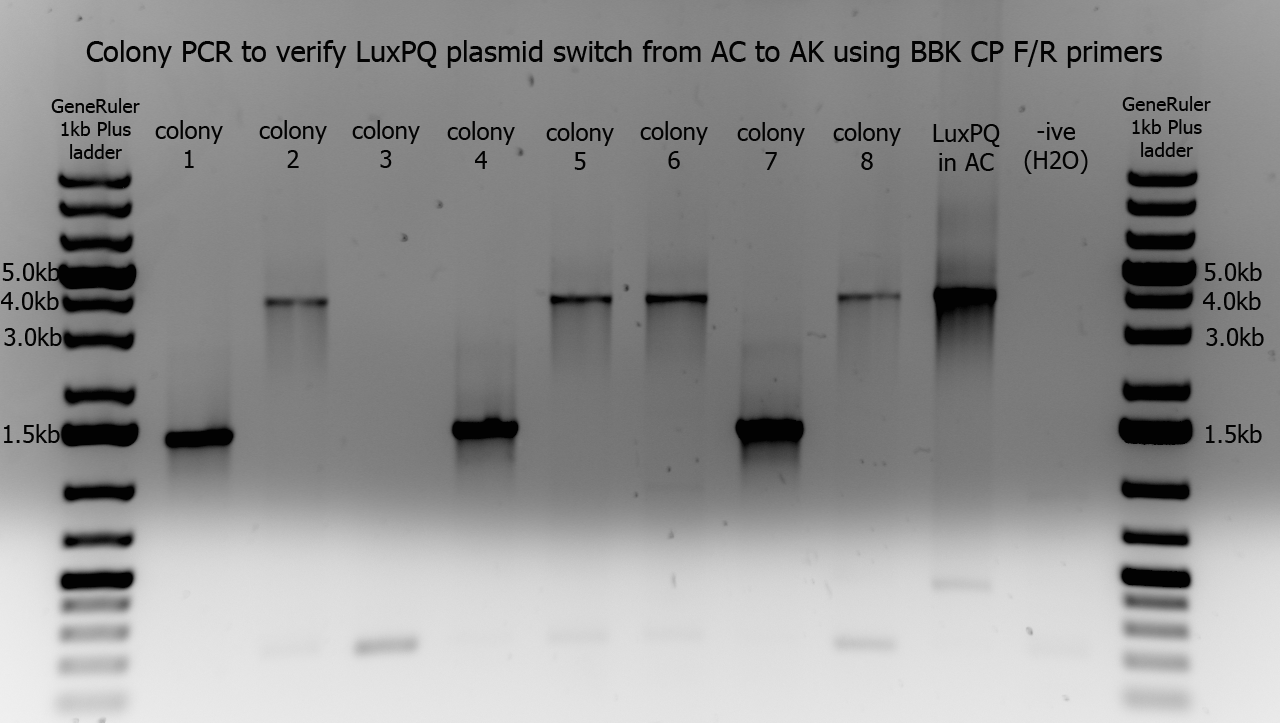|
|
| (3 intermediate revisions not shown) |
| Line 163: |
Line 163: |
| | </html> | | </html> |
| | * While clearing out some old unsuccessful plates, we found three other colonies, performed colony PCR and the 0.6% agarose gel showed no viable results. | | * While clearing out some old unsuccessful plates, we found three other colonies, performed colony PCR and the 0.6% agarose gel showed no viable results. |
| | + | |
| | <html> | | <html> |
| | </div> | | </div> |
| Line 187: |
Line 188: |
| | <div class="desc"> | | <div class="desc"> |
| | </html> | | </html> |
| - | The Matlab Help files were helpful in providing basic background about Markov Models and their uses . I also tried to recall my MATH211 notes where we had briefly studied Markov Models. | + | The Matlab Help files were helpful in providing basic background about Markov Models and their uses. I also tried to recall my MATH211 notes where we had briefly studied Markov Models. |
| | | | |
| | <html> | | <html> |
| Line 214: |
Line 215: |
| | <div class="desc"> | | <div class="desc"> |
| | </html> | | </html> |
| - | *Performed a colony PCR of J13002-LuxOD47E-B0015 to see if the B0015 terminator was seuccessfully cloned in, using BBK-CP forward and reverse primers and p Taq. Cycling conditions were the same as used for the last Colony PCR (see July 9th 2009 for details). PCR products were visualized on a 1% agarose gel and run with Generuler 1 kb+ DNA ladder along with J13002-LuxOD47E and LuxOD47E as size controls. See photo of gel below. | + | *Performed a colony PCR of J13002-LuxOD47E-B0015 to see if the B0015 terminator was successfully cloned in, using BBK-CP forward and reverse primers and p Taq. Cycling conditions were the same as used for the last Colony PCR (see July 9th 2009 for details). PCR products were visualized on a 1% agarose gel and run with Generuler 1 kb+ DNA ladder along with J13002-LuxOD47E and LuxOD47E as size controls. See photo of gel below. |
| | + | <Br> |
| | + | [[Image:2009.07.10J13002-LuxOD47E-B0015.jpg|350px]] |
| | | | |
| | *Analysis: Lanes 1-4 are J13002-LuxOD47E-B0015 trial 1 (treating B0015 as the Insert). Lanes 5-8 are trial 2 (treating J13002-LuxOD47E as the Insert). Lane 9 is J13002-LuxOD47E (a size control), Lane 10 is LuxOD47E BBK (another size control) and Lane 11 is a negative control. | | *Analysis: Lanes 1-4 are J13002-LuxOD47E-B0015 trial 1 (treating B0015 as the Insert). Lanes 5-8 are trial 2 (treating J13002-LuxOD47E as the Insert). Lane 9 is J13002-LuxOD47E (a size control), Lane 10 is LuxOD47E BBK (another size control) and Lane 11 is a negative control. |
| | | | |
| - | *This gel seems ot be kind of slanted, which isn't very good for size comparison. It looks like some of the first lanes of Trial 1 may have worked (contain the B0015 terminator) as they appear slighlty higher than other lanes, however we will run a new gel with the PCR product to be sure. | + | *This gel seems ot be kind of slanted, which isn't very good for size comparison. It looks like some of the first lanes of Trial 1 may have worked (contain the B0015 terminator) as they appear slightly higher than other lanes, however we will run a new gel with the PCR product to be sure. |
