Team:Valencia/WetLab/YeastTeam/Results
From 2009.igem.org
| Line 9: | Line 9: | ||
| - | <div style="position:relative; top:- | + | <div style="position:relative; top:-285px; left:10px; width:700px; font-family: Verdana;" align="justify"> |
<br> | <br> | ||
==Experimental results== | ==Experimental results== | ||
Revision as of 15:41, 21 October 2009
Experimental results
Our ultimate goal was to make a bio-screen made with cellular pixels (LECs). But, before to be able to build this iLCD, we had to study the behaviour of one single LEC. Therefore, we focused on the electrical excitation of our transformed yeasts.
Taking advantage of the different experimental designs we had at a glance: we had several calcium channel knock outs (mid1 and ch1), as well as functional inhibitors of the calcium channels (KCl) and divalent ion quelant (EDTA).
characterize the cell light emission if we wanted to control it better. We thought about two possible ways to make cells produce light: First, the producion of light with a chemical imput and, second, making the cells glow with electricity. we choose the calcium signaling because it is the fastest known modality of signaling in biology, and will allow for a fast refreshing rate of the screen.
Chemical input
In order to make our yeasts produce light, we firstly reproduce experiments made by Viladevall et al, after a lot of different trials, we finally could characterize the luminiscence curve in a discontinuos luminometer.
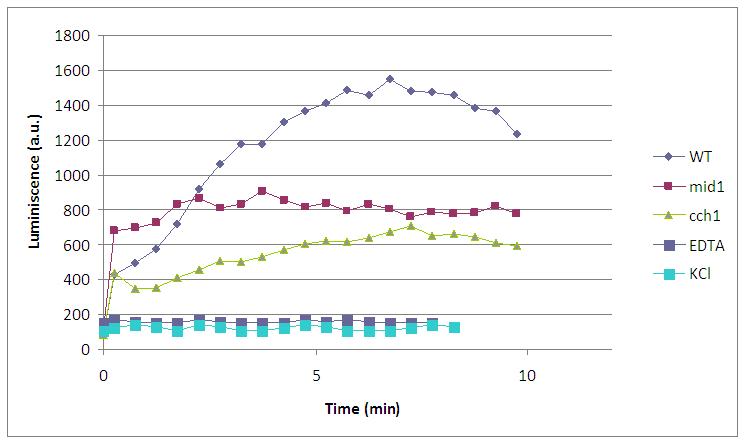
As we can see in the graph, a peak of light is emited about 450 seconds after the addition 60 microliters of KOH to 170 microliters of medium with WT transformed yeasts. Although we were almost sure that the mechanism that triggered that flash of light was the expected, we preferred to make the same experiment with different kind of controls and make sure we were not observing any artiffact:
- Mid1: one knock out mutant for a Calcium channel. Light is not observed because Ca2+ can’t enter into the cell and bind to the aequorin-coelenterazine complex.
- Cch1: another knock out mutant for Calcium channel, so the absence of light can be explainned in the same way.
- EDTA: Although every compound necessary for the reaction is present (including Ca2+ channels) light is not emited because EDTA is a divalent ion quelant, so Ca2+ is quenched and not useful for the emission.
- KCl: another negative control. The absence of the -OH group prevents the opening of calcium channels and makes yeast produce no light.
We wanted to characterize in detail this kind of response.
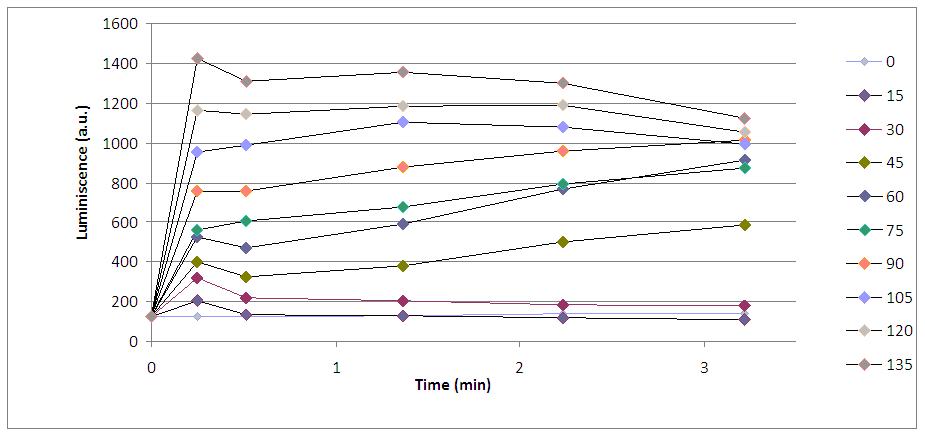
To complete the work with the chemical input, we though KOH amounts could influence in the quantity of emitted light, so we repited the experiment with different concentrations of KOH.
