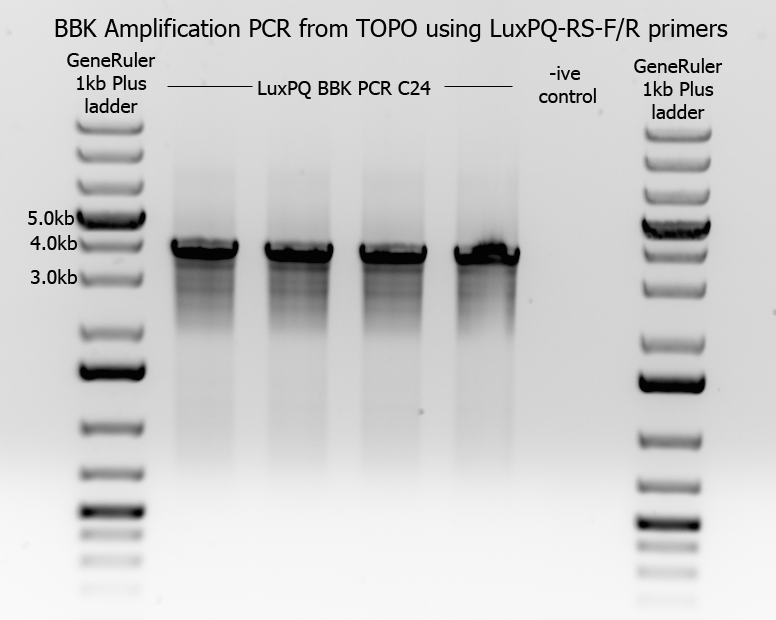Team:Calgary/29 June 2009
From 2009.igem.org
(Difference between revisions)
| (19 intermediate revisions not shown) | |||
| Line 137: | Line 137: | ||
<br> | <br> | ||
<div class="heading"> | <div class="heading"> | ||
| - | + | JUNE 29, 2009 | |
</div> | </div> | ||
<br> | <br> | ||
| Line 157: | Line 157: | ||
<br> | <br> | ||
<div class="heading"> | <div class="heading"> | ||
| - | + | Gradient PCR Verification | |
</div> | </div> | ||
<br> | <br> | ||
<div class="desc"> | <div class="desc"> | ||
</html> | </html> | ||
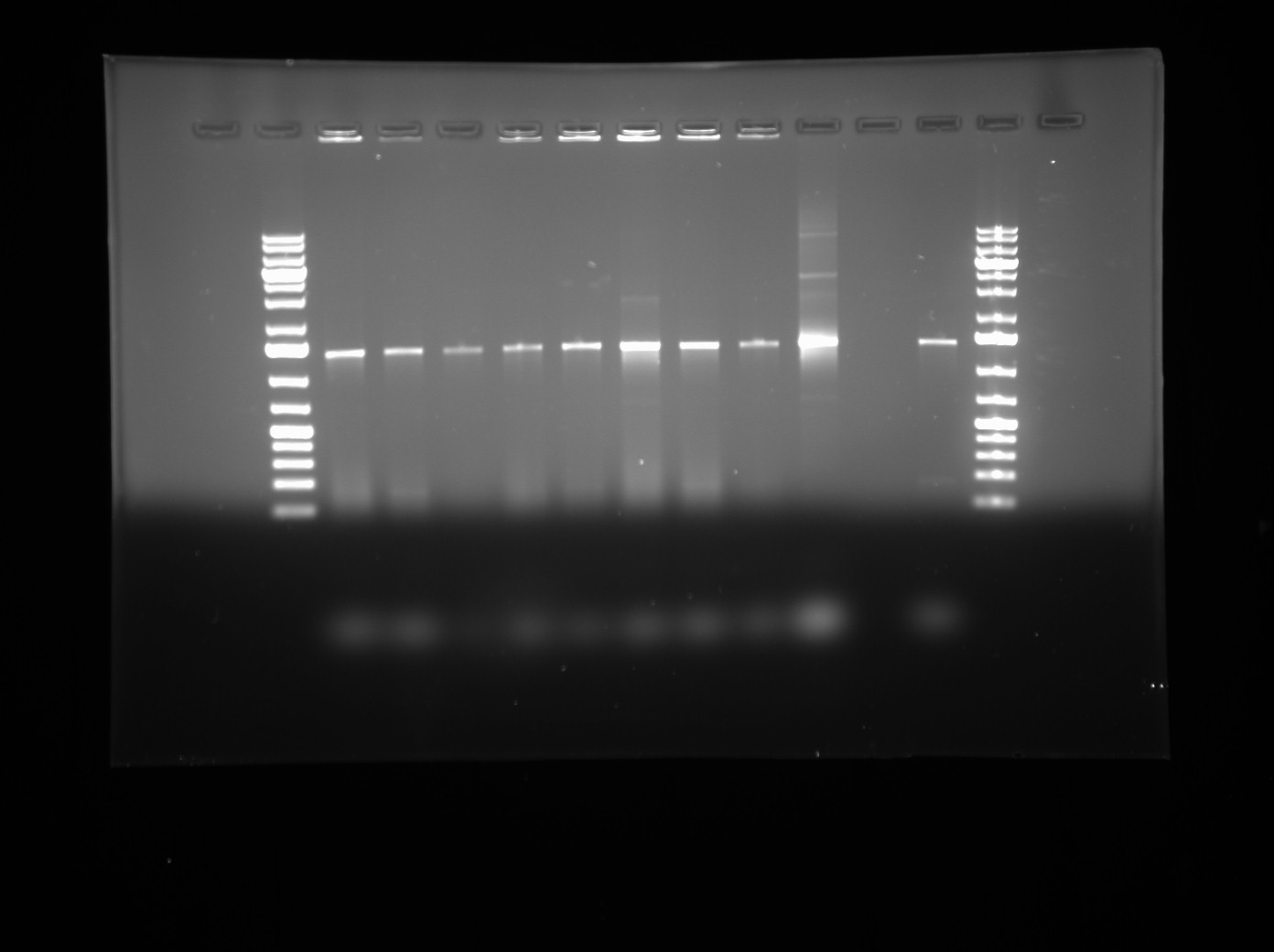
| - | + | * The gradient PCR products was verified using 0.6% agarose gel. The gel showed a 6KB band at every temperature. | |
| + | * This verifies that the biobrick sites were successfully amplified. | ||
<html> | <html> | ||
</div> | </div> | ||
</td> | </td> | ||
</tr> | </tr> | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
<tr> | <tr> | ||
| Line 209: | Line 183: | ||
<br> | <br> | ||
<div class="heading"> | <div class="heading"> | ||
| - | + | Colony PCR of BBK LuxOD47E in the psB1AC3 Vector | |
</div> | </div> | ||
<br> | <br> | ||
<div class="desc"> | <div class="desc"> | ||
</html> | </html> | ||
| - | + | *Objective: All of the plates from Friday had growth on them, so today I was able to start a colony PCR in order to verify that BBK LuxOD47E was cloned into the psB1AC3 vector. P Taq was used as well as gene specifi LuxO forward and reverse primers. Cycling conditions were the same as used for June 22nd. | |
| + | *PCR products were visualized on a 1% agarose gel run with 1.0kb+ Generuler DNA Ladder. | ||
| + | *Results: See gel photo below. Lanes 1-4 are colonies 1-4 of LuxOD47E in psB1AC3 cut with EcoRI/ PstI, lanes 5-8 are colonies 1-4 of BBK LuxOD47E in the psB1AC3 vector cut with XbaI/ PstI, lane 9 is a negative contorl and lane 10 is a positive control with C4-8 TOPO LuxOD47E DNA as the template. | ||
| + | <Br> | ||
| + | [[Image:2009.06.30ColPCRVer.jpg|350px]] | ||
| + | *Analysis: Although the DNA bands are at the expected size (~1.4 kb), the contamination in the negative contorl lane is not a good thing. We will proceed making overnight cultures and by re-doing this colony PCR. | ||
<html> | <html> | ||
| Line 235: | Line 214: | ||
<br> | <br> | ||