| - | | + | |
| - | | + | |
| | | | |
| | <html> | | <html> |
| Line 262: |
Line 263: |
| | <br> | | <br> |
| | I also worked on my US visa application and attended our weekly meetings with my supervisors. | | I also worked on my US visa application and attended our weekly meetings with my supervisors. |
| | + | <br> |
| | <html> | | <html> |
| | </div> | | </div> |
| Line 281: |
Line 283: |
| | <br> | | <br> |
| | <div class="heading"> | | <div class="heading"> |
| - | Descriptive Title of What You're Doing
| + | One more time concentration Graphs: Shifting |
| | </div> | | </div> |
| | <br> | | <br> |
| | <div class="desc"> | | <div class="desc"> |
| | </html> | | </html> |
| - | WIKI CODING HERE
| + | Mathematica graph function (a built-in function) gets a list of numbers and plot them in a way that we have the list of numbers on Y-axis and the indexes of the numbers in the list on X-axis. |
| | + | Knowing that, when we just have one cell in the simulation, and we want to plot the concentration graph of a given agent in the cell (for example AI-2), we pass the number of AI-2s in teach time step as a list to our built-in graph function and we generate it. However, when division occurs we have to pass two lists of numbers (one for first cell, and the other for the newly produced cell). That is where we are faced with a problem. |
| | + | The problem is as it was mentioned Mathematica automatically plot the graph by the indexes the numbers in the list, and the indexes for the cells existing from beginning of the simulation is as same as each time step, but for newly generated cells we need to shift the graphs to the time step they become to exist. |
| | + | To do so, we need to save the time steps that divisions occur, and then somehow differentiate the lists of numbers we are passing and add the time steps to the lists that are for new cells. |
| | + | I have written 4 functions that they all together shift the concentration graphs, so that they work perfectly with division function. |
| | | | |
| - | <html>
| + | Here is a concentration graph for AI-2 concentration: |
| - | </div>
| + | |
| - | </td>
| + | |
| - | </tr>
| + | |
| | | | |
| - | <tr>
| + | [[Image:Graph11.png]] |
| - | <td height="10px" bgcolor="#222222">
| + | |
| - | </td>
| + | |
| - | </tr>
| + | |
| | | | |
| - | <tr>
| + | AI-2 Concentration Graph. On X-axis, time steps are aligned, and on Y-axis number of AI-2 molecules are placed. This graph shows that the simulation is run for 3000 time steps and started with one cell (blue line) and other cells are generated over time by divisions (red, green, and yellow lines). This type of results provides numerical information about the concentrations of agents and also demonstrates the general trend of change of concentrations in the bacteria. demonstrates that AI-2 is produced and accumulated in the bacteria between each division, however the concentrations of accumulated AI-2 change logarithmic which reveals that some of the AI-2 are transported from inside to the outside of the bacteria. AI-2 molecules then get divided within divisions between new cells, which is depicted by those straight lines in the graph. |
