Team:Kyoto/GSDD/Experiment
From 2009.igem.org
m (→mechanism of MPR) |
|||
| (10 intermediate revisions not shown) | |||
| Line 21: | Line 21: | ||
#[[Team:Kyoto/GSDD|GSDD]] | #[[Team:Kyoto/GSDD|GSDD]] | ||
#Modelling | #Modelling | ||
| + | {{:Team:Kyoto/topicpath}} | ||
</div><!-- /#topicpath --> | </div><!-- /#topicpath --> | ||
{{:Team:Kyoto/Leftnavi}} | {{:Team:Kyoto/Leftnavi}} | ||
| Line 29: | Line 30: | ||
===Construction=== | ===Construction=== | ||
====PCR==== | ====PCR==== | ||
| - | + | <html><div style="border:1px #AAA solid;overflow:scroll;"><table class="table" style="width:850px;text-align:left;"> | |
| + | <tr><th>Part</th><th>Origin</th><th>Length /bp</th><th>Primer sequence</th></tr> | ||
| + | <tr> | ||
| + | <td rowspan="2">pTEF2</td><td rowspan="2">BBa_K165037</td><td rowspan="2">403</td><td style="text-align:left;">(Fwd) 5'- CGGAATTCGCGGCCGCTTCTAG -3'</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="text-align:left;">(Rev) 5'- GTTTTTTTGCCGGACTGCAGCG -3'</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td rowspan="2">kozac-LacI</td><td rowspan="2">BBa_C0012</td><td rowspan="2">1128</td><td style="text-align:left;">(Fwd) 5'- GCTCTAGAGCAAAAAAATGTCTGTGAATGTGAAACCAGTAACGTTATACGATGTC -3'</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="text-align:left;">(Rev) 5'- AGTGAGCTGATACCGCTCGC -3'</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td rowspan="2">SV40-ADH1</td><td rowspan="2">BBa_K105013</td><td rowspan="2">253</td><td style="text-align:left;">(Fwd)</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="text-align:left;">(Rev)</td> | ||
| + | </tr> | ||
| + | <tr><th>Part</th><th>Origin</th><th>Length /bp</th><th>Primer sequence</th></tr> | ||
| + | <tr> | ||
| + | <td rowspan="2">pTEF2</td><td rowspan="2">BBa_K165037</td><td rowspan="2">403</td><td style="text-align:left;">(Fwd) 5'- CGGAATTCGCGGCCGCTTCTAG -3'</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="text-align:left;">(Rev) 5'- GTTTTTTTGCCGGACTGCAGCG -3'</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td rowspan="2">kozac-eGFP</td><td rowspan="2">BBa_E0040</td><td rowspan="2">787</td><td style="text-align:left;">(Fwd) 5'- GCTCTAGAGCAAAAAAATGTCTCGTAAAGGAGAAGAACTTTTCACTGGAGTTG -3'</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="text-align:left;">(Rev) 5'- AGTGAGCTGATACCGCTCGC -3'</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td rowspan="2">PEST</td><td rowspan="2">pd2eGFP-Basic</td><td rowspan="2">123</td><td style="text-align:left;">(Fwd) 5'- CGGAATTCGCGGCCGCTTCTAGAACTGTACAAGAAGCTTAGCCATGGC -3'</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="text-align:left;">(Rev) 5'- CTGCAGCGGCCGCTACTAGTAAGAGCATCTACACATTGATCCTAGCAG -3'</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td rowspan="2">CEN-ARS</td><td rowspan="2">pYAC4</td><td rowspan="2">1585</td><td style="text-align:left;">(Fwd) 5'- TCGCGGCCGCTTCTAGAGTGGGTTGGAAGGCAAGAGAGC -3'</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="text-align:left;">(Rev) 5'- CTGCAGCGGCCGCTACTAGTATGTCCATGATTCGCCGGGTAAATAC -3'</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td rowspan="2">kozac-URA3</td><td rowspan="2">pYAC4</td><td rowspan="2">1057</td><td style="text-align:left;">(Fwd) 5'- CGGAATTCGCGGCCGCTTCTAGAGGCAGGAAACGAAGATAAATCATGTCGAAAG -3'</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="text-align:left;">(Rev) 5'- CTGCAGCGGCCGCTACTAGTATCAACGACAGGAGCACGATCATG -3'</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td rowspan="2">SV40-ADH1</td><td rowspan="2">BBa_K105013</td><td rowspan="2">266</td><td style="text-align:left;">(Fwd)</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="text-align:left;">(Rev)</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td rowspan="2">lacIBS</td><td rowspan="2">rpeats</td><td rowspan="2">1500</td><td style="text-align:left;">(Fwd)</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="text-align:left;">(Rev)</td> | ||
