Team:KULeuven/Modeling/Blue Light Receptor
From 2009.igem.org
(→Mathematical Model) |
Bart Bosmans (Talk | contribs) |
||
| (11 intermediate revisions not shown) | |||
| Line 3: | Line 3: | ||
{{Team:KULeuven/Common2/EndHeader}} | {{Team:KULeuven/Common2/EndHeader}} | ||
| - | + | __NOTOC__ | |
| - | + | ||
| + | =Blue Light Receptor: Modeling= | ||
==Biological Model== | ==Biological Model== | ||
| Line 31: | Line 31: | ||
| 2.3105E-3 s<sup>-1</sup> | | 2.3105E-3 s<sup>-1</sup> | ||
| | | | ||
| - | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [ | + | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [3<html>]</html>] |
|- | |- | ||
| d<sub>Proteins</sub> | | d<sub>Proteins</sub> | ||
| 1.9254E-5 s<sup>-1</sup> | | 1.9254E-5 s<sup>-1</sup> | ||
| | | | ||
| - | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [ | + | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [4<html>]</html>] |
|- | |- | ||
! colspan="4" style="border-bottom: 1px solid #003E81;" | Transcription Rates | ! colspan="4" style="border-bottom: 1px solid #003E81;" | Transcription Rates | ||
|- | |- | ||
| - | | k<sub>transcription</sub> | + | | k<sub>YcgF</sub> |
| + | | 0.0154 s<sup>-1</sup> | ||
| + | | Rate is faster than transcription rate of YcgE | ||
| + | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [1<html>]</html>] | ||
| + | |- | ||
| + | | k<sub>YcgE</sub> | ||
| 0.00848 s<sup>-1</sup> | | 0.00848 s<sup>-1</sup> | ||
| estimate | | estimate | ||
| - | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [ | + | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [1<html>]</html>] |
| + | |- | ||
| + | ! colspan="4" style="border-bottom: 1px solid #003E81;" | Translation Rates | ||
| + | |- | ||
| + | | k<sub>YcgF</sub> | ||
| + | | 0.167s<sup>-1</sup> | ||
| + | | Rate is faster than transcription rate of YcgE | ||
| + | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [5<html>]</html>] | ||
|- | |- | ||
| - | | k<sub> | + | | k<sub>YcgE</sub> |
| 0.167 s<sup>-1</sup> | | 0.167 s<sup>-1</sup> | ||
| estimate | | estimate | ||
| - | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [ | + | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [5<html>]</html>] |
|- | |- | ||
