Team:Todai-Tokyo/Notebook/LDLR
From 2009.igem.org
(→8/14) |
|||
| (137 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
{{:Team:Todai-Tokyo/Template}} | {{:Team:Todai-Tokyo/Template}} | ||
| + | <html> | ||
| + | <head> | ||
| + | <meta http-equiv="Content-Type" content="text/html; charset=Shift_JIS"> | ||
| + | <title>the notebook</title> | ||
| + | <style type="text/css"> | ||
| + | p { font-size : 10pt } | ||
| + | pz{ font-size : 30pt; color : red} | ||
| + | --> | ||
| + | .aaa { font-size : 20pt } | ||
| + | @charset "UTF-8"; | ||
| + | body { | ||
| + | background:black; | ||
| + | } | ||
| + | div#top_plan{ | ||
| + | background:#B22222; | ||
| + | color:#ffffe0; | ||
| + | margin 0 0 0 0 | ||
| + | width:1000px; | ||
| + | height:200px; | ||
| + | } | ||
| + | |||
| + | </style> | ||
| + | |||
| + | </head> | ||
| + | <body></body></html> | ||
= Plan = | = Plan = | ||
'''Aim:''' Create yeast cells that express LDLR on their cell membrane | '''Aim:''' Create yeast cells that express LDLR on their cell membrane | ||
| Line 6: | Line 31: | ||
# Clone the LDLR gene.<BR> | # Clone the LDLR gene.<BR> | ||
# Create biobrick of LDLR.<BR> | # Create biobrick of LDLR.<BR> | ||
| + | =July= | ||
| + | We got LDLR cDNA and tried cloning.<BR> | ||
| + | Both ends of LDLR contains a lot of GC,so we had hard time... | ||
=='''7/6'''== | =='''7/6'''== | ||
Overview:<BR> | Overview:<BR> | ||
| - | 1.PCR LDLR gene from plasmid containing LDLR cDNA <BR> | + | 1.[[Team:Todai-Tokyo/Protocols/PCR|PCR]] LDLR gene from plasmid containing LDLR cDNA <BR> |
2.gel-purify DNA from the PCR product <BR> | 2.gel-purify DNA from the PCR product <BR> | ||
3.insert the DNA to vector (iGEM parts) <BR> | 3.insert the DNA to vector (iGEM parts) <BR> | ||
| - | 4. | + | 4.[[Team:Todai-Tokyo/Protocols/Transformation|Transform]] into E. coli and select for ampicillin resistance<BR> |
| - | 5.check for | + | 5.check for [[Team:Todai-Tokyo/Protocols/Transformation|Transformation]] success using colony PCR by LDLR primers<BR> |
<BR> | <BR> | ||
Constructs to be created:<BR> | Constructs to be created:<BR> | ||
| Line 25: | Line 53: | ||
Transformed 1ul of each of the above into DH5a competent cells: | Transformed 1ul of each of the above into DH5a competent cells: | ||
| - | '''Transformation''' | + | '''[[Team:Todai-Tokyo/Protocols/Transformation|Transformation]]''' |
*Mix 1ul of DNA with 100ul of competent cells on ice. | *Mix 1ul of DNA with 100ul of competent cells on ice. | ||
*Leave on ice for 30 minutes. | *Leave on ice for 30 minutes. | ||
| - | *Heat shock at | + | *Heat shock at 42ºC for 45 seconds. |
*Leave on ice for 2 minutes. | *Leave on ice for 2 minutes. | ||
| - | *Add 500ul of LB and incubate at | + | *Add 500ul of LB and incubate at 37ºC for 1 hour. |
*Plate on LB-ampicillin plates. | *Plate on LB-ampicillin plates. | ||
=='''7/7'''== | =='''7/7'''== | ||
| - | Miniprep of E.coli cells containing LDLR gene with Promega, Wizard Plus SV Miniprep DNA Purification System | + | [[Team:Todai-Tokyo/Protocols/Miniprep|Miniprep]] of E.coli cells containing LDLR gene with Promega, Wizard Plus SV[[Team:Todai-Tokyo/Protocols/Miniprep|Miniprep]] DNA Purification System |
Successful | Successful | ||
| Line 49: | Line 77: | ||
0.5ul Pfu Ultra<BR> | 0.5ul Pfu Ultra<BR> | ||
15.05ul MilliQ water<BR> | 15.05ul MilliQ water<BR> | ||
| - | + | <BR> | |
Performed PCR using the following program:<BR> | Performed PCR using the following program:<BR> | ||
| - | 1. | + | 1. 95ºC 2min<BR> |
| - | 2. | + | 2. 95ºC 30sec <BR> |
| - | 3. | + | 3. 55ºC 30sec<BR> |
| - | 4. 72. | + | 4. 72.5ºC 60sec<BR> |
5. repeat 2-4 29times<BR> | 5. repeat 2-4 29times<BR> | ||
| - | 6. | + | 6. 25ºC forever<BR> |
<BR> | <BR> | ||
PCR successful<BR> | PCR successful<BR> | ||
We cut the region of LDLR out of the gel and purified PCR product using Promega kit.<BR> | We cut the region of LDLR out of the gel and purified PCR product using Promega kit.<BR> | ||
<BR> | <BR> | ||
| - | + | [[Team:Todai-Tokyo/Protocols/Infusion|Infusion]] of LDLR<BR> | |
<BR> | <BR> | ||
4ul PCR product (12.35ug/ml)<BR> | 4ul PCR product (12.35ug/ml)<BR> | ||
| Line 67: | Line 95: | ||
2ul 5×infusion reaction buffer<BR> | 2ul 5×infusion reaction buffer<BR> | ||
1ul infusion enzyme<BR> | 1ul infusion enzyme<BR> | ||
| - | + | |<BR> | |
| - | + | v<BR> | |
| - | + | 37ºC 15min<BR> | |
| - | + | 50ºC 15min<BR> | |
| + | |<BR> | ||
| + | v<BR> | ||
| + | |||
TE buffer up to 20ul<BR> | TE buffer up to 20ul<BR> | ||
| - | + | |<BR> | |
| + | v<BR> | ||
| + | |||
added 10ul of the sample to DH5α (090614) and put on ice for 15 min.<BR> | added 10ul of the sample to DH5α (090614) and put on ice for 15 min.<BR> | ||
| - | + | |<BR> | |
| - | + | v<BR> | |
| - | + | ||
| + | 42ºC 45sec<BR> | ||
| + | |<BR> | ||
| + | v<BR> | ||
added 500ul LB broth to the tube.<BR> | added 500ul LB broth to the tube.<BR> | ||
| - | + | |<BR> | |
| + | v<BR> | ||
placed it on LB ampicilin plate. <BR> | placed it on LB ampicilin plate. <BR> | ||