| - | <td>
| + | |
| - | <a name="Jamie"></a>
| + | |
| - | <br>
| + | |
| - | <div class="heading">
| + | |
| - | JAMIE
| + | |
| - | </div>
| + | |
| - | <br>
| + | |
| - | <div class="heading">
| + | |
| - | Descriptive Title of What You're Doing
| + | |
| - | </div>
| + | |
| - | <br>
| + | |
| - | <div class="desc">
| + | |
| - | </html>
| + | |
| - | WIKI CODING HERE
| + | |
| | | | |
| | <html> | | <html> |
| Line 345: |
Line 331: |
| | <br> | | <br> |
| | [[Image:2009.07.11.PQ_OU_construct_cPCR.png|700px]] | | [[Image:2009.07.11.PQ_OU_construct_cPCR.png|700px]] |
| | + | |
| | <html> | | <html> |
| | </div> | | </div> |
| Line 350: |
Line 337: |
| | </tr> | | </tr> |
| | | | |
| - | <tr>
| |
| - | <td height="10px" bgcolor="#222222">
| |
| - | </td>
| |
| - | </tr>
| |
| - |
| |
| - | <tr>
| |
| - | <td>
| |
| - | <a name="Katie"></a>
| |
| - | <br>
| |
| - | <div class="heading">
| |
| - | KATIE
| |
| - | </div>
| |
| - | <br>
| |
| - | <div class="heading">
| |
| - | Descriptive Title of What You're Doing
| |
| - | </div>
| |
| - | <br>
| |
| - | <div class="desc">
| |
| - | </html>
| |
| - | WIKI CODING HERE
| |
| - |
| |
| - | <html>
| |
| - | </div>
| |
| - | </td>
| |
| - | </tr>
| |
| | | | |
| | <tr> | | <tr> |
| Line 396: |
Line 358: |
| | </html> | | </html> |
| | Each sectional members are now put on the spot for weekly updates, so we get more in-depth look at each accomplishments. | | Each sectional members are now put on the spot for weekly updates, so we get more in-depth look at each accomplishments. |
| | + | |
| | <html> | | <html> |
| | </div> | | </div> |
| Line 424: |
Line 387: |
| | <br> | | <br> |
| | The robots now respond to more sentences, so hopefully they are more user-friendly. Still need to figure out how to make it most obvious what must be said to the robot for them to work. We also had our Second Life meeting today. | | The robots now respond to more sentences, so hopefully they are more user-friendly. Still need to figure out how to make it most obvious what must be said to the robot for them to work. We also had our Second Life meeting today. |
| - |
| |
| | | | |
| | <html> | | <html> |
| Line 480: |
Line 442: |
| | *-biobrick tower (aka Biobrick Simulator) | | *-biobrick tower (aka Biobrick Simulator) |
| | *-lab (aka Virtual Lab) | | *-lab (aka Virtual Lab) |
| - |
| |
| | | | |
| | <html> | | <html> |
| Line 558: |
Line 519: |
| | </html> | | </html> |
| | | | |
| - | Yesterday, time was spent on building a robot (Biobert) that will act | + | Yesterday, time was spent on building a robot (Biobert) that will act as a guide to our lab and also the rest of the island. It had to be built from scratch because the first one wasn't very good and creeped people out because it transformed from a pipette and had an empty souless expression. Once it was built, Mandy added the script so it could talk to people (including several secret phrases!). I designed |