<div class="heading"> | <div class="heading"> | ||
| - | + | Marketing and Ethics | |
</div> | </div> | ||
<br> | <br> | ||
<div class="desc"> | <div class="desc"> | ||
</html> | </html> | ||
| - | + | Today, I made preparations for our big bake sale event tomorrow. I also sent out the potential and current sponsors our June Newsletter along with our CTV news link. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
<br> | <br> | ||
| - | |||
| - | |||
| - | |||
<br> | <br> | ||
| - | + | I also read some ethics papers relating to Biosecurity and Bio-safety | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
<html> | <html> | ||
| Line 287: | Line 243: | ||
<br> | <br> | ||
<div class="heading"> | <div class="heading"> | ||
| - | + | Agarose gel of NotI digest | |
</div> | </div> | ||
<br> | <br> | ||
<div class="desc"> | <div class="desc"> | ||
</html> | </html> | ||
| - | + | <center>[[Image:Calgary_luxOU_luxOUfinal_NotI.jpg | 500px]]</center> | |
| + | |||
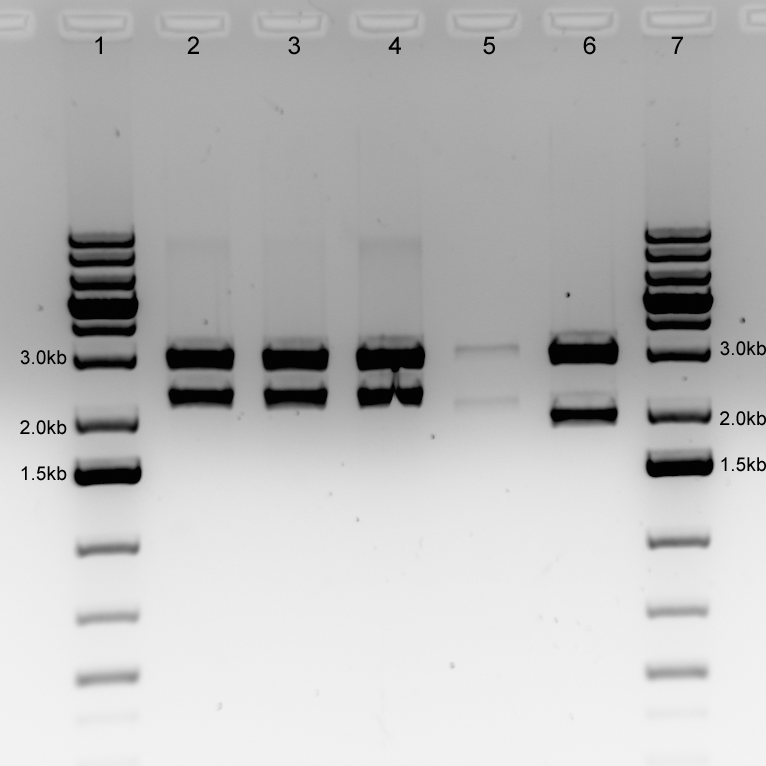
| + | Figure. 1% agarose gel (100V) for NotI verification digest of construction yielding BBa_B0015-BBa_R0040-luxOU-BBa_B0015 in psB1AC3. 200ng of plasmid was cut with NotI for 2 hours at 37oC (lanes 2-4). Digested luxOU-BBa_B0015 in psB1AC3 (lane 5) and luxOU in psB1AC3 (lane 6) were used as positive controls. Expected band sizes for the BBa_B0015-BBa_R0040-luxOU-BBa_B0015 construction in psB1AC3 are 3055bp (psB1AC3) and 2257bp. Expected band sizes for the luxOU-BBa_B0015 construction are 3055bp (psB1AC3) and 2128bp; expected band sizes for luxOU are 3055bp (psB1AC3) and 1945bp. 5μL of GeneRuler 1kb Plus DNA Ladder was loaded. | ||
| + | |||
| + | Size differences are evident in the different constructions. C1 and C7 were sent for sequencing. | ||
<html> | <html> | ||
| Line 313: | Line 273: | ||
<br> | <br> | ||
<div class="heading"> | <div class="heading"> | ||
| - | + | BBK Amplification of LuxPQ from TOPO vector | |
</div> | </div> | ||
<br> | <br> | ||
<div class="desc"> | <div class="desc"> | ||
</html> | </html> | ||
| - | + | Purpose: to amplify LuxPQ with BBK restriction sites from LuxPQ in TOPO-BL-Colony24-Mutagen C1. This was done with platinum PFX and LuxPQ-RS-F/R primers. The conditions were as follows: 94ºC for 3 minutes; 36X (94ºC for 30 seconds; 65ºC for 45 seconds; 68ºC for 4 minutes); 68ºC for 10 minutes; held at 4ºC. The products were then run on a 0.8% agarose gel (see below). Based on this gel, we have the expected size of PCR product at just under 4kb, with a clean negative control. The next step will be to purify the PCR product and then construct into a BBK vector. | |