<BR> | <BR> | ||
| Line 87: | Line 124: | ||
<BR> | <BR> | ||
put a small amount of single colony into each tube with 5ul MilliQ water<BR> | put a small amount of single colony into each tube with 5ul MilliQ water<BR> | ||
| - | + | |<BR> | |
| - | + | v<BR> | |
| - | + | ||
| + | 95ºC 5min<BR> | ||
| + | |<BR> | ||
| + | v<BR> | ||
| + | |||
PCR reaction<BR> | PCR reaction<BR> | ||
1ul 10×buffer <BR> | 1ul 10×buffer <BR> | ||
| Line 97: | Line 138: | ||
0.1ul 3’-primer<BR> | 0.1ul 3’-primer<BR> | ||
2.92ul MilliQ water<BR> | 2.92ul MilliQ water<BR> | ||
| - | + | |<BR> | |
| + | v<BR> | ||
added the PCR reaction to each tube.<BR> | added the PCR reaction to each tube.<BR> | ||
| - | + | |<BR> | |
| + | v<BR> | ||
Performed PCR using the following program:<BR> | Performed PCR using the following program:<BR> | ||
| - | 1. | + | 1. 95ºC 2min<BR> |
| - | 2. | + | 2. 95ºC 30sec <BR> |
| - | 3. | + | 3. 55ºC 30sec<BR> |
| - | 4. 72. | + | 4. 72.5ºC 120sec<BR> |
| - | 5. repeat 2-4 | + | 5. repeat 2-4 29 times<BR> |
| - | 6. | + | 6. 25ºC forever<BR> |
<BR> | <BR> | ||
PCR unsuccessful・・・.<BR> | PCR unsuccessful・・・.<BR> | ||
| Line 112: | Line 155: | ||
=='''7/25'''== | =='''7/25'''== | ||
| - | gal1 | + | gal1 [[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]] by BIG DYE<BR> |
1.8ul 5xB.D.3.1.buffer<BR> | 1.8ul 5xB.D.3.1.buffer<BR> | ||
| Line 119: | Line 162: | ||
1ul plasmid(0.15ug/ul)<BR> | 1ul plasmid(0.15ug/ul)<BR> | ||
0.5ul 5'or3'primer(3.2pmol/ul)<BR> | 0.5ul 5'or3'primer(3.2pmol/ul)<BR> | ||
| - | + | |<BR> | |
| + | v<BR> | ||
| + | |||
PCR Program<BR> | PCR Program<BR> | ||
| - | 1. | + | 1.96ºC 2min<BR> |
| - | 2. | + | 2.96ºC 10sec<BR> |
| - | 3. | + | 3.55ºC 5sec<BR> |
| - | 4. | + | 4.60ºC 3min <BR> |
5.go to 2.29times<BR> | 5.go to 2.29times<BR> | ||
| - | 6. | + | 6.25ºC forever<BR> |
| - | + | |<BR> | |
| - | + | v<BR> | |
| - | + | dd 0.5ul PHOSPHATASE ALKALINE shrimp<BR> | |
| - | + | |<BR> | |
| - | + | v<BR> | |
| + | 37ºC 1hr incubate<BR> | ||
| + | |<BR> | ||
| + | v<BR> | ||
add 1ul 3MNaOAc<BR> | add 1ul 3MNaOAc<BR> | ||
| - | + | |<BR> | |
| + | v<BR> | ||
add 25ul EtOH<BR> | add 25ul EtOH<BR> | ||
| - | + | |<BR> | |
| - | 20000xg | + | v<BR> |
| - | + | 20000xg 4ºC 10min centrifugation<BR> | |
| + | |<BR> | ||
| + | v<BR> | ||
put off supernatant<BR> | put off supernatant<BR> | ||
| - | + | |<BR> | |
| + | v<BR> | ||
dry tubes<BR> | dry tubes<BR> | ||
| - | + | |<BR> | |
| + | v<BR> | ||
add 15ul HiDi<BR> | add 15ul HiDi<BR> | ||
| - | + | |<BR> | |
| + | v<BR> | ||
put them in the sequence machine<BR> | put them in the sequence machine<BR> | ||
| Line 149: | Line 203: | ||
colony PCR again, using 7/20 protocol.<BR> | colony PCR again, using 7/20 protocol.<BR> | ||
PCR program<BR> | PCR program<BR> | ||
| - | 1. | + | 1.95ºC 2min <BR> |
| - | 2. | + | 2.95ºC 30sec<BR> |
| - | 3. | + | 3.55ºC 30sec<BR> |
| - | 4.72. | + | 4.72.5ºC 150sec<BR> |
5.go to 2. 29times<BR> | 5.go to 2. 29times<BR> | ||
| Line 158: | Line 212: | ||
=='''7/27'''== | =='''7/27'''== | ||
| - | Miniprep of E.coli cells containing LDLR gene(that yesterday picked up and cultured) with Promega, Wizard Plus SV Miniprep DNA Purification System <BR> | + | Miniprep of E.coli cells containing LDLR gene(that yesterday picked up and cultured) with Promega, Wizard Plus SV [[Team:Todai-Tokyo/Protocols/Miniprep|Miniprep]] DNA Purification System <BR> |
using AGE, found a colony that has LDLR infusion plasmid<BR> | using AGE, found a colony that has LDLR infusion plasmid<BR> | ||
| - | ==August== | + | |
| + | |||
| + | == 7/30 == | ||
| + | *[[Team:Todai-Tokyo/Protocols/sequencing|sequencing]] of LDLR | ||
| + | |||
| + | =August= | ||
| + | We found our plasmid that we got first didn't include LDLR cDNA!? | ||
| + | We came back to start line again!? | ||
| + | == 8/1 == | ||
| + | PCR of GFP for fusion<BR> | ||
| + | program<BR> | ||
| + | 1.95ºC 2min<BR> | ||
| + | 2.95ºC 30sec<BR> | ||
| + | 3.55ºC 30sec<BR> | ||
| + | 4.72.5ºC 20sec<BR> | ||
| + | 5.go to 2. 29 times<BR> | ||
| + | 6.25ºC forever<BR> | ||
| + | <BR> | ||
We found that the length of these PCR product till now was longer than that of LDLR.<BR> | We found that the length of these PCR product till now was longer than that of LDLR.<BR> | ||
| - | *read the | + | * read the [[Team:Todai-Tokyo/Protocols/Sequencing|Sequence]] of F-spe1-Gall p-LDLR-5<BR> |
| + | * read the [[Team:Todai-Tokyo/Protocols/Sequencing|Sequence]] of F-pst1-Gall p-LDLR-3<BR> | ||
| + | ->the result didn't conform with NCBI datebase<BR> | ||
*change PCR program→didn't get the PCR product whose length was correct<BR> | *change PCR program→didn't get the PCR product whose length was correct<BR> | ||
| - | |||
| + | |||
| + | == 8/2 == | ||
| + | *[[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]] of LDLR<BR> | ||
| + | |||
| + | == 8/4 == | ||
| + | |||
| + | The following were [[Team:Todai-Tokyo/Protocols/Sequencing|sequenced]] using the labeled primers: <BR> | ||