| - | as a guide to our lab and also the rest of the island. It had to be | + | it to move its arms and mouth when touched and optimized to use the least prims possible in order to make sure we have enough left for other things on the island. Today, a second robot was also made in the same fashion (Synthia) because Biobert was lonely and he needed a friend. We also had an important meeting deciding on the structure of the Second Life areas. |
| - | built from scratch because the first one wasn't very good and creeped | + | |
| - | people out because it transformed from a pipette and had an empty | + | |
| - | soulless expression. Once it was built, Mandy added the script so it
| + | |
| - | could talk to people (including several secret phrases!). I designed | + | |
| - | it to move its arms and mouth when touched and optimized to use the | + | |
| - | least prims possible in order to make sure we have enough left for | + | |
| - | other things on the island. Today, a second robot was also made in the | + | |
| - | same fashion (Synthia) because Biobert was lonely and she needed a | + | |
| - | friend. We also had an important meeting deciding on the structure of the Second Life areas. | + | |
| | <br><br> | | <br><br> |
| | <center> | | <center> |
| Line 574: |
Line 526: |
| | </center> | | </center> |
| | | | |
| - |
| |
| | <html> | | <html> |
| | </div> | | </div> |
| Line 600: |
Line 551: |
| | </html> | | </html> |
| | …was also done exactly as I described in my July 8 update. One of my tubes struggled through the lyse stage but the rest seemed to work better. The plasmid yield was not as lucrative as last time – around 70 ng/uL (100 uL sample), but it’s an improvement on the 7 ng/uL present the first time I conducted this procedure with these cells. | | …was also done exactly as I described in my July 8 update. One of my tubes struggled through the lyse stage but the rest seemed to work better. The plasmid yield was not as lucrative as last time – around 70 ng/uL (100 uL sample), but it’s an improvement on the 7 ng/uL present the first time I conducted this procedure with these cells. |
| - | <html>
| |
| | | | |
| | + | <html> |
| | </div> | | </div> |
| | </td> | | </td> |
|
|
CAROL
Colony PCR
- While clearing out some old unsuccessful plates, we found three other colonies, performed colony PCR and the 0.6% agarose gel showed no viable results.
|
|
|
CHINMOYEE
Looked at Markov Models ( Part of Optimization)
The Matlab Help files were helpful in providing basic background about Markov Models and their uses. I also tried to recall my MATH211 notes where we had briefly studied Markov Models.
|
|
|
EMILY
Colony PCR for Verification of J13002-LuxOD47E-B0015 Construction
- Performed a colony PCR of J13002-LuxOD47E-B0015 to see if the B0015 terminator was successfully cloned in, using BBK-CP forward and reverse primers and p Taq. Cycling conditions were the same as used for the last Colony PCR (see July 9th 2009 for details). PCR products were visualized on a 1% agarose gel and run with Generuler 1 kb+ DNA ladder along with J13002-LuxOD47E and LuxOD47E as size controls. See photo of gel below.