| - | ! colspan="4" style="border-bottom: 1px solid #003E81;" | | + | ! colspan="4" style="border-bottom: 1px solid #003E81;" | Dimerization Parameters |
|- | |- | ||
| - | | k<sub> | + | | k<sub>dimerization</sub> |
| - | | | + | | 8.0E-11 (s W/m^2)<sup>-1</sup> |
| - | | Rate of | + | | Rate of dimerization of YcgF |
| + | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [1<html>]</html>] | ||
| + | |- | ||
| + | | k<sub>dissociation</sub> | ||
| + | | 0.0058 s<sup>-1</sup> | ||
| + | | Rate of dissociation of the YcgF complex | ||
| [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [2<html>]</html>] | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [2<html>]</html>] | ||
|- | |- | ||
| - | | k<sub> | + | ! colspan="4" style="border-bottom: 1px solid #003E81;" | YcgF/YcgE Interaction |
| - | | | + | |- |
| - | | Rate of | + | | k<sub>bind</sub> |
| - | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [ | + | | 100 (s molecule)<sup>-1</sup> |
| + | | Rate of binding of YcgF dimer to YcgE | ||
| + | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [1<html>]</html>] | ||
| + | |- | ||
| + | | k<sub>unbind</sub> | ||
| + | | 1 s<sup>-1</sup> | ||
| + | | Rate of dissociation of the YcgF/YcgE complex | ||
| + | | [https://2009.igem.org/Team:KULeuven/Modelling/Vanillin_Production#References [1<html>]</html>] | ||
|- | |- | ||
|} | |} | ||
| Line 77: | Line 101: | ||
[2] Y. Nakasone et al., "Transient Dimerization and Conformational Change of a BLUF Protein: YcgF", Journal of the American Chemical Society, Apr. 2006 | [2] Y. Nakasone et al., "Transient Dimerization and Conformational Change of a BLUF Protein: YcgF", Journal of the American Chemical Society, Apr. 2006 | ||
| + | |||
| + | [3] J.A. Bernstein et al., “Global analysis of mRNA decay and abundance in Escherichia coli at single-gene resolution using two-color fluorescent DNA microarrays,” Proceedings of the National Academy of Sciences of the United States of America, vol. 99, Jul. 2002, pp. 9697–9702 | ||
| + | |||
| + | [4] K. Nath et al., “Protein degradation in Escherichia Coli,” The Journal of Biological Chemistry, vol. 246, Nov. 1971, pp. 6956-6967 | ||
| + | |||
| + | [5] S.L. Gotta et al., “rRNA Transcription Rate in Escherichia Coli,” Journal of Bacteriology, vol. 173, Oct. 1991, pp. 6647-6649 | ||
