally, IPTG and HSL are the input signals for our cell. IPTG is released at 15000 iterations, whereas HSL diffuses from the outside at 25000 iterations. We want a system to have AND-logic gate, so that both high IPTG and HSL concentration is required for system response. However, it is clearly visible that maximal production of GFP already starts shortly after IPTG gets into the cell. We can conclude from this that the lux-box is activated from within the cell. TThe high production of LuxI and hence, HSl, leads to a high concentration of the HSl-LuxR complex. Moreover, the HSL-LuxR complex is ver stable. This complex cannot diffuse out of the cell as HSL can, so that its concentration can build up very much and hence, activate the lux-box.
Possible Solution
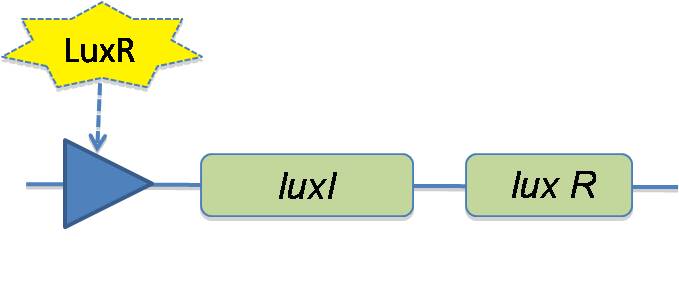
To try to overcome this problem, we have redesigned the system to have LuxI and LuxR on the same promoter but with a lux box in front of both. For this model, we no longer assume the trivial process of repression of LacI being lifted and Quorum Sensing (QS) being turned on. The LacI repression is now a repression / induction hill function with a Kd of 1200 molecules per cell for IPTG forming the LacI-IPTG complex. We assume IPTG leaks into the cell from outside. We only set the outside concentration of IPTG. For quorum sensing all simply set an elevated exterior HSL concentration which is free to diffuse in. We now include a feedback loop into the QS mechanism. To do this we attach a lux box onto the plasmid producing LuxR and LuxI, as follows:
However, the production strength of this lux box is smaller than that of the lux box for producing X, Y and cI. The strengths are as follows:
Production max for X, Y and cI = 0.44 PoPS (Polymerases-per-second)
Production min for X Y and cI = 0.013 PoPS
Production max for QS feedback = 0.002 PoPS
Production min for QS feedback = 0.00015 PoPS
(check our "parameters" section for references)
An important new parameter is KP. This is the dissociation constant for “P” activating the lux boxes, where P is the LuxR-HSL complex. In the next section we show how the most important results obtained with this model.
Level of KP required for system to function
here we use a Monte Carlo simulation to determine the value of KP in molecules per cell that is required for the system to function (note that the IPTG and HSL input levels added are both 10000 molecules)
Relationship between KTetR and KCI when KP is 50 and KLacI is 700
We notice that KP must be around 50 molecules to let the system function. We will now explore the relationship between KTetR and KCI at the value of KP=50 molecules per cell. for this simulation we set the HSL and IPTG input levels to 50,000 molecules. In our next section we will try to calculate the minimum levels of HSL and IPTG required to activate the system correctly.
The graph shows a great deal of stochastic fluctuation, which shows this system is not very robust. Increasing KP above 50 greatly increases this problem, as does decreasing HSL outside below 50,000.
Our estimation for KTetR and KCI suggest that KTetR is less than KCI. We assume the worst case value for these two dissociation constants and set them equal to each other, at KTetR = KCI = 7000 molecules per cell.