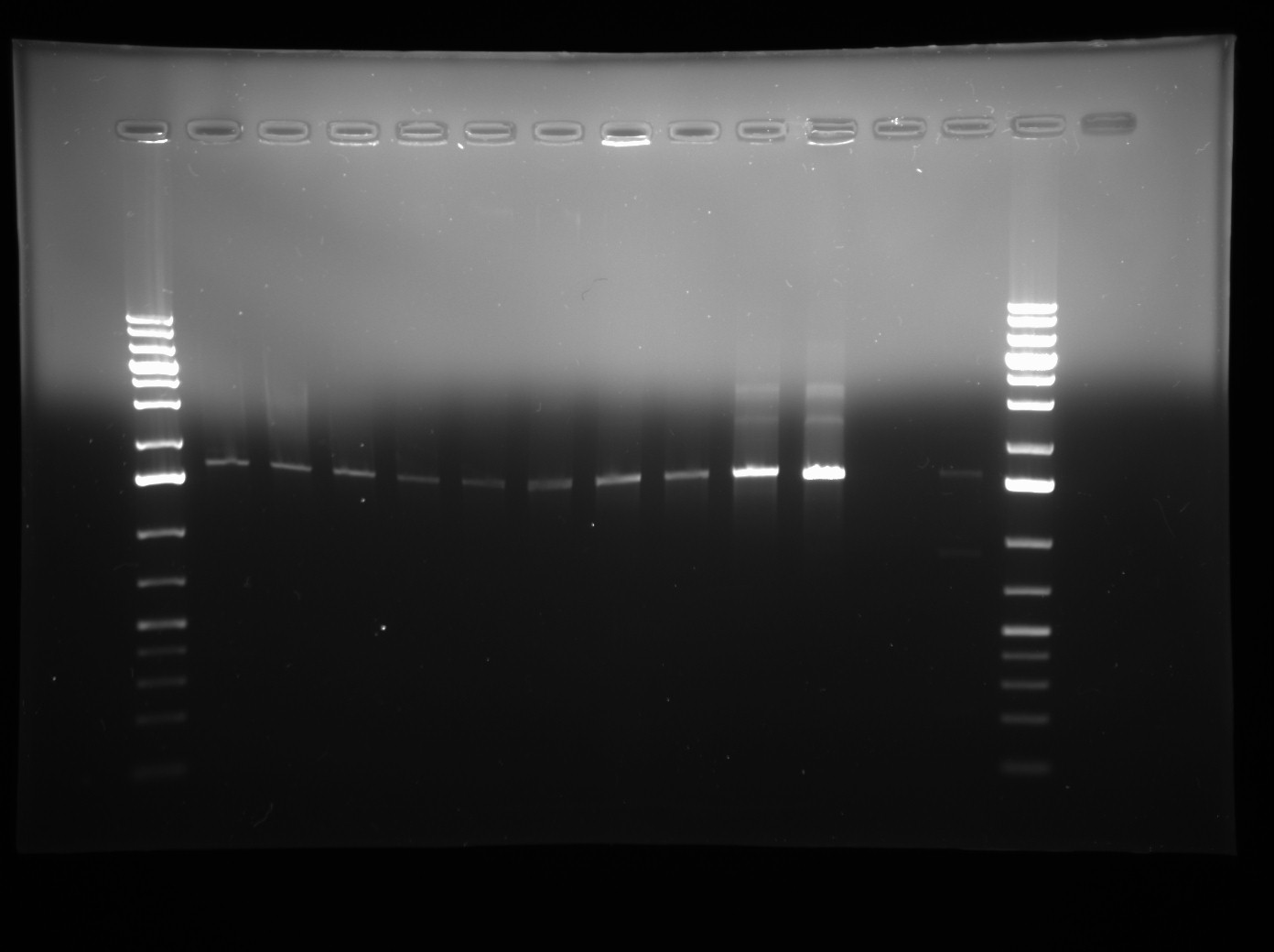
- Analysis: Lanes 1-4 are J13002-LuxOD47E-B0015 trial 1 (treating B0015 as the Insert). Lanes 5-8 are trial 2 (treating J13002-LuxOD47E as the Insert). Lane 9 is J13002-LuxOD47E (a size control), Lane 10 is LuxOD47E BBK (another size control) and Lane 11 is a negative control.
- This gel seems ot be kind of slanted, which isn't very good for size comparison. It looks like some of the first lanes of Trial 1 may have worked (contain the B0015 terminator) as they appear slightly higher than other lanes, however we will run a new gel with the PCR product to be sure.
|
|
|
FAHD
Marketing for July 10th 2009
TODAY, I concentrated my energies on marketing our iGEM project. The following are the companies I had contacted:
1) Contacted Sigma Aldrich
2) Contacted Nexen
3) Contacted Neuroimage Inc.
I will make follow up phone calls next week.
I also worked on my US visa application and attended our weekly meetings with my supervisors.
|
|
|
IMAN
One more time concentration Graphs: Shifting
Mathematica graph function (a built-in function) gets a list of numbers and plot them in a way that we have the list of numbers on Y-axis and the indexes of the numbers in the list on X-axis.