| + | </tr> | ||
| + | </table></div> | ||
| + | </html> | ||
====MPR==== | ====MPR==== | ||
| Line 37: | Line 102: | ||
Initially, to design a microgene, a pair of MPR primers was synthesized so that (i) the pair contained complementary bases in their 3' region; (ii) the primers had mismatched bases at their 3'-OH ends. These mismatched nucleotides at the 3'-OH ends of MPR primers are critical for successful polymerization of the microgene. During MPR, the primer pairs are reacted under conditions similar to PCR—i.e. a thermal cycle reaction is repeated in the presence of a thermostable archaeal DNA polymerase and dNTP (but without any template DNA). Without the 3'-OH end mismatch, the primer dimer that corresponds to one unit of the microgene would be amplified; including the 3'-OH end mismatch in the MPR primers and using a DNA polymerase having 3'–5' exonuclease activity enables large DNAs consisting of tandem repeats of the microgene to be synthesized. Moreover, nucleotide insertions and deletions randomly occur at end-joining junctions between microgenes, resulting in synthesis of combinatorial libraries of the repetitive sequences from a single microgene. | Initially, to design a microgene, a pair of MPR primers was synthesized so that (i) the pair contained complementary bases in their 3' region; (ii) the primers had mismatched bases at their 3'-OH ends. These mismatched nucleotides at the 3'-OH ends of MPR primers are critical for successful polymerization of the microgene. During MPR, the primer pairs are reacted under conditions similar to PCR—i.e. a thermal cycle reaction is repeated in the presence of a thermostable archaeal DNA polymerase and dNTP (but without any template DNA). Without the 3'-OH end mismatch, the primer dimer that corresponds to one unit of the microgene would be amplified; including the 3'-OH end mismatch in the MPR primers and using a DNA polymerase having 3'–5' exonuclease activity enables large DNAs consisting of tandem repeats of the microgene to be synthesized. Moreover, nucleotide insertions and deletions randomly occur at end-joining junctions between microgenes, resulting in synthesis of combinatorial libraries of the repetitive sequences from a single microgene. | ||
| - | Based on the above mechanism, we used sets of primer pairs that have 7 complementary base pairs in the 3’ region and have mismatch pairs at their 3’-OH ends. | + | Based on the above mechanism, we used sets of primer pairs that have 7 complementary base pairs in the 3’ region and have mismatch pairs at their 3’-OH ends. |
| - | + | ||
| - | + | {| class="table" style="margin:auto;" | |
| + | |- | ||
| + | ! !!Sequence!!loop Tm!!Tm!!GC | ||
| + | |- | ||
| + | |MPR primer 1 (Fwd) | ||
| + | |style="text-align:left;"|5'-GAAATGTGAGCGCTCACAACT-C<span style="color:blue;">GGCGGGG</span><span style="color:red;">A</span>-3' (31nt)||64 deg C||86 deg C||58% | ||
| + | |- | ||
| + | |MPR primer 2 (Rev) | ||
| + | |style="text-align:left;"|5'-CACAACCTAGGGTCGACTGTGGG<span style="color:blue;">CCCCGCC</span><span style="color:red;">A</span>-3'||56 deg C||72 deg C||68% | ||
| + | |} | ||
| - | [[Image:MPR( | + | [[Image:MPR(MPR).png|600px|center|Fig.1]] |
| - | + | [[Image:MPR(反応組成).png|250px|center|Fig.2]] | |
| + | =====Reaction===== | ||
# Set PCR tube into themal cycler. | # Set PCR tube into themal cycler. | ||
| - | # After it becomes | + | # After it becomes 94°C, add Vent DNA polymerase 2.6 ul (2 units/ul). (Hot start) |
[[Image:MPR(reaction).png|250px|center|Fig.1]] | [[Image:MPR(reaction).png|250px|center|Fig.1]] | ||
===Measurement=== | ===Measurement=== | ||
| + | ====transformation of yeast==== | ||
| + | 1.Yeast 10^7/ul 1ml | ||
| + | |||
| + | 2.centrifuge 5000rpm 3min | ||
| + | |||
| + | 3.remove the top of layer | ||
| + | |||
| + | 4.add MilliQ 1ml | ||
| + | |||
| + | 5.centrifuge 5000rpm 3min | ||
| + | |||