| - | + | [[Image:2009.06.30.luxPQ_BBK_Amp.png|700px]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
<html> | <html> | ||
| Line 370: | Line 306: | ||
<div class="desc"> | <div class="desc"> | ||
</html> | </html> | ||
| - | Mandy and I were assigned as partners and we were able to find a possible application of our signalling circuit involving the antifreeze proteins. There are variety of organisms that express Antifreeze proteins (AFPs), including fish (Arctic cod), bacteria (Pseudomonas putida), insects, and plants. Since we only work with bacteria, we attempted to find the most suitable antifreeze protein, and the antifreeze protein in <i>Marinomonas primoryensis</i> seemed to be most ideal. | + | Mandy and I were assigned as partners and we were able to find a possible application of our signalling circuit involving the antifreeze proteins. There are variety of organisms that express Antifreeze proteins (AFPs), including fish (Arctic cod), bacteria (<i>Pseudomonas putida</i>), insects, and plants. Since we only work with bacteria, we attempted to find the most suitable antifreeze protein, and the antifreeze protein in <i>Marinomonas primoryensis</i> seemed to be most ideal. |
<br>Unlike other plant and bacterial antifreeze proteins, it is found to be hyperactive, meaning it is stable in low temperatures. Also, this antifreeze protein, like the one found in animals, utilizes a freeze-avoidant strategy which depresses the freezing point of the cell, while antifreeze proteins from other plants and bacteria utilize freeze-tolerant method, which does let the cell freeze, but it inhibits the growth of ice crystals; thus preventing the cell from damage. | <br>Unlike other plant and bacterial antifreeze proteins, it is found to be hyperactive, meaning it is stable in low temperatures. Also, this antifreeze protein, like the one found in animals, utilizes a freeze-avoidant strategy which depresses the freezing point of the cell, while antifreeze proteins from other plants and bacteria utilize freeze-tolerant method, which does let the cell freeze, but it inhibits the growth of ice crystals; thus preventing the cell from damage. | ||
