Team:Lethbridge/Modeling
From 2009.igem.org
(→Lumazine Synthase Structural Modelling) |
(→Lumazine Synthase Structural Modelling) |
||
| (7 intermediate revisions not shown) | |||
| Line 3: | Line 3: | ||
<body style="background-color:#062770;text-align:justify"> | <body style="background-color:#062770;text-align:justify"> | ||
</html> | </html> | ||
| + | |||
<html> | <html> | ||
<head> | <head> | ||
| Line 238: | Line 239: | ||
</ul></li> | </ul></li> | ||
| - | <li><a class="qmparent" href="https://2009.igem.org/Team:Lethbridge/Ethics" style="width: | + | <li><a class="qmparent" href="https://2009.igem.org/Team:Lethbridge/Ethics" style="width:40px;">Ethics and Safety</a> |
<ul> | <ul> | ||
<li><a href="https://2009.igem.org/Team:Lethbridge/Ethics#Ethics_and_SynBio" style="width:80px;">Ethics and Synbio</a></li> | <li><a href="https://2009.igem.org/Team:Lethbridge/Ethics#Ethics_and_SynBio" style="width:80px;">Ethics and Synbio</a></li> | ||
<li><a href="https://2009.igem.org/Team:Lethbridge/Ethics#The_Survey">The Survey</a></li> | <li><a href="https://2009.igem.org/Team:Lethbridge/Ethics#The_Survey">The Survey</a></li> | ||
| - | </ul></li> | + | <li><a href="https://2009.igem.org/Team:Lethbridge/Ethics#Safety">Safety</a></li> |
| + | </ul></li> | ||
| - | <li><a class="qmparent" href="https://2009.igem.org/Team:Lethbridge/Notebook" style="width: | + | <li><a class="qmparent" href="https://2009.igem.org/Team:Lethbridge/Notebook" style="width:3px;">Notebook</a> |
<ul> | <ul> | ||
| Line 267: | Line 269: | ||
<li><a href="https://2009.igem.org/Team:Lethbridge/Team_Meetings#September">September</a></li> | <li><a href="https://2009.igem.org/Team:Lethbridge/Team_Meetings#September">September</a></li> | ||
<li><a href="https://2009.igem.org/Team:Lethbridge/Team_Meetings#October">October</a></li> | <li><a href="https://2009.igem.org/Team:Lethbridge/Team_Meetings#October">October</a></li> | ||
| - | </ul></li> | + | <li><a href="https://2009.igem.org/Team:Lethbridge/Team_Meetings#Events">Events</a></li> |
| + | </ul></li> | ||
<li><a href="https://2009.igem.org/Team:Lethbridge/Modeling" style="width:10px;">Modeling</a></li> | <li><a href="https://2009.igem.org/Team:Lethbridge/Modeling" style="width:10px;">Modeling</a></li> | ||
| Line 281: | Line 284: | ||
<li><a href="https://2009.igem.org/Team:Lethbridge/Collaboration" style="width:25px;">Collaboration</a></li> | <li><a href="https://2009.igem.org/Team:Lethbridge/Collaboration" style="width:25px;">Collaboration</a></li> | ||
<li><a href="https://2009.igem.org/Team:Lethbridge/Criteria" style="width:1px;">Judging</a></li> | <li><a href="https://2009.igem.org/Team:Lethbridge/Criteria" style="width:1px;">Judging</a></li> | ||
| - | <li><a href="https://2009.igem.org/Team:Lethbridge/videos" style="width: | + | <li><a href="https://2009.igem.org/Team:Lethbridge/videos" style="width:117px;">Extra Information on SynBio</a></li> |
<li class="qmclear"> </li></ul> | <li class="qmclear"> </li></ul> | ||
| Line 289: | Line 292: | ||
</body> | </body> | ||
</html> | </html> | ||
| - | |||
===Lumazine Synthase Structural Modelling=== | ===Lumazine Synthase Structural Modelling=== | ||
| - | + | The X-ray crystal structure of lumazine synthase (LS) from Aquifex aeolicus was solved in 2001 by Zhang et al to 1.6 Å resolution with 5 monomers in the asymmetric unit (PDB ID 1HQK). The protein forms different multimeric forms in different organisms; for example, Saccharomyces cerevisiae LS forms a homopentamer; A. aeolicus LS forms either a 60 subunit icosahedral capsid (T=1; Zhang et al, 2001) or a 180 subunit capsid (T=3; Seebeck et al, 2005). The monomeric form of LS consists of a core 4 stranded β-sheet flanked by two α-helices on one side and three on the opposite side. The electrostatic surface potential map of wild-type lumazine synthase is shown in Figure 1. | |