| + | 6.1MLioAc 36ul, 2mg/ml sssDNA 25ul, plusmid DNA 3ul, MilliQ 40ul | ||
| + | |||
| + | 7.vortex | ||
| + | 8.add 50%PEG#4000 240ul | ||
| + | |||
| + | 9.vortex | ||
| + | 10.RT 30min, 42℃20min | ||
| + | |||
| + | 11.centrifuge 5000rpm 1min | ||
| + | |||
| + | 12.remove the top of layer | ||
| + | |||
| + | |||
| + | |||
| + | 13.add MilliQ 50ul | ||
| + | |||
====Evaluation for the performance of Timer Vector==== | ====Evaluation for the performance of Timer Vector==== | ||
We evaluate Timer Vector by observing the expression of GFP. Timer Vector is inserted into yeast which is transformed by lacI expression vector. Timer Vector has LacI repetitive sequence in the both ends. It protects from the degradation by exonuclease, so GFP is expressed. The repetitive sequence becomes shorter every time a cell divides. At last, the repetitive sequence is completely lost. Timer Vector is degraded by exonuclease, and stop the expression of GFP. | We evaluate Timer Vector by observing the expression of GFP. Timer Vector is inserted into yeast which is transformed by lacI expression vector. Timer Vector has LacI repetitive sequence in the both ends. It protects from the degradation by exonuclease, so GFP is expressed. The repetitive sequence becomes shorter every time a cell divides. At last, the repetitive sequence is completely lost. Timer Vector is degraded by exonuclease, and stop the expression of GFP. | ||
| Line 58: | Line 159: | ||
In the anticipated result, the colony stops the expression of GFP at the almost same time. The repetitive sequence becomes shorter by about 200 bps per cell division, and it is completely lost after about 7-8 times of cell divisions. This leads the suggestion that observed colonies are composed of over 2<sup>7</sup> yeast. | In the anticipated result, the colony stops the expression of GFP at the almost same time. The repetitive sequence becomes shorter by about 200 bps per cell division, and it is completely lost after about 7-8 times of cell divisions. This leads the suggestion that observed colonies are composed of over 2<sup>7</sup> yeast. | ||
| + | [[Image:Transformed_merge.jpg|400px|thumb|center|Fig The transformed BY782 by 832, which expresses EGFP.]] | ||
</div><!-- /#rightcontents --> | </div><!-- /#rightcontents --> | ||
Latest revision as of 02:54, 22 October 2009
Experiment
Construction
PCR
| Part | Origin | Length /bp | Primer sequence |
|---|---|---|---|
| pTEF2 | BBa_K165037 | 403 | (Fwd) 5'- CGGAATTCGCGGCCGCTTCTAG -3' |
| (Rev) 5'- GTTTTTTTGCCGGACTGCAGCG -3' | |||
| kozac-LacI | BBa_C0012 | 1128 | (Fwd) 5'- GCTCTAGAGCAAAAAAATGTCTGTGAATGTGAAACCAGTAACGTTATACGATGTC -3' |
| (Rev) 5'- AGTGAGCTGATACCGCTCGC -3' | |||
| SV40-ADH1 | BBa_K105013 | 253 | (Fwd) |
| (Rev) | |||
| Part | Origin | Length /bp | Primer sequence |
| pTEF2 | BBa_K165037 | 403 | (Fwd) 5'- CGGAATTCGCGGCCGCTTCTAG -3' |
| (Rev) 5'- GTTTTTTTGCCGGACTGCAGCG -3' | |||
| kozac-eGFP | BBa_E0040 | 787 | (Fwd) 5'- GCTCTAGAGCAAAAAAATGTCTCGTAAAGGAGAAGAACTTTTCACTGGAGTTG -3' |
| (Rev) 5'- AGTGAGCTGATACCGCTCGC -3' | |||
| PEST | pd2eGFP-Basic | 123 | (Fwd) 5'- CGGAATTCGCGGCCGCTTCTAGAACTGTACAAGAAGCTTAGCCATGGC -3' |
| (Rev) 5'- CTGCAGCGGCCGCTACTAGTAAGAGCATCTACACATTGATCCTAGCAG -3' | |||
| CEN-ARS | pYAC4 | 1585 | (Fwd) 5'- TCGCGGCCGCTTCTAGAGTGGGTTGGAAGGCAAGAGAGC -3' |
| (Rev) 5'- CTGCAGCGGCCGCTACTAGTATGTCCATGATTCGCCGGGTAAATAC -3' | |||
| kozac-URA3 | pYAC4 | 1057 | (Fwd) 5'- CGGAATTCGCGGCCGCTTCTAGAGGCAGGAAACGAAGATAAATCATGTCGAAAG -3' |
| (Rev) 5'- CTGCAGCGGCCGCTACTAGTATCAACGACAGGAGCACGATCATG -3' | |||
| SV40-ADH1 | BBa_K105013 | 266 | (Fwd) |
| (Rev) | |||
| lacIBS | rpeats | 1500 | (Fwd) |
| (Rev) |
MPR
We constructed repetitive sequence by MPR(Microgene Polymerization Reaction). The sequence can be divided into some parts according to the length. They were inserted into PSB1A2 which has blunt end. In this process, we dephosphorylate PSB1A2, and phosphorylate repetetitive sequence.