| + | * 1)F-spe1-Gall p-LDLR-5<BR> | ||
| + | * 2)F-pst1-Gall p-LDLR-3<BR> | ||
| + | |||
| + | '''Results''':<BR> | ||
| + | Sequence read failed | ||
| + | |||
| + | == 8/5 == | ||
| + | *[[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]] of LDLR<BR> | ||
| + | |||
| + | == 8/6 == | ||
| + | |||
| + | *[[Team:Todai-Tokyo/Protocols/PCR|PCR]] of LDLR1<BR> | ||
| + | *[[Team:Todai-Tokyo/Protocols/PCR|PCR]] of LDLR2<BR> | ||
| + | |||
| + | LDLR1 and LDLR2 is combination of Gal1, LDLR and GFP.<BR> | ||
| + | |||
| + | == 8/7 == | ||
| + | *[[Team:Todai-Tokyo/Protocols/PCR|PCR]] of LDLR | ||
| + | using the labeled primers: <BR> | ||
| + | * 1)F-spe1-Gal1-LDLR-5'<BR> | ||
| + | * 2)F-spe1-Gal1-LDLR-3'<BR> | ||
| + | |||
| + | |||
| + | *[[Team:Todai-Tokyo/Protocols/PCR|PCR]] of GFP | ||
| + | using the labeled primers: <BR> | ||
| + | * 1)F-spe1-Gal1-LDLR-GFP-5'<BR> | ||
| + | * 2)F-pst1-Gal1-LDLR-GFP-3'<BR> | ||
| + | |||
| + | == 8/8 == | ||
| + | *[[Team:Todai-Tokyo/Protocols/Transformation|Transformation]] of P3 21D<BR> | ||
| + | *[[Team:Todai-Tokyo/Protocols/Transformation|Transformation]] of P1 23L<BR> | ||
| + | |||
| + | |||
| + | *[[Team:Todai-Tokyo/Protocols/PCR|PCR]] of LDLR | ||
| + | using the labeled primers: <BR> | ||
| + | * 1)F-spe1-Gal1-LDLR-5'<BR> | ||
| + | * 2)F-spe1-Gal1-LDLR-3'<BR> | ||
| + | |||
| + | using the following program; | ||
| + | |||
| + | program<BR> | ||
| + | 1.95ºC 2min<BR> | ||
| + | 2.95ºC 30sec<BR> | ||
| + | 3.52~55ºC 30sec<BR> | ||
| + | 4.72.5ºC 60sec<BR> | ||
| + | 5.go to 2. 29 times<BR> | ||
| + | 6.25ºC forever<BR> | ||
| + | <BR> | ||
| + | |||
| + | Result;<BR> | ||
| + | The length of LDLR is about 2583 bp.<BR> | ||
| + | |||
| + | *[[Team:Todai-Tokyo/Protocols/PCR|PCR]] of LDLR | ||
| + | using the labeled primers: <BR> | ||
| + | * 1)F-spe1-Gal1-LDLR-5'<BR> | ||
| + | * 2)F-spe1-Gal1-LDLR-3'<BR> | ||
| + | |||
| + | using the following program; | ||
| + | |||
| + | program<BR> | ||
| + | 1.95ºC 2min<BR> | ||
| + | 2.95ºC 30sec<BR> | ||
| + | 3.57ºC 30sec<BR> | ||
| + | 4.72.5ºC 60sec<BR> | ||
| + | 5.go to 2. 29 times<BR> | ||
| + | 6.25ºC forever<BR> | ||
| + | <BR> | ||
| + | |||
| + | The change point of this program is the temperature at third step.<BR> | ||
| + | This temperature is 57ºC but previous is 52~55ºC.<BR> | ||
| + | |||
| + | result;<BR> | ||
| + | The length of LDLR is 2500~3000bp.<BR> | ||
| + | |||
| + | == 8/9 == | ||
| + | *[[Team:Todai-Tokyo/Protocols/PCR|PCR]] of LDLR | ||
| + | |||
| + | == 8/10 == | ||
| + | *[[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]] of LDLR <BR> | ||
| + | The following were sequenced using the labeled primers<BR> | ||
| + | |||
| + | 1)1D-LDLR-5' <BR> | ||
| + | 2)1D-LDLR-3' <BR> | ||
| + | |||
| + | Results:<BR> | ||
| + | Sequence read failed | ||
| + | |||
| + | |||
| + | *PCR of LDLR(1st time) | ||
| + | using the labeled primers: <BR> | ||
| + | 1)F-spe1-Gal1-LDLR-GFP-5'<BR> | ||
| + | 2)F-spe1-Gal1-LDLR-GFP-3'<BR> | ||
| + | |||
| + | using the following program; | ||
| + | |||
| + | program<BR> | ||
| + | 1.95ºC 2min<BR> | ||
| + | 2.95ºC 30sec<BR> | ||
| + | 3.55ºC 30sec<BR> | ||
| + | 4.72.5ºC 60sec<BR> | ||
| + | 5.go to 2. 29 times<BR> | ||
| + | 6.25ºC forever<BR> | ||
| + | <BR> | ||
| + | |||
| + | Result;<BR> | ||
| + | PCR is sucessful.<BR> | ||
| + | |||
| + | *PCR of LDLR(2nd time) | ||
| + | using the labeled primers is same as 1st time's: <BR> | ||
| + | |||
| + | using the following program; | ||
| + | |||
| + | program<BR> | ||
| + | 1.95ºC 2min<BR> | ||
| + | 2.96ºC 30sec<BR> | ||
| + | 3.55ºC 30sec<BR> | ||
| + | 4.60.0ºC 3min<BR> | ||
| + | 5.go to 2. 29 times<BR> | ||
| + | 6.25ºC forever<BR> | ||
| + | <BR> | ||
| + | |||
| + | == 8/12 == | ||
| + | *[[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]] of LDLR | ||
| + | Result;<BR> | ||
| + | NNNNN can't read. | ||
| + | *electrophorese LDLR+GFP | ||
| + | RESULT:the band across 3000-3500bp not found. | ||
| + | -> need to rePCR LDLR+GFP. | ||
| + | |||
| + | == 8/13 == | ||
| + | *measure the absorbance of LDLR2cDNA | ||
| + | ->534ug/mL | ||
| + | +PCR | ||
| + | objects:LDLR2, yqiT, GFP | ||
| + | program | ||
| + | 1.95ºC 2min<BR> | ||
| + | 2.96ºC 30sec<BR> | ||
| + | 3.55ºC 30sec<BR> | ||
| + | 4.60.0ºC 3min<BR> | ||
| + | 5.go to 2. 29 times<BR> | ||
| + | 6.25ºC forever<BR> | ||
| + | *electrophorese the above 3 | ||
| + | result -> | ||
| + | other than GFP not found | ||
| + | <- maybe DNAs dissolved by UV or contamination; | ||
| + | |||
| + | == 8/14 == | ||
| + | *[[Team:Todai-Tokyo/Protocols/PCR|PCR]] <BR> | ||
| + | objects: LDLR GFP<BR> | ||
| + | LDLR<BR> | ||
| + | primer<BR> | ||
| + | F_S_GalI_LDLR_5'<BR> | ||
| + | F_PstI_GalI_LDLR_3'<BR> | ||
| + | |||
| + | GFP<BR> | ||
| + | primer <BR> | ||
| + | F_S_GalI_LDLR_GFP 5'<BR> | ||
| + | F_P_GalI_LDLR_GFP 3'<BR> | ||
| + | <BR> | ||
| + | *electrophorese PCR products<BR> | ||
| + | ->failed (LDLR+GFP) | ||
| + | |||
| + | == 8/15 == | ||