As we can see, the volume of KOH added (from 15 microliters to 120) is related to the luminiscent peak. Although there is not linearly proportional, luminiscence intensity is increased when we increase the quantity of KOH we put in the sample (always 170 microliters of medium with yeasts).
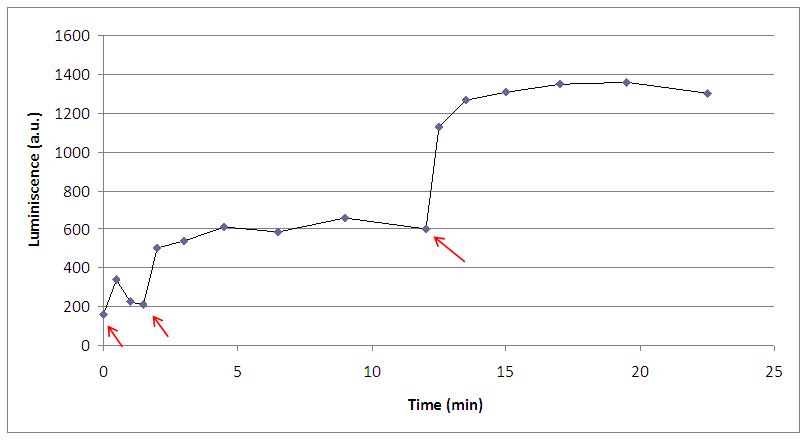
Characterising the response to the KOH we also found interesting to determinate the reproducibility of the process.
By adding 30 microliters of KOH at certain times (arrows), we discovered that after the first peak, cells couldn’t return to the basals levels, and every new shock make yeasts produce light in higher levels than the last one.
Electrical input
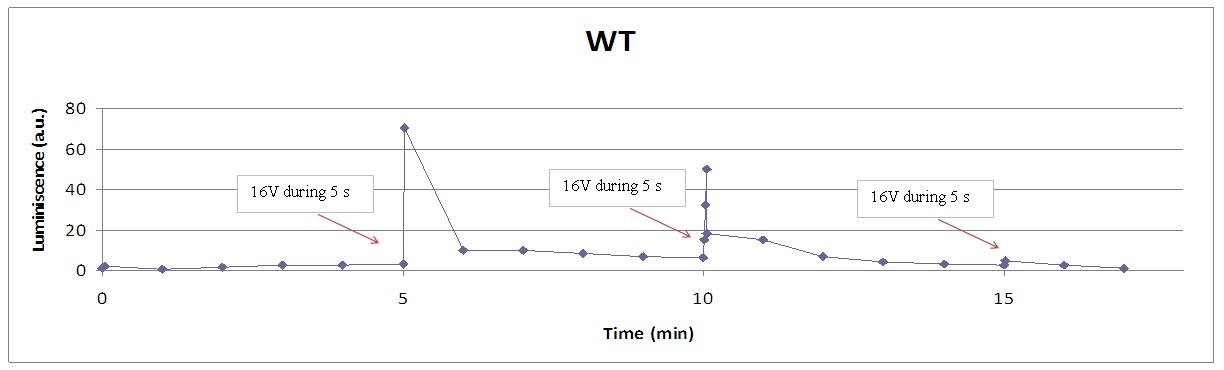
When the experiments with an alkali input showed us that yeasts were able to produce light because of their transformation, we tried our ambitious goal: stimulate calcium channels with an electrical input.