| + | [[Image:Modeling_fig_1.png|350px|center]] | ||
| + | By introducing glutamate residues at positions 83, 86, 120 and 123 (Seebeck et al, 2005), the interior face of the 5mer becomes much more electronegative (Figure 2). The overall formal charge for the wild-type 5mer was calculated using Pymol (Delano Scientific) as -15, or -3 per monomer. By introducing the mutations listed above, the overall formal charge for the mutant lumazine 5mer was calculated as -40 (-13.3 per monomer). As a result, the overall formal charge on the wild type 60mer is -180, compared to -480 for the mutant 60mer. | ||
| + | [[Image:Modeling_fig_2.png|350px|center]] | ||
| - | |||
| + | To model the overall structure of the lumazine synthase, the pdb file for the biological unit was downloaded from the protein databank (http://www.rcsb.org/pdb/home/home.do). A 6 subunit ring is shown below in two different orientations, demonstrating the symmetry of the overall structure in Figure 3. | ||
| + | [[Image:Modeling_fig_3.png|350px|center]] | ||
| - | + | Based on this symmetry, the intersubunit distances were determined. At any given point across the lumazine ring, the distance varied from 83.00 to 89.03 Å. As the 60 subunit lumazine capsid structurally resembles a sphere, the internal volume was calculated (V = 4/3πr3) as between 299 and 369 nm3. The introduced mutations had no effect on the intersubunit distance (figure 4) (83.02 to 89.02 Å). | |
| - | [[Image: | + | [[Image:Modeling_fig_4.png|350px|center]] |
| + | The overall structure of the capsid is shown below, with a single 5mer removed for ease of viewing the interior of the capsid: | ||
| - | [[Image: | + | [[Image:Modeling_fig_5.png|350px|center]] |
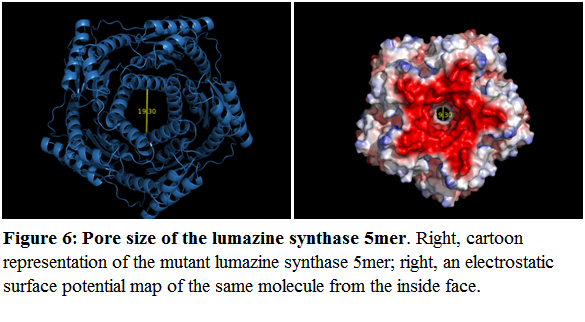
| + | However, the pore size of the lumazine 5mer to allow entry of our proposed yellow and cyan fluorescent protein is an issue. The pore size of the mutant lumazine 5mer is 19.3 angstroms in diameter at its smallest point (Figure 6): | ||
| + | |||
| + | |||
| + | [[Image:Modeling_fig_6.png|350px|center]] | ||
| - | |||
| - | |||
| - | |||
| - | As a result, the dimensions of the 236 amino acid cyan fluorescent protein (CFP) and yellow fluorescent protein (YFP) were determined and shown in | + | As a result, the dimensions of the 236 amino acid cyan fluorescent protein (CFP) and yellow fluorescent protein (YFP) were determined and shown in Figure 7. |
| - | [[Image: | + | [[Image:Modeling_fig_7.png|350px|center]] |
| - | As the proteins effectively resemble cylinders, the total volume of these proteins was determined (V = πr2h) using the longest dimensions (height = 40.28 Å; radius = 20.94 Å) as 55.49 nm3, meaning that up to 5 or 6 fluorescent proteins could fit within the 60 subunit lumazine capsid. However, none of the dimensions of the fluorescent proteins allows for targeting to a fully formed capsid; they simply could not fit through the lumazine synthase pores. Thus, one of two options is available: the proteins could be targeted to the lumazine microcompartment PRIOR to its completion, or the fluorescent proteins must be co-translationally inserted into the microcompartment prior to folding into their native, functional conformations. As it is unknown if ribosomes associate with lumazine synthase capsids, this mechanism seems unlikely; thus, targeting and insertion into the microcompartment prior to its completion seems most likely. This actually has implications for targeting other metabolic proteins, as once inside the microcompartment, they