| + | rePCR LDLR+GFP | ||
| + | electrophorese LDLR+GFP | ||
| + | == 8/19 == | ||
| + | |||
| + | 1 Colony PCR of LDLR<BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) J63010(P17D)_seq_5’<BR> | ||
| + | 2) J63010(P17D)_seq_3’<BR> | ||
| + | Result:<BR> | ||
| + | successful <BR> | ||
| + | |||
| + | 2 PCR of LDLR for fusion<BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) F_S_Gal1_LDLR_5’new<BR> | ||
| + | 2)F_LDLR_3’new<BR> | ||
| + | |||
| + | 3 PCR of GFP for fusion<BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) F_S_Gal1_LDLR_GFP_5’<BR> | ||
| + | 2) F_S_Gsl1_LDLR_GFP_3’<BR><BR> | ||
| + | |||
| + | == 8/21 == | ||
| + | |||
| + | 1 PCR of LDLR<BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) F_S_Gal1_LDLR_5’new<BR> | ||
| + | 2)F_LDLR_3’<BR><BR> | ||
| + | |||
| + | 2 [[Team:Todai-Tokyo/Protocols/Infusion|Infusion]] of LDLR and GFP<BR><BR> | ||
| + | |||
| + | == 8/22 == | ||
| + | |||
| + | 1 [[Team:Todai-Tokyo/Protocols/Miniprep|Miniprep]] ofLDLR<BR><BR> | ||
| + | |||
| + | == 8/23 == | ||
| + | |||
| + | 1 [[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]] of LDLR<BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) J_P17D_5’<BR> | ||
| + | 2) J_P17D_3’<BR><BR> | ||
| + | |||
| + | == 8/25 == | ||
| + | |||
| + | 1 PCR of LDLR<BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) F_S_Gp_LDLR_out_5’<BR> | ||
| + | 2) F_P_Gp_LDLR_out_3’<BR><BR> | ||
| + | |||
| + | == 8/26 == | ||
| + | |||
| + | 1 PCR of LDLR<BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) F_S_Gp_LDLR_out_5’<BR> | ||
| + | 2) F_P_Gp_LDLR_out_3’<BR><BR> | ||
| + | |||
| + | 2 [[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]] of LDLR cDNA<BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) F_S_Gp_LDLR_out_5’<BR> | ||
| + | 2)F_P_Gp_LDLR_out_3’<BR> | ||
| + | Result : failed<BR><BR> | ||
| + | |||
| + | == 8/27 == | ||
| + | |||
| + | 1 PCR of LDLR <BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) F_S_Gp_LDLR_out_5’<BR> | ||
| + | 2) F_P_Gp_LDLR_out_3'<BR><BR> | ||
| + | |||
| + | 2 [[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]] of LDLR cDNA<BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) F_S_Gp_LDLR_out_5’<BR> | ||
| + | 2)F_P_Gp_LDLR_out_3’<BR> | ||
| + | Result : failed<BR><BR> | ||
| + | |||
| + | == 8/30 == | ||
| + | |||
| + | *PCR of LDLR<BR> | ||
| + | using the labeled primers: <BR> | ||
| + | 1)F-spe1-Gal1-LDLR-GFP-5'<BR> | ||
| + | 2)F-spe1-Gal1-LDLR-GFP-3'<BR> | ||
| + | |||
| + | == 8/31 == | ||
| + | |||
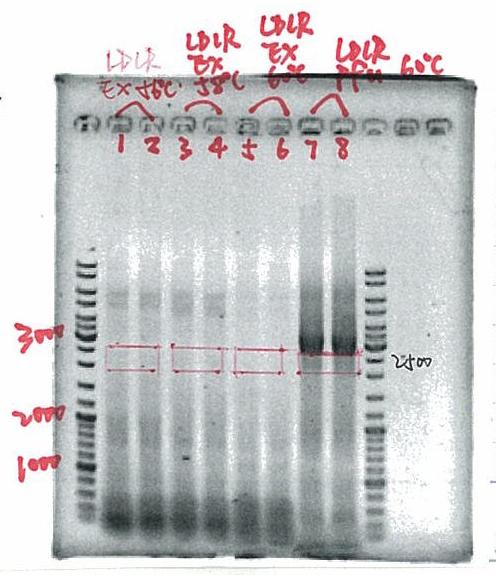
| + | [[Image:Ldl.jpg|right|150px]] | ||
| + | [[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]] of LDLR<BR> | ||
| + | The following were sequenced using the labeled primers<BR> | ||
| + | |||
| + | 1)1D-LDLR-5' <BR> | ||
| + | 2)1D-LDLR-3' <BR> | ||
| + | |||
| + | Results:<BR> | ||
| + | Sequence read failed<BR><BR><BR> | ||
| + | |||
| + | =September= | ||
| + | We got new LDLR cDNA!<BR> | ||
| + | *look for the best PCR program<BR> | ||
| + | →DMSO 5% or 10%<BR> | ||
| + | →annealing temparature 55ºC or 60ºC<BR><BR> | ||
| + | result<BR> | ||
| + | the best program<BR> | ||
| + | 10% DMSO<BR> | ||
| + | 1.95ºC 2min<BR> | ||
| + | 2.95ºC 30sec<BR> | ||
| + | 3.60ºC 30sec<BR> | ||
| + | 4.72.5ºC 1min<BR> | ||
| + | 5.go to 2. 29 times<BR> | ||
| + | 6.25ºC forever<BR> | ||
| + | purify the PCR product using Promega kit and insert it in iGEM part(plate1-7D:Gal1 promoter) <BR> | ||
| + | And, insert it and GFP in iGEM part(plate1-7D:Gal1 promoter) | ||
| + | |||
| + | =='''9/15'''== | ||
| + | 1. PCR + Gel Extraction<BR> | ||
| + | *LDLR<BR> | ||
| + | |||
| + | 2. PCR<BR> | ||
| + | *LDLR (annealing temperature:57)<BR> | ||
| + | *LDLR (annealing temperature:60)<BR> | ||
| + | |||
| + | 3. PCR<BR> | ||
| + | *LDLR: tested different concentrations of template (do not contain DMSO)<BR> | ||
| + | *1. X 1/10<BR> | ||
| + | *2. X 1/5<BR> | ||
| + | *3. X 1 (normal)<BR> | ||
| + | *4. X 5<BR> | ||
| + | *5. X 10<BR> | ||
| + | |||
| + | =='''9/16'''== | ||
| + | 1. PCR | ||
| + | *LDLR: tested different concentrations of template<BR> | ||
| + | *1. X 1/10 + 20%DMSO<BR> | ||
| + | *2. X 1/5 + 20%DMSO<BR> | ||
| + | *3. X 1 (normal) + 20%DMSO<BR> | ||
| + | *4. X 5 + 20%DMSO<BR> | ||
| + | *5. X 10 + 20%DMSO<BR> | ||
| + | |||
| + | 2. PCR<BR> | ||
| + | *LDLR: tested different conditions<BR> | ||
| + | *1. cDNA1, old primer, 20% DMSO<BR> | ||
| + | *2. cDNA1, old primer, no DMSO<BR> | ||
| + | *3. cDNA1, new primer, 20% DMSO<BR> | ||
| + | *4. cDNA1, new primer, no DMSO<BR> | ||
| + | *5. cDNA2, old primer, 20% DMSO<BR> | ||
| + | *6. cDNA2, old primer, no DMSO<BR> | ||
| + | *7. cDNA2, new primer, 20% DMSO<BR> | ||
| + | *8. cDNA2, new primer, no DMSO<BR> | ||
| + | *9. control<BR> | ||
| + | |||
| + | =='''9/24'''== | ||
| + | 1. PCR | ||
| + | * GEP | ||
| + | * YCplac111 | ||
| + | |||
| + | =='''9/25'''== | ||
