Team:Calgary/Lab/Chassis Selection
From 2009.igem.org
(Difference between revisions)
(New page: {{Template:CalgaryHeader}} <html> <body> <style> p{ padding: 5px 5px 5px 5px; } .heading{ font: century gothic; color: #ffffff; } .name{ font: century gothic; color: #ffffff; padding: ...) |
|||
| (32 intermediate revisions not shown) | |||
| Line 71: | Line 71: | ||
<div class="heading">CHASSIS SELECTION</div> | <div class="heading">CHASSIS SELECTION</div> | ||
<div class="desc"> | <div class="desc"> | ||
| - | <img src="http://i1001.photobucket.com/albums/af132/igemcalgary/l.gif" align="left" hspace=10> | + | <img src="http://i1001.photobucket.com/albums/af132/igemcalgary/l.gif" align="left" hspace=10><br> |
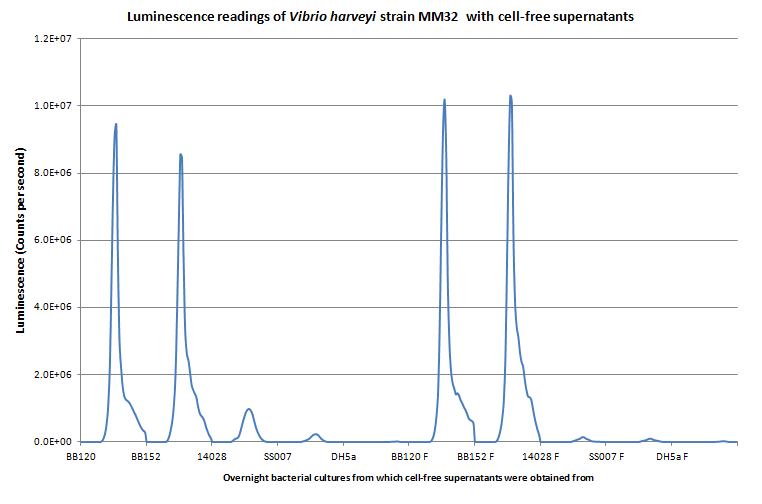
| + | We need to answer two important questions for setting up the AI-2 signalling system in <i>E. coli</i>: (1)can we isolate AI-2 and (2) do we have suitable chassis to house the system. Here we answer both questions (see figures). Vibrio harveyi MM32 (BB120 luxN::Tn5 luxS::Tn5)<sup>1</sup> is an ideal reporter strain as it cannot respond to AHL nor synthesize AI-2, respectively. In addition, since E. coli produce AI-2, we need to set our system up in cells that do not produce AI-2 (i.e. DH5α cells) and thus, we need to verify that these cells do not produce AI-2.<br><br> | ||
| + | </html> | ||
| + | |||
| + | <center>[[Image:Calgary_Chassis1.png | 700px]] | ||
| + | |||
| + | <b>Figure 1. Luminescence readings for AI-2 activity test in cell-free supernatants of various strains of bacteria with V.harveyi MM32 reporter strain taken over a 20 hour read period.</b> MM32 is responsive (i.e. luminescent) to the AI-2 present in the supernatant of V. harveyi BB120, a wildtype strain, and BB152, a strain deficient in AHL production. Salmonella typhimurium 14028 produces AI-2 but due to differences in boration, specificity to the V. harveyi reporter strain, and therefore luminescence, decreases. S. typhimurium SS007 and E. coli DH5α are incapable of producing AI-2 and serves as a negative control. 90μL of freshly diluted (1:5000) MM32 cells in autoinducer bioassay (AB) media was added to 10μL of cell free supernatant. Readings taken with VICTOR every 30 minutes; absorbance readings not shown. 3 trials in total; general trends are consistent (data not shown). | ||
| + | |||
| + | [[Image:Calgary_Chassis2.png | 700px]] | ||
| + | |||
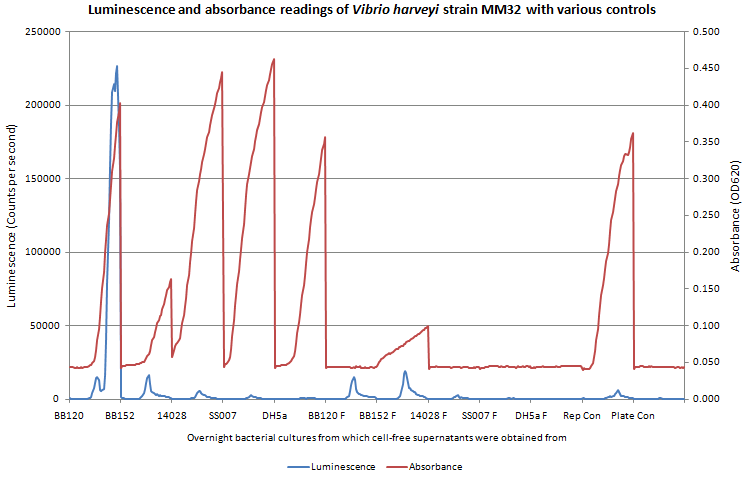
| + | <b>Figure 2. Luminescence readings for controls used in the AI-2 activity test of cell-free supernatants from various strains of bacteria with V.harveyi MM32 reporter strain taken over a 20 hour read period.</b> A comparison was made between unfiltered and filtered (indicated by “F”) CF supernatants. The presence of growth curves in the unfiltered CF supernatants stresses the importance of filtering supernatants. Presence of cells in CF supernatants explains the luminescence seen in the BB120 and BB152 experiments. The presence of luminescence and growth in BB120 F and BB152 F requires investigation into why cells were still present in the CF supernatant. Growth in the reporter control (dilute reporter cells with sterile LB media) indicates the lack of auto-luminescent cells. Lack of growth and fluorescence in the plate control (sterile LB media) shows the sterile plate. 90μL of sterile autoinducer bioassay (AB) media was added to 10μL of cell free supernatant (or LB, as indicated). Rep con stands for reporter control and Plate con stands for plate control. Readings taken with VICTOR every 30 minutes. | ||
| + | |||
| + | </center> | ||
| + | <html> | ||
</div> | </div> | ||
<br> | <br> | ||
Latest revision as of 03:59, 22 October 2009
UNIVERSITY OF CALGARY

 "
"