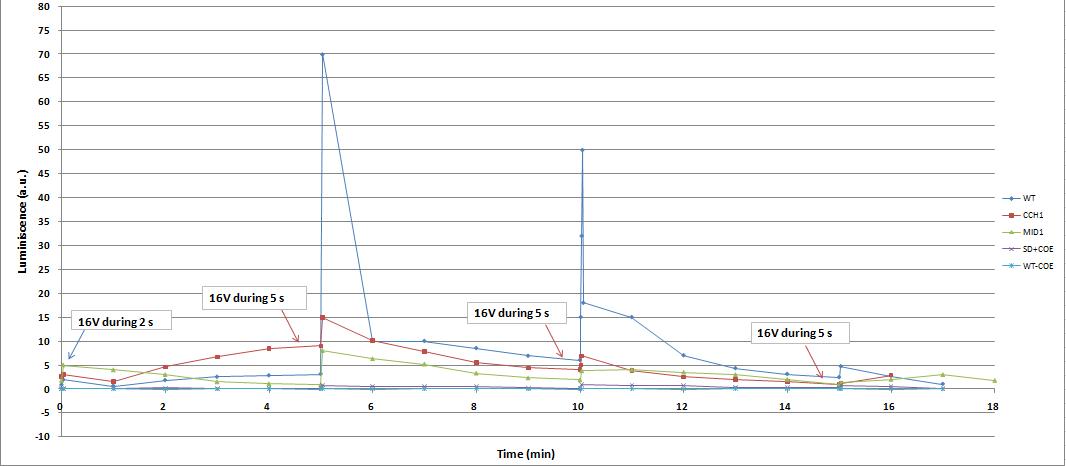
We use two luminometers, one luminometer discontinuous and the other is continuous. Each luminometer uses different units, depends on the manufacturer. For this reasons we can't compare directly the results obtained with one luminometer with the results of the other. According this, when we only compare results in the same graph if they were obtained with the same luminometer. However, an increase (or not) in the luminosity, means the same at two luminometers and the experiments are complementary and reaffirms our conclusions.
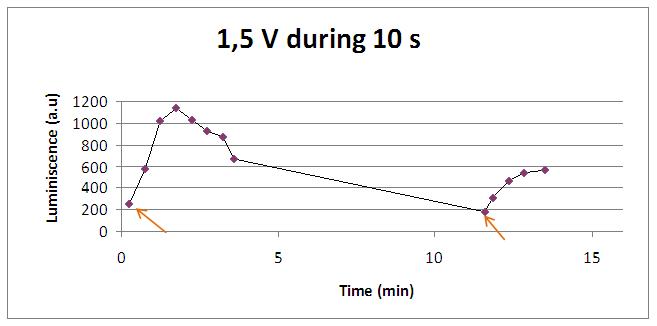
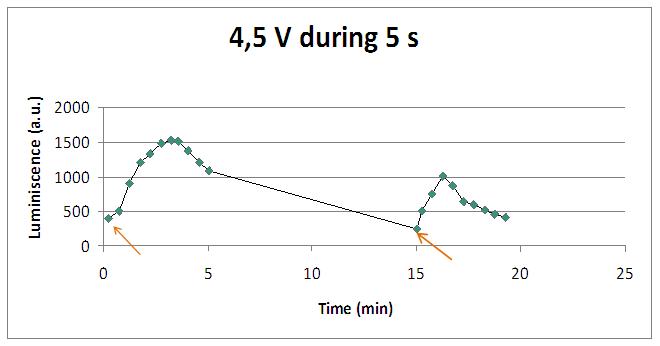
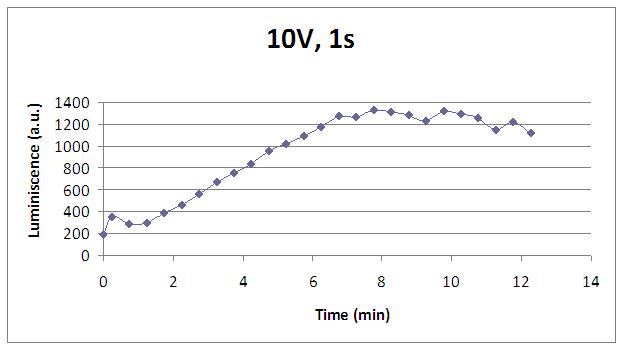
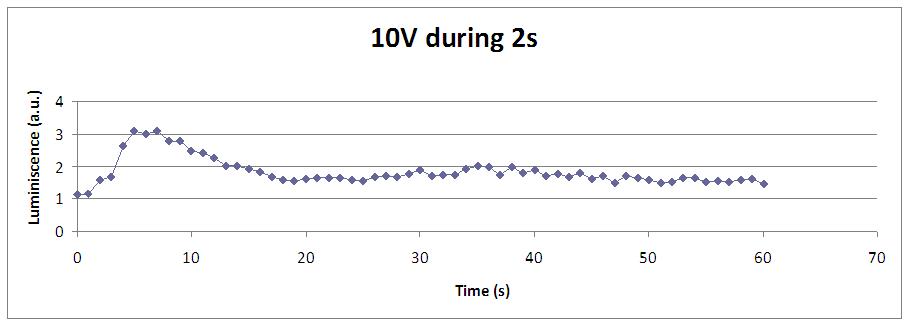
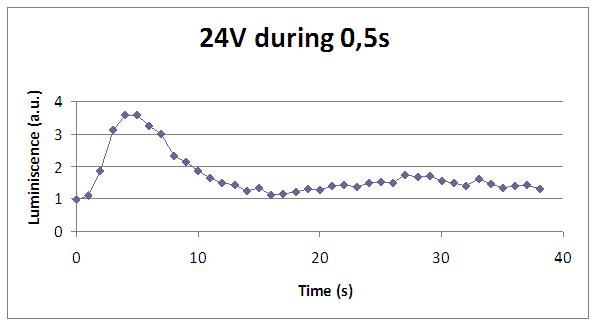
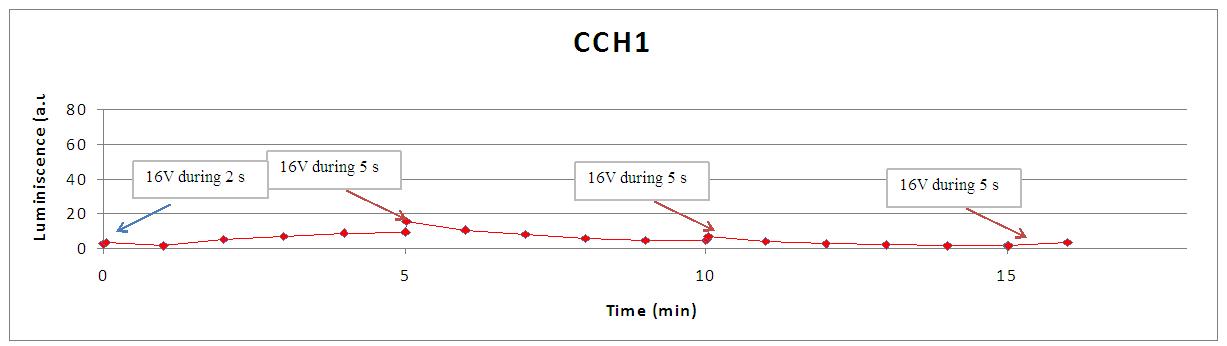
We reproduced the mentionated Viladevall et al's protocol, incubating the transformed yeasts with coelenterazine, but changing the KOH by electricity. Surprisingly, we found that light was also produced in a very similar way. We tried with different times and voltages in order to find the optim conditions for a big peak of light. Some of our graphics are theese:
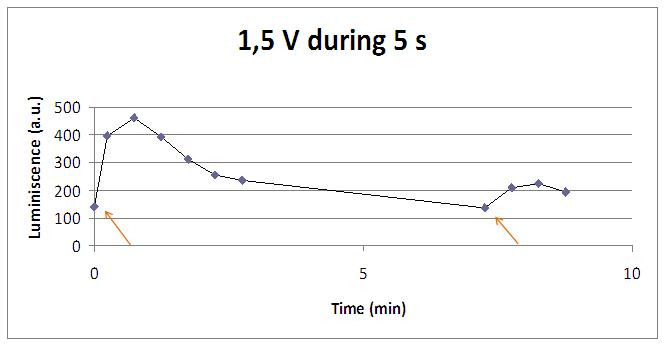
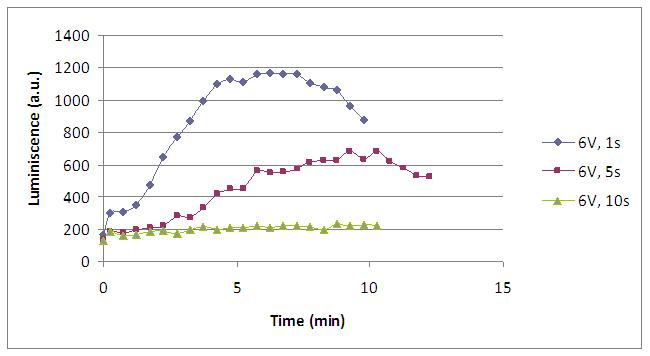
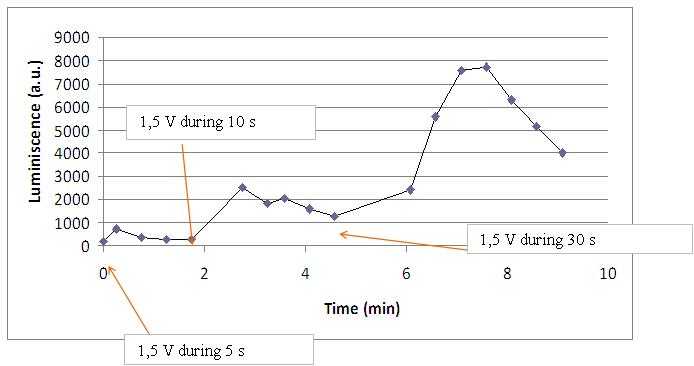
We realised that the time of exposure to the electrical stimulus was crucial, even more that the aplied voltage. That means, if we increased the voltage at very short times, cells could produce a more abrupt peak of light. But if we increased the time of exposure to the electricity, we observe a less defined response, with more flattened peaks.
That’s probably because a big exposure time of electrical input damages and killes the yeasts, making them to release their components to the medium, including the aequorin-coelenterazine-Ca2+ complex, so the emission of light is more uniform in time, instead of the production of the flash produced by the Calcium enetering in the cell.
In the case of very little voltages (like 1,5V) this observation is not carried out by our yeasts. The reason must be that the electrical input is too low, so yeasts don’t die so easily as with more elevated voltage, and a better response is produce with a more prolongated electrical shock.