Knowing that, when we just have one cell in the simulation, and we want to plot the concentration graph of a given agent in the cell (for example AI-2), we pass the number of AI-2s in teach time step as a list to our built-in graph function and we generate it. However, when division occurs we have to pass two lists of numbers (one for first cell, and the other for the newly produced cell). That is where we are faced with a problem.
The problem is as it was mentioned Mathematica automatically plot the graph by the indexes the numbers in the list, and the indexes for the cells existing from beginning of the simulation is as same as each time step, but for newly generated cells we need to shift the graphs to the time step they become to exist.
To do so, we need to save the time steps that divisions occur, and then somehow differentiate the lists of numbers we are passing and add the time steps to the lists that are for new cells.
I have written 4 functions that they all together shift the concentration graphs, so that they work perfectly with division function.
Here is a concentration graph for AI-2 concentration:
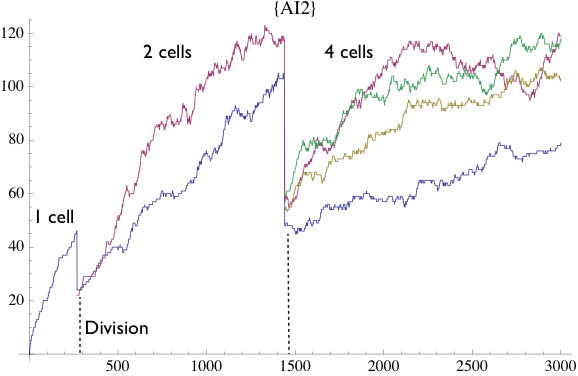
AI-2 Concentration Graph. On X-axis, time steps are aligned, and on Y-axis number of AI-2 molecules are placed. This graph shows that the simulation is run for 3000 time steps and started with one cell (blue line) and other cells are generated over time by divisions (red, green, and yellow lines). This type of results provides numerical information about the concentrations of agents and also demonstrates the general trend of change of concentrations in the bacteria. demonstrates that AI-2 is produced and accumulated in the bacteria between each division, however the concentrations of accumulated AI-2 change logarithmic which reveals that some of the AI-2 are transported from inside to the outside of the bacteria. AI-2 molecules then get divided within divisions between new cells, which is depicted by those straight lines in the graph.
|
|
|
JEREMY
Colony PCR to verify LuxPQ plasmid switch (from AC to AK – see July 8) and to verify PQ-B-R-OU-B (July 9) construct
Purpose: To perform a colony PCR to verify the presence of the above constructs. Each Colony pTaq PCR reaction used the BBK CP F/R primers, each with the following similar conditions: 94ºC for 6 minutes; 36X (94ºC for 30 seconds; 55ºC for 45 seconds; 72ºC for 4 minutes for plasmid switch (or 6 min 10 sec for PQ-B-R-OU-B construct); 72ºC for 10 minutes; held at 4ºC. The products were then run on a 0.8% agarose gel (see below). For the plasmid switch, we can see that colonies 2, 5, 6, and 8 gave the desired size of LuxPQ (3.8kb), whereas in the signaling construct verification, we see desired sizes (~6.0kb) for colonies 11, 12 and 13. Next steps will be to isolate plasmid and proceed with further verification techniques on the colonies that appear to be what we want.