| + | |||
<html> | <html> | ||
</div> | </div> | ||
| Line 391: | Line 328: | ||
<br> | <br> | ||
<div class="heading"> | <div class="heading"> | ||
| - | + | Restriction Digest in Second Life | |
</div> | </div> | ||
<br> | <br> | ||
<div class="desc"> | <div class="desc"> | ||
</html> | </html> | ||
| - | + | More objects (restriction enzymes) for restriction digest were made. I have scripted the phosphatase treatment and built a heating block. The heating block and water bath still need to be scripted. Notecards are still required for restriction. Other than this, I made a video for the blog showcasing what we've started to do in the virtual labs. We also met with Sonja today to discuss the Sim's layout, and how to make the island more educational (e.g. levels in biobricker, buddies that help you out). | |
| + | <br> | ||
| + | Made my video for the blog :) | ||
<html> | <html> | ||
| Line 417: | Line 356: | ||
<br> | <br> | ||
<div class="heading"> | <div class="heading"> | ||
| - | + | New Version of the HUD | |
</div> | </div> | ||
<br> | <br> | ||
<div class="desc"> | <div class="desc"> | ||
</html> | </html> | ||
| - | + | Retrospective Notebook: This entry was not written on this day, but derived later from working notes I made that day. | |
| + | |||
| + | I figured out Second Life's very backwards link numbering system today. When prims (primitive objects) are linked together into a larger object, each one is assigned a 'link number' within the larger object. Then, scripts running in the prims can send messages to scripts in other prims via a 'link message' system, using the link number as an address (e.g. Prim number two says 'hello!' to prim number five. Prim five responds with 'get off my lawn' to prim number two.) The problem is that if one wants to use these link numbers to send messages, you have to know what they are, and link numbers are assigned invisibly and automatically by the SL game engine when the prims are linked together. | ||
| + | |||
| + | I did some experimentation, and as it turns out, link numbers are assigned in the reverse order that you selected the prims before linking them. So to assign the link numbers predictable, we hold down shift (as in windows explorer) and select the object we want to have the highest link number, and select the next highest, and then the next highest, etc. | ||