| + | *Miniprep of E.coli cells containing GFP gene (those yesterday picked up and cultured) with Promega, Wizard Plus SV [[Team:Todai-Tokyo/Protocols/Miniprep|Miniprep]] DNA Purification System <BR> | ||
| + | *read the [[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]] of LDLR cDNA<BR> | ||
| + | Results:<BR> | ||
| + | Sequence read failed<BR><BR><BR> | ||
| + | |||
| + | |||
| + | *PCR of LDLR cDNA(Pfu UltraII)<BR> | ||
| + | Results:<BR> | ||
| + | PCR failed<BR><BR><BR> | ||
| + | |||
| + | =='''9/26'''== | ||
| + | *PCR of LDLR for infusion<BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) F_S_Gal1_LDLR_5’new<BR> | ||
| + | 2)F_pstI_Gal1p_LDLR_3’new<BR> | ||
| + | Results:<BR> | ||
| + | PCR failed<BR><BR><BR> | ||
| + | |||
| + | *PCR of LDLR for fusion<BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) F_S_Gal1_LDLR_5’new<BR> | ||
| + | 2)F_LDLR_3’new<BR> | ||
| + | Results:<BR> | ||
| + | PCR failed<BR><BR><BR> | ||
| + | |||
| + | *PCR of GFP for fusion<BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) F_S_Gal1_LDLR_GFP_5’<BR> | ||
| + | 2) F_p_Gal1_LDLR_GFP_3’<BR><BR> | ||
| + | Results:<BR> | ||
| + | PCR failed<BR><BR><BR> | ||
| + | |||
| + | =='''9/27'''== | ||
| + | *PCR of LDLR <BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) F_S_Gp_LDLRout_5'<BR> | ||
| + | 2)F_P_Gp_LDLRout_3'<BR> | ||
| + | Results:<BR> | ||
| + | PCR failed<BR><BR><BR> | ||
| + | |||
| + | *PCR of LDLR <BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) F_S_Gal1_LDLR_5'new<BR> | ||
| + | 2)F_P_Gal1p_LDLR_3'<BR> | ||
| + | Results:<BR> | ||
| + | PCR failed<BR><BR><BR> | ||
| + | |||
| + | *PCR of LDLR <BR> | ||
| + | Using the labeled primers:<BR> | ||
| + | 1) F_S_Gal1_LDLR_5'new<BR> | ||
| + | 2)F_LDLR_3'<BR> | ||
| + | Results:<BR> | ||
| + | PCR failed<BR><BR><BR> | ||
| + | |||
| + | *read the [[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]] of LDLR cDNA<BR> | ||
| + | Results:<BR> | ||
| + | Sequence read failed<BR><BR><BR> | ||
| + | |||
| + | =='''9/28'''== | ||
| + | *PCR of BC LDLR <BR> | ||
| + | primers:<BR> | ||
| + | F_S_Gal1_LDLR_5'new<br> | ||
| + | F_pst1_Gal1p_LDLR_3'<br><br> | ||
| + | |||
| + | *again, PCR of LFL, HEP and BC LDLR<br> | ||
| + | primer:<br> | ||
| + | 1)F_S_Gp_LDLR_out_5'<br> | ||
| + | 1)F_P_Gp_LDLR_out_3'<br> | ||
| + | 2)F_S_Gal1p_LDLR_5'new<br> | ||
| + | 2)F_pst1_Gal1p_LDLR_3'<br> | ||
| + | 3)F_S_Gal1_LDLR_5'new<br> | ||
| + | 3)F_LDLR_3'primer<br><br> | ||
| + | |||
| + | =='''9/29'''== | ||
| + | |||
| + | *sequencing of YCplaC111<br><br> | ||
| + | |||
| + | *infusion of LDLR<br> | ||
| + | vector:P17D<br><br> | ||
| + | |||
| + | =='''9/30'''== | ||
| + | *colony PCR of LDLR and P17D+LDLR and P17D+LDLR+GFP<br> | ||
| + | primer:<br> | ||
| + | F_S_Gal1_LDLR_5'new<br> | ||
| + | F_Pst1_Gal1p_LDLR_3'<br><br> | ||
| + | |||
| + | *again, colony PCR of P17D+LDLR+GFP<br> | ||
| + | primer:<br> | ||
| + | F_S_Gal1_LDLR_5'new<br> | ||
| + | F_Pst1_Gal1p_LDLR_3'<br><br> | ||
| + | |||
| + | =October= | ||
| + | LDLR made a debut as an iGEM part!<BR> | ||
| + | That was very long way... | ||
| + | |||
| + | =='''10/4'''== | ||
| + | *PCR of GFP for LDLR primer<br> | ||
| + | primer:<br> | ||
| + | F_S_Gal1_LDLR_GFP_5'<br> | ||
| + | F_P_Gal1_LDLR_GRP_3'<br> | ||
| + | |||
| + | |||
| + | *colony PCR of LDLR+GRP<br> | ||
| + | primer:<br> | ||
| + | P17Dseq_5'<br> | ||
| + | P17Dseq_3'<br> | ||
| + | |||
| + | *infusion of LDLF+GFP+P17D<br> | ||
| + | vector:P17D digested with S and P<br> | ||
| + | |||
| + | =='''10/5'''== | ||
| + | *check of plasmids including LDLR by AGE<BR> | ||
| + | 1.LDLR3 091004<BR> | ||
| + | 2.LDLR5 091004<BR> | ||
| + | 3.LDLR+GFP 1 091004<BR> | ||
| + | 4.LDLR+GFP 2 091004<BR> | ||
| + | 1&2->OK,3&4->wouldn't have LDLR+GFP<BR> | ||
| + | *[[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]]<BR> | ||
| + | 1.LDLR3 091004<BR> | ||
| + | 2.LDLR5 091004<BR> | ||
| + | 3.LDLR+GFP 1 091004<BR> | ||
| + | 4.LDLR+GFP 2 091004<BR> | ||
| + | 5~10.YCplacIII(whose restriction site may be changed) <BR> | ||
| + | primer<BR> | ||
| + | 1~4:P1-7Dseq.5'or3'<BR> | ||
| + | 5~10:YCplacIII seq. primer5'or3'<BR> | ||
| + | *PCR for LDLR+GFP fusion(Pfu UltraII)<BR> | ||
| + | template:HEP-LDLR cDNA<BR> | ||
| + | primer:<BR> | ||
| + | F_S_Gal1_LDLR_5'new<BR> | ||
| + | F_P_GAl1_LDLR_GFP_5'<BR> | ||
| + | |||
| + | =='''10/7'''== | ||
| + | *PCR for GFP fusion(Pfu UltraII)<BR> | ||
| + | primer<BR> | ||
| + | F_S_Gal1_LDLR_GFP_5'<BR> | ||
| + | F_P_Gal1_LDLR_GFP_3'<BR> | ||
| + | |||
| + | =='''10/8'''== | ||
| + | 1. PCR of Gal1+LDLR<BR> | ||
| + | P1-7D seq5'<BR> | ||
| + | P1-7D seq3'<BR> | ||
| + | (Pfu ULtraII)<BR> | ||
| + | |||
| + | =='''10/10'''== | ||
| + | *YCplacIII quick change<BR> | ||
| + | PCR<BR> | ||
| + | YCplacIII5'<BR> | ||
| + | YCplacIII3' <BR> | ||
| + | =='''10/11'''== | ||
| + | *PCR<BR> | ||
| + | template:HEP-LDLR cDNA<BR> | ||
| + | primer:<BR> | ||
| + | F_S_Gal1_LDLR_5'new<BR> | ||
| + | 3'-GFP<BR> | ||
| + | *[[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]]<BR> | ||