would be protected from degradation by cellular proteases, and their metabolic products could likely diffuse through the pores in the lumazine synthase capsid | + | As the proteins effectively resemble cylinders, the total volume of these proteins was determined (V = πr2h) using the longest dimensions (height = 40.28 Å; radius = 20.94 Å) as 55.49 nm3, meaning that up to 5 or 6 fluorescent proteins could fit within the 60 subunit lumazine capsid. However, none of the dimensions of the fluorescent proteins allows for targeting to a fully formed capsid; they simply could not fit through the lumazine synthase pores. Thus, one of two options is available: the proteins could be targeted to the lumazine microcompartment PRIOR to its completion, or the fluorescent proteins must be co-translationally inserted into the microcompartment prior to folding into their native, functional conformations. As it is unknown if ribosomes associate with lumazine synthase capsids, this mechanism seems unlikely; thus, targeting and insertion into the microcompartment prior to its completion seems most likely. This actually has implications for targeting other metabolic proteins, as once inside the microcompartment, they would be protected from degradation by cellular proteases, and their metabolic products could likely diffuse through the pores in the lumazine synthase capsid. |
| - | |||
All pictures generated were produced using the software Pymol (Delano Scientific). Electrostatic surface potentials and formal charges at neutral pH were calculated using the same software. Mutations in the lumazine synthase gene were introduced by hand. | All pictures generated were produced using the software Pymol (Delano Scientific). Electrostatic surface potentials and formal charges at neutral pH were calculated using the same software. Mutations in the lumazine synthase gene were introduced by hand. | ||
Latest revision as of 02:38, 22 October 2009
- Home
- Team
- Project
- Ethics and Safety
- Notebook
- Meetings
- Modeling
- Parts
- Collaboration
- Judging
- Extra Information on SynBio
Lumazine Synthase Structural Modelling
The X-ray crystal structure of lumazine synthase (LS) from Aquifex aeolicus was solved in 2001 by Zhang et al to 1.6 Å resolution with 5 monomers in the asymmetric unit (PDB ID 1HQK). The protein forms different multimeric forms in different organisms; for example, Saccharomyces cerevisiae LS forms a homopentamer; A. aeolicus LS forms either a 60 subunit icosahedral capsid (T=1; Zhang et al, 2001) or a 180 subunit capsid (T=3; Seebeck et al, 2005). The monomeric form of LS consists of a core 4 stranded β-sheet flanked by two α-helices on one side and three on the opposite side. The electrostatic surface potential map of wild-type lumazine synthase is shown in Figure 1.
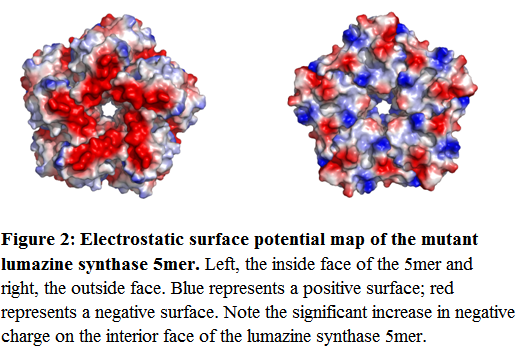
By introducing glutamate residues at positions 83, 86, 120 and 123 (Seebeck et al, 2005), the interior face of the 5mer becomes much more electronegative (Figure 2). The overall formal charge for the wild-type 5mer was calculated using Pymol (Delano Scientific) as -15, or -3 per monomer. By introducing the mutations listed above, the overall formal charge for the mutant lumazine 5mer was calculated as -40 (-13.3 per monomer). As a result, the overall formal charge on the wild type 60mer is -180, compared to -480 for the mutant 60mer.
To model the overall structure of the lumazine synthase, the pdb file for the biological unit was downloaded from the protein databank (http://www.rcsb.org/pdb/home/home.do). A 6 subunit ring is shown below in two different orientations, demonstrating the symmetry of the overall structure in Figure 3.