| + | |||
| + | This is just what I'd needed to simplify the heads up display. Instead of fiddling with the display tiles names each time I wanted to roll out an updated script, I could create a single tile with the new script. Then, copy that tile four times to get five tiles in a row, copy those five to get a row of fifteen tiles, and then copy that row eight times to get the full 8*15 block. And then, select them from last to first, one at a time, selecting the HUD's background last (where the HUD's main script lives), link them together, and voila. Updated HUD. Thankfully, the selection system is forgiving, so that when I inevitably misclick the 100th tile when I wanted the 101st, I can shift click it again to unselect, and continue without restarting. | ||
| + | |||
| + | Using link numbers also had a secondary benefit: the HUD's main script no longer needed to send each 'display character x at tile y' message to every tile, and let the tiles determine whether it was intended for them or not. Each of the 120 tiles would end up rejecting 119 messages! Instead, I could now do a little math to calculate which tile needed to get which message. The background object got link number '1', so the first tile of the first row was number 2, the first of the second row was number 17, through to 121, the last tile in the bottom row. The higher efficiency made the HUD's screen refresh speed go up by an order of magnitude, giving you the speedy screen you're familiar with today. | ||
| + | |||
| + | And if my decision to change systems needed any more justification, I discovered two more typos in the names of the tiles from the last version while I was building the new version. Out with the old, in with the new! | ||
<html> | <html> | ||
| Line 443: | Line 392: | ||
<br> | <br> | ||
<div class="heading"> | <div class="heading"> | ||
| - | + | Second Bake sale | |
</div> | </div> | ||
<br> | <br> | ||
<div class="desc"> | <div class="desc"> | ||
</html> | </html> | ||