| + | LDLR5<BR> | ||
| + | LDLR3<BR> | ||
| + | |||
| + | =='''10/12'''== | ||
| + | 1. sequencing<BR> | ||
| + | *LDLR<BR> | ||
| + | *P2,10F<BR> | ||
| + | *YCplac111<BR> | ||
| + | *Gal1<BR> | ||
| + | |||
| + | 2. PCR LDLR from HEP<BR> | ||
| + | |||
| + | =='''10/13'''== | ||
| + | 1. PCR<BR> | ||
| + | *LDLR + GFP<BR> | ||
| + | *LDLR<BR> | ||
| + | |||
| + | =='''10/15'''== | ||
| + | 1. [[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]]<BR> | ||
| + | *LDLR<BR> | ||
| + | |||
| + | =='''10/16'''== | ||
| + | 1. PCR<BR> | ||
| + | *LDLR<BR> | ||
| + | *YCplac111<BR> | ||
| + | |||
| + | 2. Restriction Enzyme digestion<BR> | ||
| + | *YCplac111 with E,P<BR> | ||
| + | *YCplac111 with X,P<BR> | ||
| + | |||
| + | 3. Colony PCR<BR> | ||
| + | *LDLR + GFP<BR> | ||
| + | *LDLR + P1,7D<BR> | ||
| + | |||
| + | =='''10/18'''== | ||
| + | 1. [[Team:Todai-Tokyo/Protocols/Sequencing|Sequencing]]<BR> | ||
| + | *LDLR<BR> | ||
| + | |||
| + | |||
| + | 2. PCR of LDLR<BR> | ||
| + | tested the following conditions;<BR> | ||
| + | *1. DMSO 10%, 60ºC(for annealing)<BR> | ||
| + | *2. DMSO 15%, 55ºC<BR> | ||
| + | *3. DMSO 15%, 60ºC<BR> | ||
| + | *4. DMSO 20%, 55ºC<BR> | ||
| + | *5. DMSO 20%, 60ºC<BR> | ||
| + | *6. DMSO 10%, 58ºC<BR> | ||
| + | |||
| + | 3. Ligation<BR> | ||
| + | *YcplacIII + LDLR<BR> | ||
| + | |||
| + | |||
| + | 4. PCR of LDLR<BR> | ||
| + | tested the following conditions;<BR> | ||
| + | *1. DMSO 10%, 60ºC(for annealing)<BR> | ||
| + | *2. DMSO 15%, 55ºC<BR> | ||
| + | *3. DMSO 15%, 60ºC<BR> | ||
| + | *4. DMSO 20%, 55ºC<BR> | ||
| + | *5. DMSO 20%, 60ºC<BR> | ||
| + | *6. DMSO 10%, 58ºC<BR> | ||
| + | |||
| + | 5. PCR of LDLR | ||
| + | tested the following conditions;<BR> | ||
| + | *1. DMSO 10%, 60ºC(for annealing)<BR> | ||
| + | *2. DMSO 15%, 55ºC<BR> | ||
| + | *3. DMSO 15%, 60ºC<BR> | ||
| + | *4. DMSO 20%, 55ºC<BR> | ||
| + | *5. DMSO 20%, 60ºC<BR> | ||
| + | *6. DMSO 10%, 58ºC<BR> | ||
| + | *7. 40ul system, DMSO 10%, different primer (YcplacIII)<BR> | ||
| + | |||
| + | 6. Gel electrophoresis<BR> | ||
| + | *lane 2~15; LDLR<BR> | ||
| + | |||
| + | 7. sequencing<BR> | ||
| + | *LDLR+P1 7D<BR> | ||
| + | |||
| + | 8. Ligation<BR> | ||
| + | *LDLR and YcplacIII<BR> | ||
| + | |||
| + | =='''10/19'''== | ||
| + | colony PCR<BR> | ||
| + | *YcplacIII+LDLR<BR> | ||
| + | |||
| + | colony PCR (again)<BR> | ||
| + | *YcplacIII+LDLR<BR> | ||
Latest revision as of 03:53, 22 October 2009
| Home | The Team | The Project | Parts Submitted to the Registry | Modeling | Notebook | Protocols | Ethics |
|---|
Contents |
Plan
Aim: Create yeast cells that express LDLR on their cell membrane
Methods:
- Clone the LDLR gene.
- Create biobrick of LDLR.
July
We got LDLR cDNA and tried cloning.
Both ends of LDLR contains a lot of GC,so we had hard time...
7/6
Overview:
1.PCR LDLR gene from plasmid containing LDLR cDNA
2.gel-purify DNA from the PCR product
3.insert the DNA to vector (iGEM parts)
4.Transform into E. coli and select for ampicillin resistance
5.check for Transformation success using colony PCR by LDLR primers
Constructs to be created:
pGal1-Kozak-LDLR-terminator
Obtaining DNA:
Resuspended DNA in the following wells with 10ul water:
plate 1 7D
pGal1(including Kozak sequence)
[http://partsregistry.org/wiki/index.php?title=Part:BBa_J63006 Gal1 promoter]
Transformed 1ul of each of the above into DH5a competent cells:
- Mix 1ul of DNA with 100ul of competent cells on ice.
- Leave on ice for 30 minutes.
- Heat shock at 42ºC for 45 seconds.
- Leave on ice for 2 minutes.
- Add 500ul of LB and incubate at 37ºC for 1 hour.
- Plate on LB-ampicillin plates.
7/7
Miniprep of E.coli cells containing LDLR gene with Promega, Wizard Plus SVMiniprep DNA Purification System
Successful
7/19
PCR of LDLR
0.4ul 50uM 5’primer
0.4ul 50uM 3’primer
1.6ul 2.5mM dNTP
2ul 10×Pfu Ultra 2 buffer
0.05ul LDLR plasmid
0.5ul Pfu Ultra
15.05ul MilliQ water
Performed PCR using the following program:
1. 95ºC 2min
2. 95ºC 30sec
3. 55ºC 30sec
4. 72.5ºC 60sec
5. repeat 2-4 29times
6. 25ºC forever
PCR successful
We cut the region of LDLR out of the gel and purified PCR product using Promega kit.
Infusion of LDLR
4ul PCR product (12.35ug/ml)
3ul vector (iGEM parts plate1 D-3, 14.95ug/ml)
2ul 5×infusion reaction buffer
1ul infusion enzyme
|
v
37ºC 15min
50ºC 15min
|
v
TE buffer up to 20ul
|
v
added 10ul of the sample to DH5α (090614) and put on ice for 15 min.
|
v
42ºC 45sec
|
v
added 500ul LB broth to the tube.
|
v
placed it on LB ampicilin plate.
7/20
colony PCR
put a small amount of single colony into each tube with 5ul MilliQ water
|
v
95ºC 5min
|
v
PCR reaction
1ul 10×buffer
0.8ul 2.5mMdNTP
0.08ul Ex-Taq
0.1ul 5’-primer
0.1ul 3’-primer
2.92ul MilliQ water
|
v
added the PCR reaction to each tube.
|
v
Performed PCR using the following program:
1. 95ºC 2min
2. 95ºC 30sec
3. 55ºC 30sec
4. 72.5ºC 120sec
5. repeat 2-4 29 times
6. 25ºC forever
PCR unsuccessful・・・.
7/25
gal1 Sequencing by BIG DYE
1.8ul 5xB.D.3.1.buffer
0.4ul B.D.3.1.