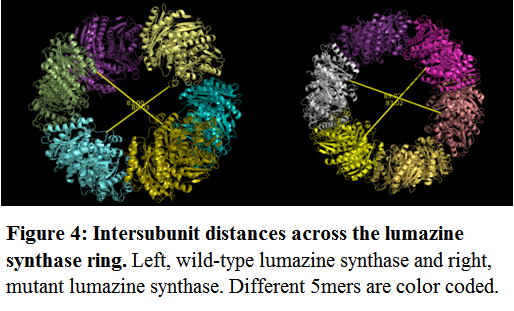
Based on this symmetry, the intersubunit distances were determined. At any given point across the lumazine ring, the distance varied from 83.00 to 89.03 Å. As the 60 subunit lumazine capsid structurally resembles a sphere, the internal volume was calculated (V = 4/3πr3) as between 299 and 369 nm3. The introduced mutations had no effect on the intersubunit distance (figure 4) (83.02 to 89.02 Å).
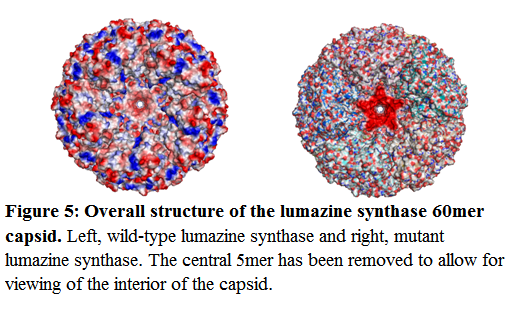
The overall structure of the capsid is shown below, with a single 5mer removed for ease of viewing the interior of the capsid:
However, the pore size of the lumazine 5mer to allow entry of our proposed yellow and cyan fluorescent protein is an issue. The pore size of the mutant lumazine 5mer is 19.3 angstroms in diameter at its smallest point (Figure 6):
As a result, the dimensions of the 236 amino acid cyan fluorescent protein (CFP) and yellow fluorescent protein (YFP) were determined and shown in Figure 7.
As the proteins effectively resemble cylinders, the total volume of these proteins was determined (V = πr2h) using the longest dimensions (height = 40.28 Å; radius = 20.94 Å) as 55.49 nm3, meaning that up to 5 or 6 fluorescent proteins could fit within the 60 subunit lumazine capsid. However, none of the dimensions of the fluorescent proteins allows for targeting to a fully formed capsid; they simply could not fit through the lumazine synthase pores. Thus, one of two options is available: the proteins could be targeted to the lumazine microcompartment PRIOR to its completion, or the fluorescent proteins must be co-translationally inserted into the microcompartment prior to folding into their native, functional conformations. As it is unknown if ribosomes associate with lumazine synthase capsids, this mechanism seems unlikely; thus, targeting and insertion into the microcompartment prior to its completion seems most likely. This actually has implications for targeting other metabolic proteins, as once inside the microcompartment, they would be protected from degradation by cellular proteases, and their metabolic products could likely diffuse through the pores in the lumazine synthase capsid.
All pictures generated were produced using the software Pymol (Delano Scientific). Electrostatic surface potentials and formal charges at neutral pH were calculated using the same software. Mutations in the lumazine synthase gene were introduced by hand.
References
Seebeck, F.P, Woycechowsky, K.J., Zhuang, W., Rabe, J.P., and Hilvert, D. A simple tagging method for protein encapsulation. J. Am. Chem. Soc. 2006. 128, 4516-4517.
L. Xing L., J. Nilsson J., X.F. Zhang, X.F., M.Fischer, M., A.Bacher, A., W. Meining, W., R. Ladenstain, R., and Cheng, R.H. The icosahedral 180 subunit capsid of the Aquifex aeolicus numazine synthase IDEA mutant dose not follow the quasi-equivalence. Microsc Microanal. 2006. 12.
Zhang, X., Meining, W., Fischer, M., Bacher, A., Ladenstein, R. X-ray structure analysis and crystallographic refinement of lumazine synthase from the hyperthermophile Aquifex aeolicus at 1.6 A resolution: determinants of thermostability revealed from structural comparisons. J. Mol. Biol. 2001. 306, 1099-1114.
 "
"