Required input levels of IPTG and HSL
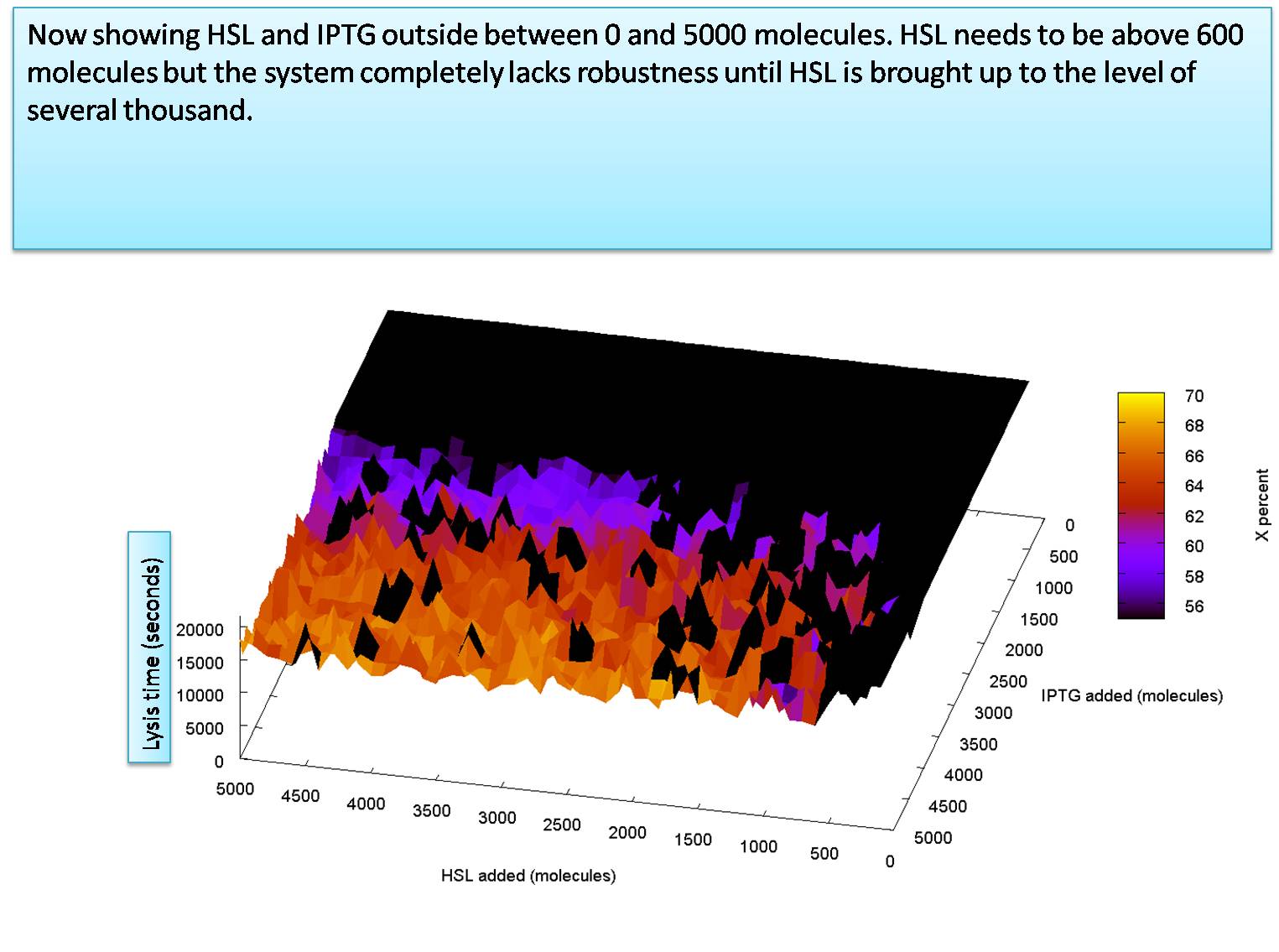
We start by going from 5,000 molecules to 100,000 molecules for HSL and from 10 molecules to 5,000 molecules for IPTG. we see that the sysem requires only around 2000 molecules of IPTG outside, although it benefits slighty when it is exposed to more. Note that the graphs color bar only extends from 55% of X production to 70% of X production. There appears to be no disernable effect in increasing the HSL outside, although when it is above 70,000 it seems to reduce the stochastic effects that stop the system lysing.
We now look at a graph showing HSL at values below 5000 molecules.