6.3ul MilliQ water
1ul plasmid(0.15ug/ul)
0.5ul 5'or3'primer(3.2pmol/ul)
|
v
PCR Program
1.96ºC 2min
2.96ºC 10sec
3.55ºC 5sec
4.60ºC 3min
5.go to 2.29times
6.25ºC forever
|
v
dd 0.5ul PHOSPHATASE ALKALINE shrimp
|
v
37ºC 1hr incubate
|
v
add 1ul 3MNaOAc
|
v
add 25ul EtOH
|
v
20000xg 4ºC 10min centrifugation
|
v
put off supernatant
|
v
dry tubes
|
v
add 15ul HiDi
|
v
put them in the sequence machine
7/26
colony PCR again, using 7/20 protocol.
PCR program
1.95ºC 2min
2.95ºC 30sec
3.55ºC 30sec
4.72.5ºC 150sec
5.go to 2. 29times
but,unsucessful.
7/27
Miniprep of E.coli cells containing LDLR gene(that yesterday picked up and cultured) with Promega, Wizard Plus SV Miniprep DNA Purification System
using AGE, found a colony that has LDLR infusion plasmid
7/30
- sequencing of LDLR
August
We found our plasmid that we got first didn't include LDLR cDNA!? We came back to start line again!?
8/1
PCR of GFP for fusion
program
1.95ºC 2min
2.95ºC 30sec
3.55ºC 30sec
4.72.5ºC 20sec
5.go to 2. 29 times
6.25ºC forever
We found that the length of these PCR product till now was longer than that of LDLR.
->the result didn't conform with NCBI datebase
- change PCR program→didn't get the PCR product whose length was correct
8/2
- Sequencing of LDLR
8/4
The following were sequenced using the labeled primers:
- 1)F-spe1-Gall p-LDLR-5
- 2)F-pst1-Gall p-LDLR-3
Results:
Sequence read failed
8/5
- Sequencing of LDLR
8/6
LDLR1 and LDLR2 is combination of Gal1, LDLR and GFP.
8/7
- PCR of LDLR
using the labeled primers:
- 1)F-spe1-Gal1-LDLR-5'
- 2)F-spe1-Gal1-LDLR-3'
- PCR of GFP
using the labeled primers:
- 1)F-spe1-Gal1-LDLR-GFP-5'
- 2)F-pst1-Gal1-LDLR-GFP-3'
8/8
- Transformation of P3 21D
- Transformation of P1 23L
- PCR of LDLR
using the labeled primers:
- 1)F-spe1-Gal1-LDLR-5'
- 2)F-spe1-Gal1-LDLR-3'
using the following program;
program
1.95ºC 2min
2.95ºC 30sec
3.52~55ºC 30sec
4.72.5ºC 60sec
5.go to 2. 29 times
6.25ºC forever
Result;
The length of LDLR is about 2583 bp.
- PCR of LDLR
using the labeled primers:
- 1)F-spe1-Gal1-LDLR-5'
- 2)F-spe1-Gal1-LDLR-3'
using the following program;
program
1.95ºC 2min
2.95ºC 30sec
3.57ºC 30sec
4.72.5ºC 60sec
5.go to 2. 29 times
6.25ºC forever
The change point of this program is the temperature at third step.
This temperature is 57ºC but previous is 52~55ºC.
result;
The length of LDLR is 2500~3000bp.
8/9
- PCR of LDLR
8/10
- Sequencing of LDLR
The following were sequenced using the labeled primers
1)1D-LDLR-5'
2)1D-LDLR-3'
Results:
Sequence read failed
- PCR of LDLR(1st time)
using the labeled primers:
1)F-spe1-Gal1-LDLR-GFP-5'
2)F-spe1-Gal1-LDLR-GFP-3'
using the following program;
program
1.95ºC 2min
2.95ºC 30sec
3.55ºC 30sec
4.72.5ºC 60sec
5.go to 2. 29 times
6.25ºC forever
Result;
PCR is sucessful.
- PCR of LDLR(2nd time)
using the labeled primers is same as 1st time's:
using the following program;
program
1.95ºC 2min
2.96ºC 30sec
3.55ºC 30sec
4.60.0ºC 3min
5.go to 2. 29 times
6.25ºC forever
8/12
- Sequencing of LDLR
Result;
NNNNN can't read.
- electrophorese LDLR+GFP
RESULT:the band across 3000-3500bp not found. -> need to rePCR LDLR+GFP.
8/13
- measure the absorbance of LDLR2cDNA
->534ug/mL
+PCR
objects:LDLR2, yqiT, GFP
program
1.95ºC 2min
2.96ºC 30sec
3.55ºC 30sec
4.60.0ºC 3min
5.go to 2. 29 times
6.25ºC forever
- electrophorese the above 3
result -> other than GFP not found <- maybe DNAs dissolved by UV or contamination;
8/14
objects: LDLR GFP
LDLR
primer
F_S_GalI_LDLR_5'
F_PstI_GalI_LDLR_3'
GFP
primer
F_S_GalI_LDLR_GFP 5'
F_P_GalI_LDLR_GFP 3'
- electrophorese PCR products
->failed (LDLR+GFP)
8/15
rePCR LDLR+GFP electrophorese LDLR+GFP
8/19
1 Colony PCR of LDLR
Using the labeled primers:
1) J63010(P17D)_seq_5’
2) J63010(P17D)_seq_3’
Result:
successful
2 PCR of LDLR for fusion
Using the labeled primers:
1) F_S_Gal1_LDLR_5’new
2)F_LDLR_3’new
3 PCR of GFP for fusion
Using the labeled primers:
1) F_S_Gal1_LDLR_GFP_5’
2) F_S_Gsl1_LDLR_GFP_3’
8/21
1 PCR of LDLR
Using the labeled primers:
1) F_S_Gal1_LDLR_5’new
2)F_LDLR_3’
2 Infusion of LDLR and GFP
8/22
1 Miniprep ofLDLR
8/23
1 Sequencing of LDLR
Using the labeled primers:
1) J_P17D_5’
2) J_P17D_3’
8/25
1 PCR of LDLR
Using the labeled primers:
1) F_S_Gp_LDLR_out_5’
2) F_P_Gp_LDLR_out_3’
8/26
1 PCR of LDLR
Using the labeled primers:
1) F_S_Gp_LDLR_out_5’
2) F_P_Gp_LDLR_out_3’
2 Sequencing of LDLR cDNA
Using the labeled primers:
1) F_S_Gp_LDLR_out_5’
2)F_P_Gp_LDLR_out_3’
Result : failed
8/27
1 PCR of LDLR
Using the labeled primers:
1) F_S_Gp_LDLR_out_5’
2) F_P_Gp_LDLR_out_3'
2 Sequencing of LDLR cDNA
Using the labeled primers:
1) F_S_Gp_LDLR_out_5’
2)F_P_Gp_LDLR_out_3’
Result : failed
8/30
- PCR of LDLR
using the labeled primers:
1)F-spe1-Gal1-LDLR-GFP-5'
2)F-spe1-Gal1-LDLR-GFP-3'
8/31
Sequencing of LDLR
The following were sequenced using the labeled primers
1)1D-LDLR-5'
2)1D-LDLR-3'
Results:
Sequence read failed
September
We got new LDLR cDNA!