| + | |||
{{Team:KULeuven/Common2/PageFooter}} | {{Team:KULeuven/Common2/PageFooter}} | ||
Latest revision as of 12:49, 18 October 2009
Blue Light Receptor: Modeling
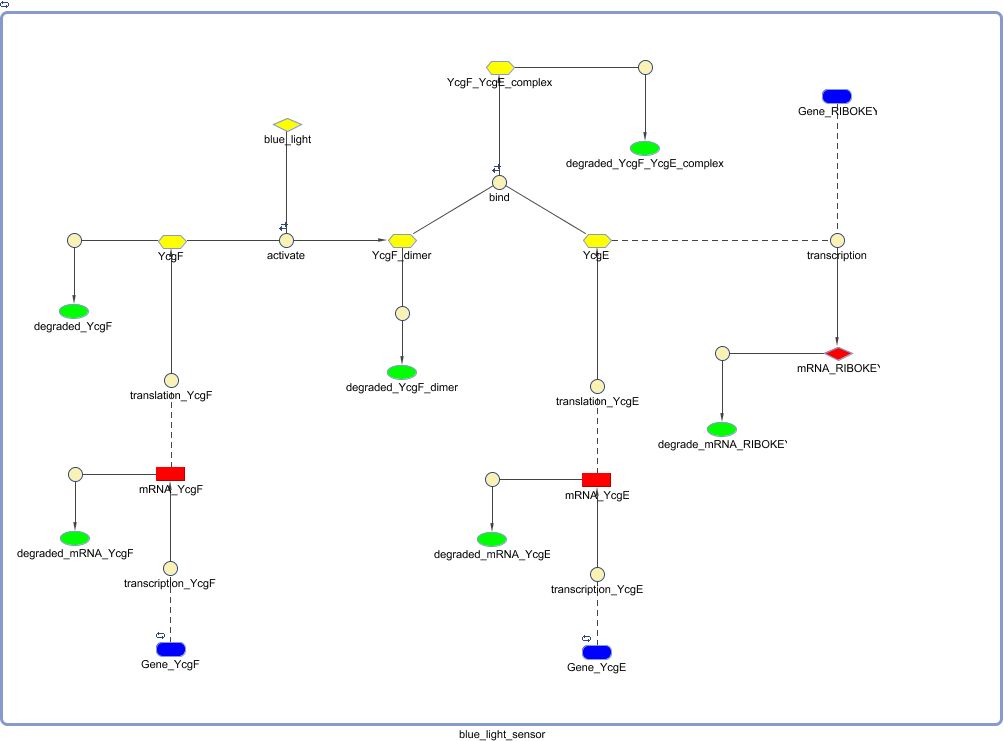
Biological Model
Mathematical Model
| Name | Value | Comments | Reference |
|---|---|---|---|
| Degradation Rates | |||
| dmRNA | 2.3105E-3 s-1 | [3] | |
| dProteins | 1.9254E-5 s-1 | [4] | |
| Transcription Rates | |||
| kYcgF | 0.0154 s-1 | Rate is faster than transcription rate of YcgE | [1] |
| kYcgE | 0.00848 s-1 | estimate | [1] |
| Translation Rates | |||
| kYcgF | 0.167s-1 | Rate is faster than transcription rate of YcgE | [5] |
| kYcgE | 0.167 s-1 | estimate | [5] |
| Dimerization Parameters | |||
| kdimerization | 8.0E-11 (s W/m^2)-1 | Rate of dimerization of YcgF | [1] |
| kdissociation | 0.0058 s-1 | Rate of dissociation of the YcgF complex | [2] |
| YcgF/YcgE Interaction | |||
| kbind | 100 (s molecule)-1 | Rate of binding of YcgF dimer to YcgE | [1] |
| kunbind | 1 s-1 | Rate of dissociation of the YcgF/YcgE complex | [1] |
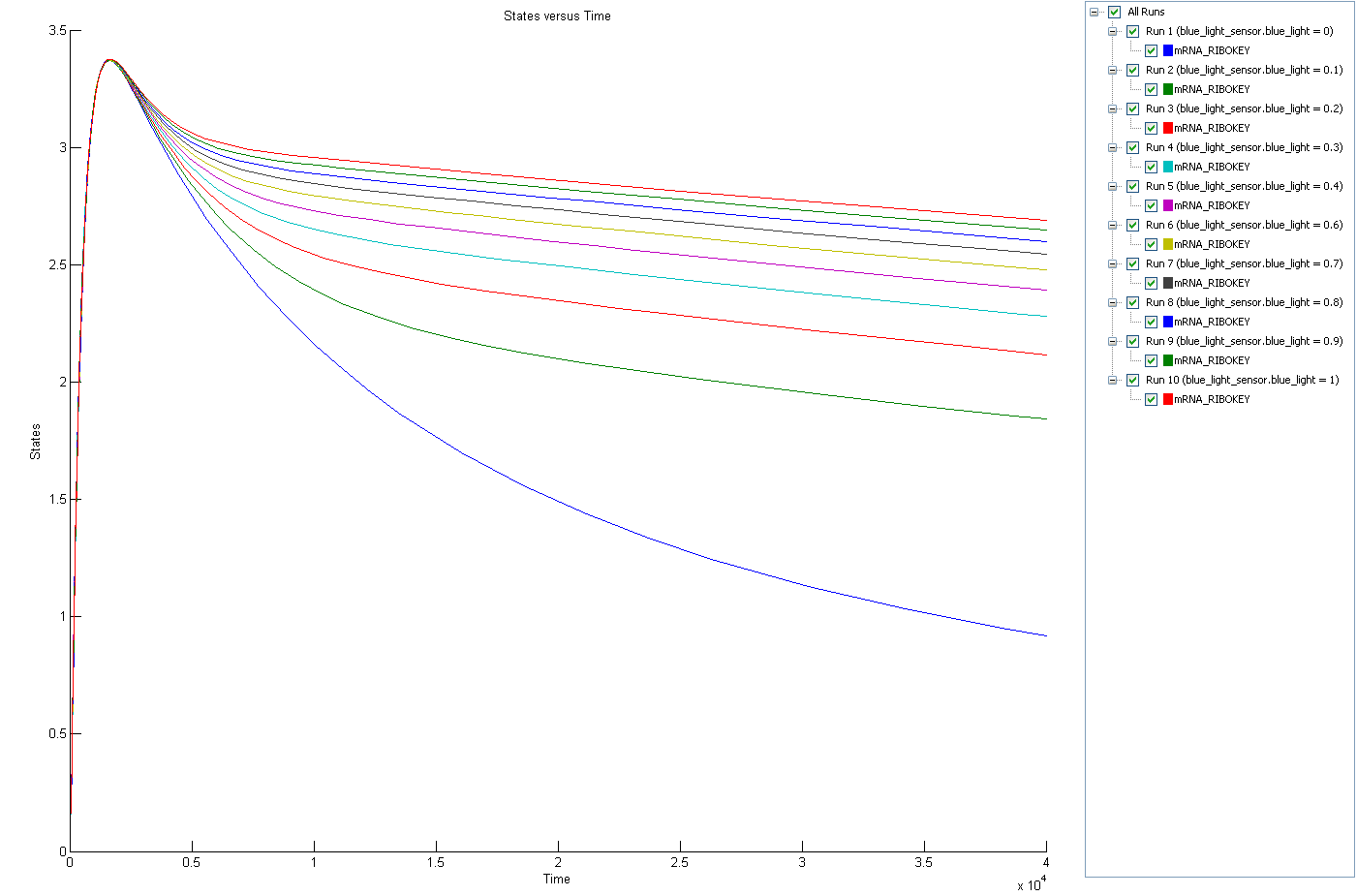
Simulation
The amount of produced RIBOKEY is positively correlated on the incident blue radiation. The build up of key in the begin period of is due to the relative slow build up of the active state of YcgF protein in compared to the build up of YcgE.
References
[1] Natalia Tschowri, Susan Busse and Regine Hengge, "The BLUF-EAL protein YcgF acts as a direct anti-repressor in a blue-light response of Escherichia coli"
[2] Y. Nakasone et al., "Transient Dimerization and Conformational Change of a BLUF Protein: YcgF", Journal of the American Chemical Society, Apr. 2006
[3] J.A. Bernstein et al., “Global analysis of mRNA decay and abundance in Escherichia coli at single-gene resolution using two-color fluorescent DNA microarrays,” Proceedings of the National Academy of Sciences of the United States of America, vol. 99, Jul. 2002, pp. 9697–9702
[4] K. Nath et al., “Protein degradation in Escherichia Coli,” The Journal of Biological Chemistry, vol. 246, Nov. 1971, pp. 6956-6967
[5] S.L. Gotta et al., “rRNA Transcription Rate in Escherichia Coli,” Journal of Bacteriology, vol. 173, Oct. 1991, pp. 6647-6649
 "
"