Optimal Operation
Here are the following assumptions that need to make to get the optimal system performance
KP has to be ~50 molecules per cell
KTetR has to be ~3000 molecules per cell
KCI has to be ~ 5000 molecules per cell
the input level of HSL has to be ~10,000 molecules
the input level of IPTG has to be ~5,000 molecules
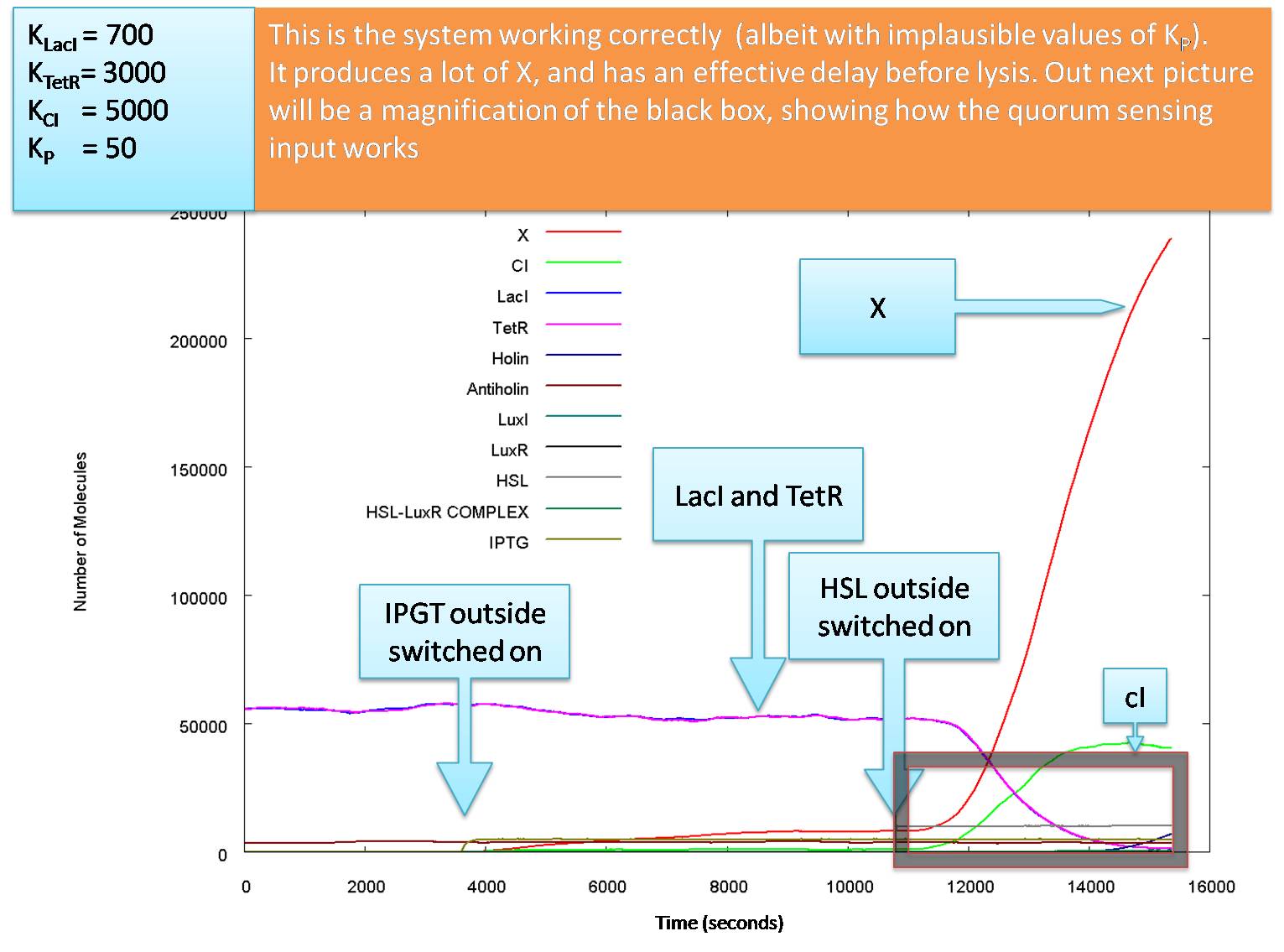
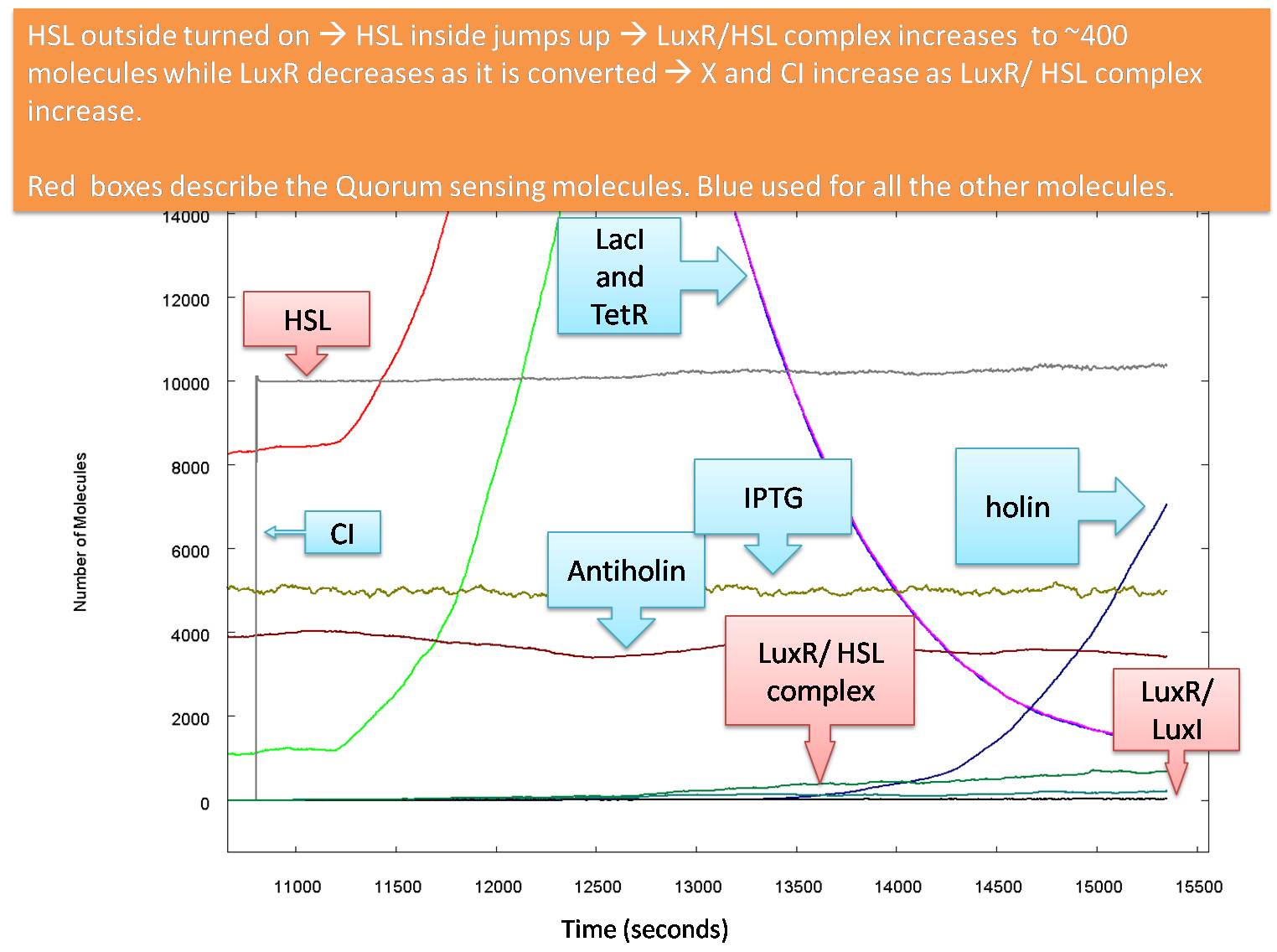
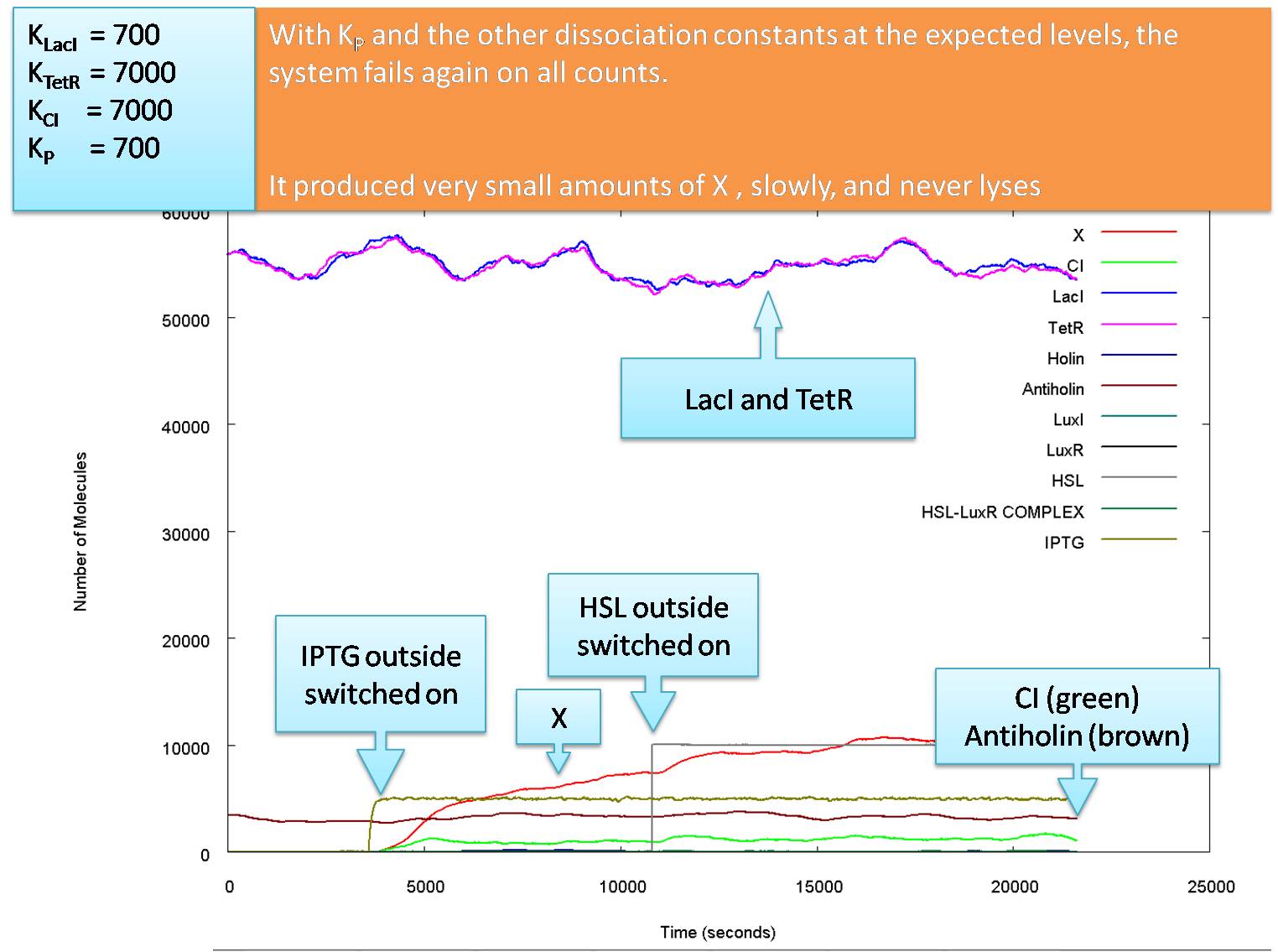
Actual Operation
The graphs above show how the system would function with our carefully calculated optimum parameter set. However, we cannot guarantee that the actual parameters will be close to these estimates. while the input levels of HSL and IPTG are reasonable, our values of KCI and KTetR may well be almost equal to each other, and around 7000 molecules per cell. While changing these to 7000 would reduce the level of the glue production at lysis the killing blow for this model is that KP is almost definitely larger than 50 molecules per cell.
The following graph shows how the system functions at the values of Kd we believe to be most likely:
Conclusions
As we have seen above, our system is very stochastically unstable, and extremly sensitive to the parameters we have investigated. As our best estimates of these parameters do not fall close to the optimal range required for the quorum sensing system we must redesign it. In the next section we amended our quorum sensing system again, to try to produce the correct response to our inputs.
 Back to Sensitivity Simulations Back to Sensitivity Simulations
|
Continue to the Amended Model 
|
Bibliography
(1) Goryachev. Systems analysis of a quorum sensing network: Design constraints imposed by the functional requirements, network topology and kinetic constants. BioSystems 2006;83(2-3 SPEC. ISS.):178.
(2) James. Luminescence control in the marine bacterium Vibrio fischeri: An analysis of the dynamics of lux regulation. J.Mol.Biol. 2000;296(4):1127.
(3) Ward JP. Mathematical modelling of quorum sensing in bacteria. IMA. 2001;18:263-292.
 "
"