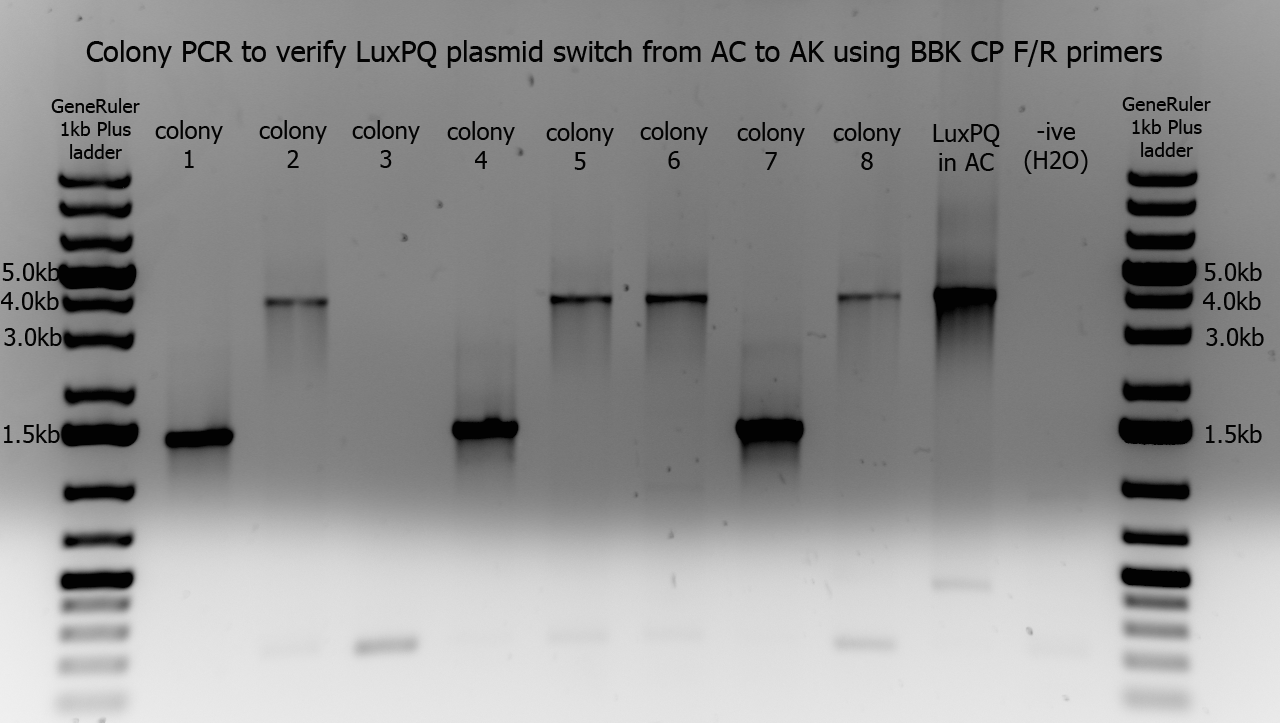
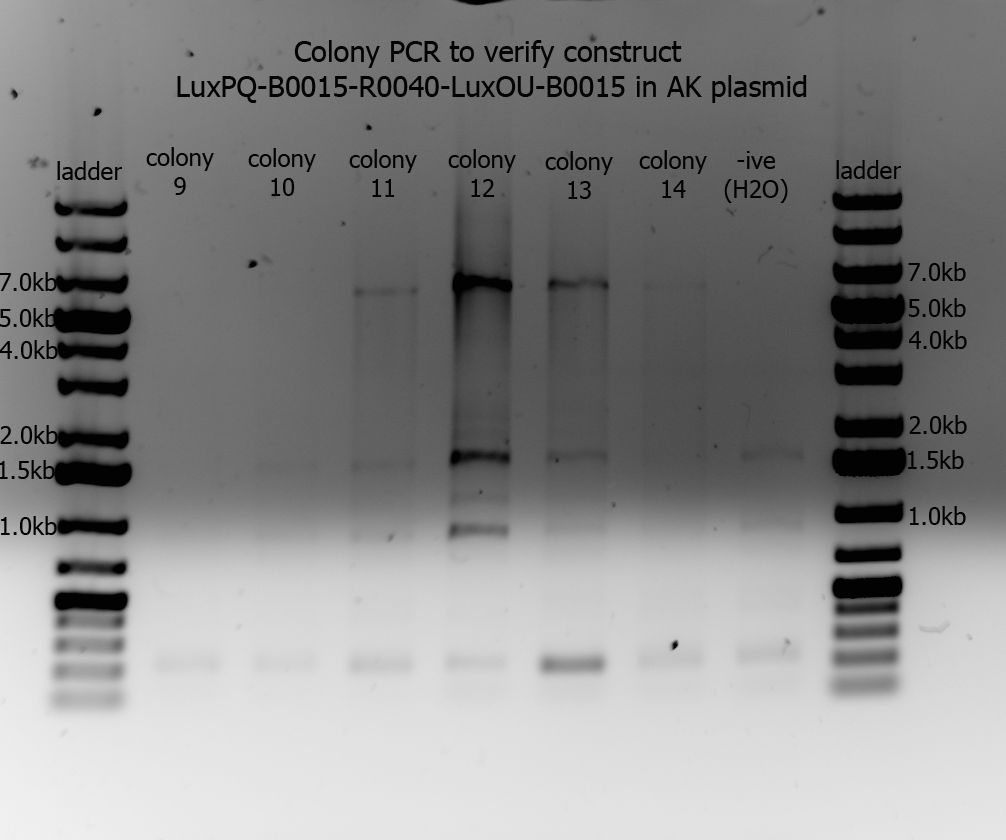
|
|
|
KEVIN
Weekly meeting: Second life
Each sectional members are now put on the spot for weekly updates, so we get more in-depth look at each accomplishments.