This graph clearly show us that using a same voltage, we obtain a better response with the shortest time of the electrical input.
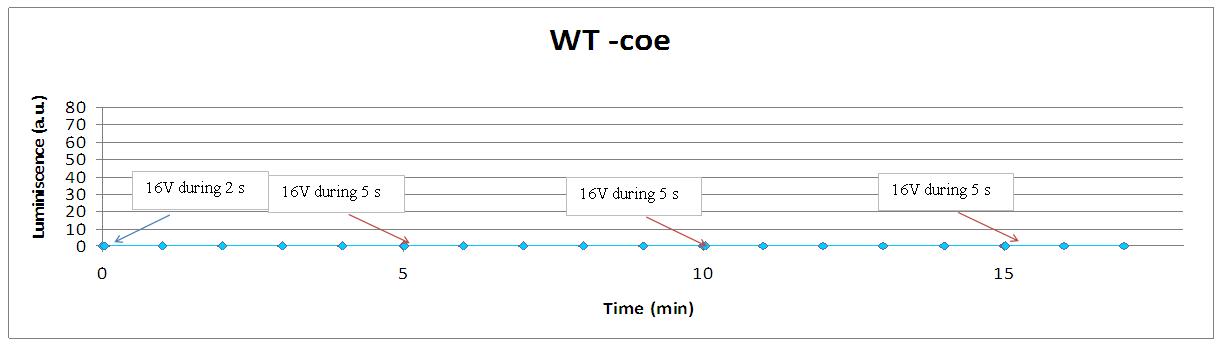
Our controls discard the idea of an artifact. For example, light could be made by a spark produced during the discharge. It was not very probable, because the peak observed was produce near 400 seconds after the stimulus. But, another time, when cells without coelenterazine or mutants are used, we see no light.
Studying the repetibility of the process, this is a little different from the chemical stimulus, but the system has a similar behaviour, and we can stimulate several times the same sample getting a response. However, every next shock produces a fewer peak of light. We hace two hypothesis: one of them is that a part of our yeasts die meanwhile the electrical stimulus. The other one is that coelenterazine is not reusable, so a proportion of it runs down in every emission of light.
 "
"