mechanism of MPR
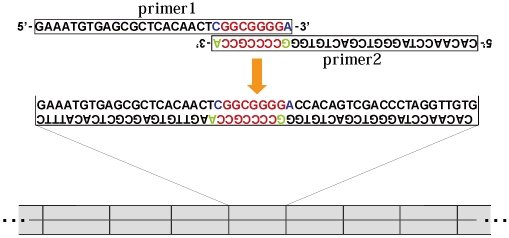
Initially, to design a microgene, a pair of MPR primers was synthesized so that (i) the pair contained complementary bases in their 3' region; (ii) the primers had mismatched bases at their 3'-OH ends. These mismatched nucleotides at the 3'-OH ends of MPR primers are critical for successful polymerization of the microgene. During MPR, the primer pairs are reacted under conditions similar to PCR—i.e. a thermal cycle reaction is repeated in the presence of a thermostable archaeal DNA polymerase and dNTP (but without any template DNA). Without the 3'-OH end mismatch, the primer dimer that corresponds to one unit of the microgene would be amplified; including the 3'-OH end mismatch in the MPR primers and using a DNA polymerase having 3'–5' exonuclease activity enables large DNAs consisting of tandem repeats of the microgene to be synthesized. Moreover, nucleotide insertions and deletions randomly occur at end-joining junctions between microgenes, resulting in synthesis of combinatorial libraries of the repetitive sequences from a single microgene. Based on the above mechanism, we used sets of primer pairs that have 7 complementary base pairs in the 3’ region and have mismatch pairs at their 3’-OH ends.
| Sequence | loop Tm | Tm | GC | |
|---|---|---|---|---|
| MPR primer 1 (Fwd) | 5'-GAAATGTGAGCGCTCACAACT-CGGCGGGGA-3' (31nt) | 64 deg C | 86 deg C | 58% |
| MPR primer 2 (Rev) | 5'-CACAACCTAGGGTCGACTGTGGGCCCCGCCA-3' | 56 deg C | 72 deg C | 68% |
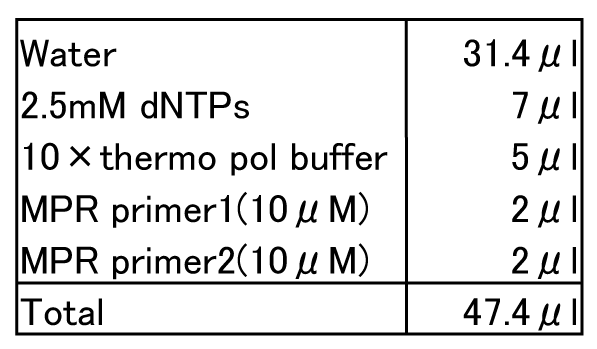
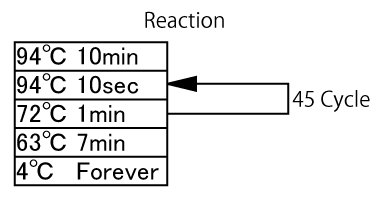
Reaction
- Set PCR tube into themal cycler.
- After it becomes 94°C, add Vent DNA polymerase 2.6 ul (2 units/ul). (Hot start)
Measurement
transformation of yeast
1.Yeast 10^7/ul 1ml
2.centrifuge 5000rpm 3min
3.remove the top of layer
4.add MilliQ 1ml
5.centrifuge 5000rpm 3min
6.1MLioAc 36ul, 2mg/ml sssDNA 25ul, plusmid DNA 3ul, MilliQ 40ul
7.vortex 8.add 50%PEG#4000 240ul
9.vortex 10.RT 30min, 42℃20min
11.centrifuge 5000rpm 1min
12.remove the top of layer
13.add MilliQ 50ul
Evaluation for the performance of Timer Vector
We evaluate Timer Vector by observing the expression of GFP. Timer Vector is inserted into yeast which is transformed by lacI expression vector. Timer Vector has LacI repetitive sequence in the both ends. It protects from the degradation by exonuclease, so GFP is expressed. The repetitive sequence becomes shorter every time a cell divides. At last, the repetitive sequence is completely lost. Timer Vector is degraded by exonuclease, and stop the expression of GFP.
The expression of GFP is observed by fluorescence microscope. We observe the yeast in liquid culture medium on a glass schale. Liquid culture allows us to choose single yeast to focus on. ConcanavalinA is added into the culture medium. This prevents the yeast from diffusing, and yeast forms colonies in culture medium. We use CSM liquid culture medium without Uracil. The yeast we use can’t synthesize Uracil. Timer Vector has Uracil synthetic gene, so only transformed yeast grow.
In the anticipated result, the colony stops the expression of GFP at the almost same time. The repetitive sequence becomes shorter by about 200 bps per cell division, and it is completely lost after about 7-8 times of cell divisions. This leads the suggestion that observed colonies are composed of over 27 yeast.
 "
"