|
|
|
MANDY
Wiki and Second Life Robots
Fixed most of the first lab challenge.
Tried to fix wiki CSS coding, but it didn’t work (at Lethbridge workshop, we learned that the wiki can’t handle css positioning so we are switching to use tables to divide the parts of the page into sections).
The robots now respond to more sentences, so hopefully they are more user-friendly. Still need to figure out how to make it most obvious what must be said to the robot for them to work. We also had our Second Life meeting today.
|
|
|
PATRICK
Reorganization and Planning
Retrospective Notebook: This entry was not written on this day, but derived later from working notes I made that day.
Had a meeting today, got lots more feedback for the whole Second Life project from the point of view of an educator, extremely useful!
Produced a tentative list of levels for the simulator today:
- 1 my first transcription + biobrick parts
- 2 repressor
- 3 inducible repressor
- 4 repressor w. alosteric inhibition / lacI
- 5 activator
- 6 inducible activator
- 7 inhibitable activator
- 8 2 circuits. one constituitive, represses the other
- 9 bistable switch
- 10 repressilator
Began reorganizing the HUD's screens to be easier to navigate and understand. Came up with the idea of a Home screen, which is a base camp for each level, and links to all of the other features of the simulator HUD.
Also established the progression for visitors to the island, and filled in a few gaps:
Progression
- -basic controls
- -synthetic kingdonm
- -molecular bio basics
- --dna
- --central dogma
- --big picture: synthetic biology
- -biobrick tower (aka Biobrick Simulator)
- -lab (aka Virtual Lab)
|
|
|
PRIMA
Mini-prep Lux OD47A from original plates and liquid cultures
Purpose: purify upto 20 (microg) of high-copy plasmid DNA from 1.5ml overnight cultures of E.coli in LB medium.
Procedures: followed the Qiagen mini-prep procedures.
I shadowed Vicky today.
Colonies 1 & 2 - Lux OD47A BBK from original plates
Colonies 3 & 4 - Lux OD47A BBK from liquid cultures
We used 2 samples of original Colony 6 and 2 samples of plate cultures.
The following chart is showing the Nano-drop results (concentration of DNA) obtained after isolating the plasmids.
| Plasmid | 260/280 | 260/230 | Concentration [ng/μL]
|
| Lux OD47A -plate | 1.88 | 2.25 | 92.7
|
| Lux OD47A -plate | 1.84 | 1.84 | 84.8
|
| Lux OD47A - liquid culture | 1.91 | 2.20 | 69.6
|
| Lux OD47A - liquid culture | 1.98 | 2.37 | 56.6
|
|
|
|
STEFAN
Robots!
Yesterday, time was spent on building a robot (Biobert) that will act as a guide to our lab and also the rest of the island. It had to be built from scratch because the first one wasn't very good and creeped people out because it transformed from a pipette and had an empty souless expression. Once it was built, Mandy added the script so it could talk to people (including several secret phrases!). I designed
it to move its arms and mouth when touched and optimized to use the least prims possible in order to make sure we have enough left for other things on the island. Today, a second robot was also made in the same fashion (Synthia) because Biobert was lonely and he needed a friend. We also had an important meeting deciding on the structure of the Second Life areas.

|
|
|
VICKI
Plasmid isolation
…was also done exactly as I described in my July 8 update. One of my tubes struggled through the lyse stage but the rest seemed to work better. The plasmid yield was not as lucrative as last time – around 70 ng/uL (100 uL sample), but it’s an improvement on the 7 ng/uL present the first time I conducted this procedure with these cells.
|

 "
"