| - | + | This was iGEM Calgary's Second bake sale. The team divided up the task of baking goodies. We met up at Emily's house yesterday to bake cupcakes, muffins, cakes and much more. We also had cinnamon buns, cheesecakes, samosas, cookies and the list goes on. I booked the Hippocrates site the week before. I had to be at the sale all day, heating up food, handing out the goods and keeping track of the float. I also helped to clean up at the end of the event. we successfully raised $370.00. | |
<html> | <html> | ||
| Line 462: | Line 411: | ||
<tr> | <tr> | ||
<td> | <td> | ||
| - | <a name=" | + | <a name="Vicki"></a> |
<br> | <br> | ||
<div class="heading"> | <div class="heading"> | ||
| - | + | VICKI | |
</div> | </div> | ||
<br> | <br> | ||
<div class="heading"> | <div class="heading"> | ||
| - | + | Plasmid isolation of overnight cultures and subsequent NotI restriction digest | |
</div> | </div> | ||
<br> | <br> | ||
| Line 475: | Line 424: | ||
</html> | </html> | ||
WIKI CODING HERE | WIKI CODING HERE | ||
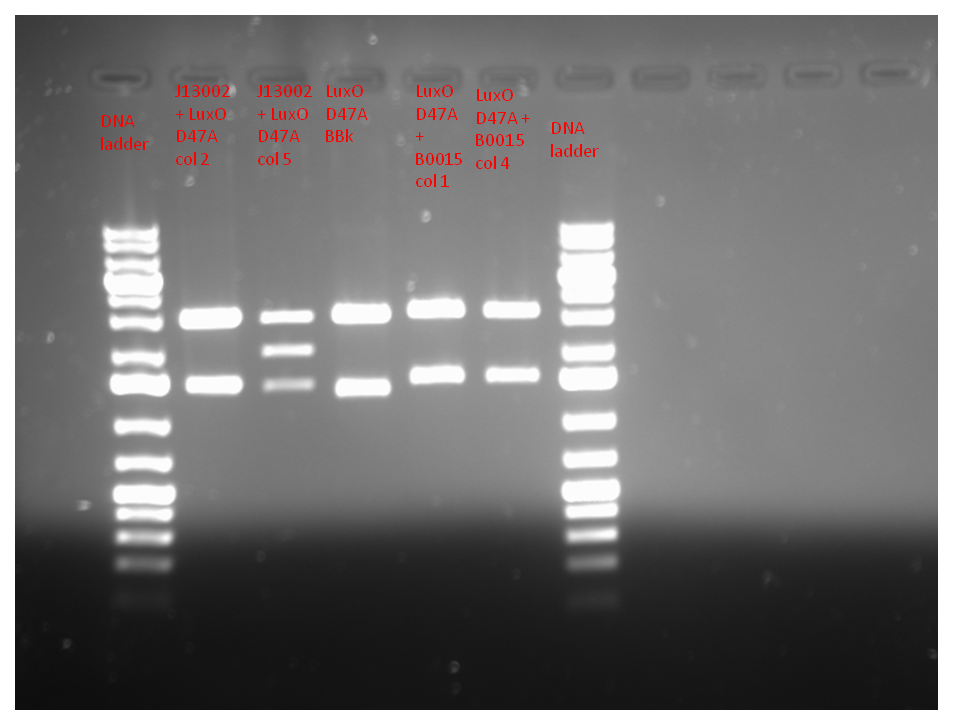
| + | Purpose: to confirm if a construct of the appropriate size can be excised from the plasmids transformed into TOP10 competent cells | ||
| - | + | Materials and methods | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | The plasmid isolation and restriction digest were performed in accordance with the procedure outlined on the protocol page. We expect to see 2 pieces from the J13002 + LuxOD47A BBk, one at 1685 bp and one at 3055 bp (for the AC plasmid); 2 pieces from the LuxOD47A BBk + B0015, one at 1750 bp and one at 3189 bp (for the AK plasmid); and 2 pieces from the LuxOD47A BBk size control, one at 1610 bp and one at 3055 bp (for the AC plasmid). | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | Results | |
| - | + | ||
| - | + | Attached below. Lane 1 is the DNA ladder; lanes 2 and 3 are J13002 + LuxOD47A BBk (colonies 2 and 5 respectively); lane 4 is LuxOD47A BBk size control; lanes 5 and 6 are LuxOD47A BBk + B0015 (colonies 1 and 4 respectively); and lane 7 is a DNA ladder. The DNA ladder used was the GeneElute 1kb+. The bands for the mutant and mutant + B0015 are in line with what we expected. | |
| - | <br> | + | |
| - | + | <br><br> | |
| - | + | <center> | |
| - | + | [[Image:June29.png|700px]] | |
| - | <br> | + | </center> |
| - | < | + | |
| - | + | ||
| - | </ | + | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
<html> | <html> | ||
Latest revision as of 06:57, 20 October 2009
UNIVERSITY OF CALGARY

 "
"