- look for the best PCR program
→DMSO 5% or 10%
→annealing temparature 55ºC or 60ºC
result
the best program
10% DMSO
1.95ºC 2min
2.95ºC 30sec
3.60ºC 30sec
4.72.5ºC 1min
5.go to 2. 29 times
6.25ºC forever
purify the PCR product using Promega kit and insert it in iGEM part(plate1-7D:Gal1 promoter)
And, insert it and GFP in iGEM part(plate1-7D:Gal1 promoter)
9/15
1. PCR + Gel Extraction
- LDLR
2. PCR
- LDLR (annealing temperature:57)
- LDLR (annealing temperature:60)
3. PCR
- LDLR: tested different concentrations of template (do not contain DMSO)
- 1. X 1/10
- 2. X 1/5
- 3. X 1 (normal)
- 4. X 5
- 5. X 10
9/16
1. PCR
- LDLR: tested different concentrations of template
- 1. X 1/10 + 20%DMSO
- 2. X 1/5 + 20%DMSO
- 3. X 1 (normal) + 20%DMSO
- 4. X 5 + 20%DMSO
- 5. X 10 + 20%DMSO
2. PCR
- LDLR: tested different conditions
- 1. cDNA1, old primer, 20% DMSO
- 2. cDNA1, old primer, no DMSO
- 3. cDNA1, new primer, 20% DMSO
- 4. cDNA1, new primer, no DMSO
- 5. cDNA2, old primer, 20% DMSO
- 6. cDNA2, old primer, no DMSO
- 7. cDNA2, new primer, 20% DMSO
- 8. cDNA2, new primer, no DMSO
- 9. control
9/24
1. PCR
- GEP
- YCplac111
9/25
- Miniprep of E.coli cells containing GFP gene (those yesterday picked up and cultured) with Promega, Wizard Plus SV Miniprep DNA Purification System
- read the Sequencing of LDLR cDNA
Results:
Sequence read failed
- PCR of LDLR cDNA(Pfu UltraII)
Results:
PCR failed
9/26
- PCR of LDLR for infusion
Using the labeled primers:
1) F_S_Gal1_LDLR_5’new
2)F_pstI_Gal1p_LDLR_3’new
Results:
PCR failed
- PCR of LDLR for fusion
Using the labeled primers:
1) F_S_Gal1_LDLR_5’new
2)F_LDLR_3’new
Results:
PCR failed
- PCR of GFP for fusion
Using the labeled primers:
1) F_S_Gal1_LDLR_GFP_5’
2) F_p_Gal1_LDLR_GFP_3’
Results:
PCR failed
9/27
- PCR of LDLR
Using the labeled primers:
1) F_S_Gp_LDLRout_5'
2)F_P_Gp_LDLRout_3'
Results:
PCR failed
- PCR of LDLR
Using the labeled primers:
1) F_S_Gal1_LDLR_5'new
2)F_P_Gal1p_LDLR_3'
Results:
PCR failed
- PCR of LDLR
Using the labeled primers:
1) F_S_Gal1_LDLR_5'new
2)F_LDLR_3'
Results:
PCR failed
- read the Sequencing of LDLR cDNA
Results:
Sequence read failed
9/28
- PCR of BC LDLR
primers:
F_S_Gal1_LDLR_5'new
F_pst1_Gal1p_LDLR_3'
- again, PCR of LFL, HEP and BC LDLR
primer:
1)F_S_Gp_LDLR_out_5'
1)F_P_Gp_LDLR_out_3'
2)F_S_Gal1p_LDLR_5'new
2)F_pst1_Gal1p_LDLR_3'
3)F_S_Gal1_LDLR_5'new
3)F_LDLR_3'primer
9/29
- sequencing of YCplaC111
- infusion of LDLR
vector:P17D
9/30
- colony PCR of LDLR and P17D+LDLR and P17D+LDLR+GFP
primer:
F_S_Gal1_LDLR_5'new
F_Pst1_Gal1p_LDLR_3'
- again, colony PCR of P17D+LDLR+GFP
primer:
F_S_Gal1_LDLR_5'new
F_Pst1_Gal1p_LDLR_3'
October
LDLR made a debut as an iGEM part!
That was very long way...
10/4
- PCR of GFP for LDLR primer
primer:
F_S_Gal1_LDLR_GFP_5'
F_P_Gal1_LDLR_GRP_3'
- colony PCR of LDLR+GRP
primer:
P17Dseq_5'
P17Dseq_3'
- infusion of LDLF+GFP+P17D
vector:P17D digested with S and P
10/5
- check of plasmids including LDLR by AGE
1.LDLR3 091004
2.LDLR5 091004
3.LDLR+GFP 1 091004
4.LDLR+GFP 2 091004
1&2->OK,3&4->wouldn't have LDLR+GFP
1.LDLR3 091004
2.LDLR5 091004
3.LDLR+GFP 1 091004
4.LDLR+GFP 2 091004
5~10.YCplacIII(whose restriction site may be changed)
primer
1~4:P1-7Dseq.5'or3'
5~10:YCplacIII seq. primer5'or3'
- PCR for LDLR+GFP fusion(Pfu UltraII)
template:HEP-LDLR cDNA
primer:
F_S_Gal1_LDLR_5'new
F_P_GAl1_LDLR_GFP_5'
10/7
- PCR for GFP fusion(Pfu UltraII)
primer
F_S_Gal1_LDLR_GFP_5'
F_P_Gal1_LDLR_GFP_3'
10/8
1. PCR of Gal1+LDLR
P1-7D seq5'
P1-7D seq3'
(Pfu ULtraII)
10/10
- YCplacIII quick change
PCR
YCplacIII5'
YCplacIII3'
10/11
- PCR
template:HEP-LDLR cDNA
primer:
F_S_Gal1_LDLR_5'new
3'-GFP
LDLR5
LDLR3
10/12
1. sequencing
- LDLR
- P2,10F
- YCplac111
- Gal1
2. PCR LDLR from HEP
10/13
1. PCR
- LDLR + GFP
- LDLR
10/15
1. Sequencing
- LDLR
10/16
1. PCR
- LDLR
- YCplac111
2. Restriction Enzyme digestion
- YCplac111 with E,P
- YCplac111 with X,P
3. Colony PCR
- LDLR + GFP
- LDLR + P1,7D
10/18
1. Sequencing
- LDLR
2. PCR of LDLR
tested the following conditions;
- 1. DMSO 10%, 60ºC(for annealing)
- 2. DMSO 15%, 55ºC
- 3. DMSO 15%, 60ºC
- 4. DMSO 20%, 55ºC
- 5. DMSO 20%, 60ºC
- 6. DMSO 10%, 58ºC
3. Ligation
- YcplacIII + LDLR
4. PCR of LDLR
tested the following conditions;
- 1. DMSO 10%, 60ºC(for annealing)
- 2. DMSO 15%, 55ºC
- 3. DMSO 15%, 60ºC
- 4. DMSO 20%, 55ºC
- 5. DMSO 20%, 60ºC
- 6. DMSO 10%, 58ºC
5. PCR of LDLR
tested the following conditions;
- 1. DMSO 10%, 60ºC(for annealing)
- 2. DMSO 15%, 55ºC
- 3. DMSO 15%, 60ºC
- 4. DMSO 20%, 55ºC
- 5. DMSO 20%, 60ºC
- 6. DMSO 10%, 58ºC
- 7. 40ul system, DMSO 10%, different primer (YcplacIII)
6. Gel electrophoresis
- lane 2~15; LDLR
7. sequencing
- LDLR+P1 7D
8. Ligation
- LDLR and YcplacIII
10/19
colony PCR
- YcplacIII+LDLR
colony PCR (again)
- YcplacIII+LDLR
| Home | The Team | The Project | Parts Submitted to the Registry | Modeling | Notebook | Protocols | Ethics |
|---|
 "
"