Team:Lethbridge/Project
From 2009.igem.org
(→Motility Assay) |
(→Electron microscopy) |
||
| Line 542: | Line 542: | ||
The Magnetite nanoparticle producing construct, pLacI:sRBS:mms6:dT, is expected to produce uniform nanoparticles. In order to visualize the size and shape of the nanoparticles we intend to use electron microscopy. With the aid of the electron microscope it is possible to visualize particles on the nanoscale. For this we will need to overexpress the protein (see gene expression) with IPTG. The magnetite, Fe3O4, nanoparticles form in the presence of FeCl2. Varying concentrations of FeCl2 will be added to the cells. After 24 hours, the cells will be opened up using chemical, mechanical and chemical/mechanical techniques, in order to determine if this poses any significant difference on the nanoparticles themselves. The nanoparticles are insoluble, and therefore can be filtered off from the cell lysate and washed. There are different types of filters which can be used to collect nanoparticles, we will use thin polycarbonate membrane as our filter of choice since the nanoparticles which we expect to obtain are on the smaller scale (6-20 nm in diameter). We will then place the sample under the electron microscope to visualize our magnetite nanoparticles. | The Magnetite nanoparticle producing construct, pLacI:sRBS:mms6:dT, is expected to produce uniform nanoparticles. In order to visualize the size and shape of the nanoparticles we intend to use electron microscopy. With the aid of the electron microscope it is possible to visualize particles on the nanoscale. For this we will need to overexpress the protein (see gene expression) with IPTG. The magnetite, Fe3O4, nanoparticles form in the presence of FeCl2. Varying concentrations of FeCl2 will be added to the cells. After 24 hours, the cells will be opened up using chemical, mechanical and chemical/mechanical techniques, in order to determine if this poses any significant difference on the nanoparticles themselves. The nanoparticles are insoluble, and therefore can be filtered off from the cell lysate and washed. There are different types of filters which can be used to collect nanoparticles, we will use thin polycarbonate membrane as our filter of choice since the nanoparticles which we expect to obtain are on the smaller scale (6-20 nm in diameter). We will then place the sample under the electron microscope to visualize our magnetite nanoparticles. | ||
| + | |||
| + | <html> | ||
| + | <head> | ||
| + | |||
| + | <!--%%%%%%%%%%%% QuickMenu Styles [Keep in head for full validation!] %%%%%%%%%%%--> | ||
| + | <style type="text/css"> | ||
| + | |||
| + | |||
| + | /*!!!!!!!!!!! QuickMenu Core CSS [Do Not Modify!] !!!!!!!!!!!!!*/ | ||
| + | .qmmc .qmdivider{display:block;font-size:1px;border-width:0px;border-style:solid;position:relative;z-index:1;}.qmmc .qmdividery{float:left;width:0px;}.qmmc .qmtitle{display:block;cursor:default;white-space:nowrap;position:relative;z-index:1;}.qmclear {font-size:1px;height:0px;width:0px;clear:left;line-height:0px;display:block;float:none !important;}.qmmc {position:relative;zoom:1;z-index:10;}.qmmc a, .qmmc li {float:left;display:block;white-space:nowrap;position:relative;z-index:1;}.qmmc div a, .qmmc ul a, .qmmc ul li {float:none;}.qmsh div a {float:left;}.qmmc div{visibility:hidden;position:absolute;}.qmmc .qmcbox{cursor:default;display:block;position:relative;z-index:1;}.qmmc .qmcbox a{display:inline;}.qmmc .qmcbox div{float:none;position:static;visibility:inherit;left:auto;}.qmmc li {z-index:auto;}.qmmc ul {left:-10000px;position:absolute;z-index:10;}.qmmc, .qmmc ul {list-style:none;padding:0px;margin:0px;}.qmmc li a {float:none;}.qmmc li:hover>ul{left:auto;}#qm0 ul {top:100%;}#qm0 ul li:hover>ul{top:0px;left:100%;} | ||
| + | |||
| + | |||
| + | /*!!!!!!!!!!! QuickMenu Styles [Please Modify!] !!!!!!!!!!!*/ | ||
| + | |||
| + | |||
| + | /* QuickMenu 0 */ | ||
| + | |||
| + | /*"""""""" (MAIN) Container""""""""*/ | ||
| + | #qm0 | ||
| + | { | ||
| + | width:auto; | ||
| + | background-color:transparent; | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" (MAIN) Items""""""""*/ | ||
| + | #qm0 a | ||
| + | { | ||
| + | padding:5px 60px 3px 0px; | ||
| + | margin:0px 5px 0px 0px; | ||
| + | color:#FFD600; | ||
| + | font-family:Arial; | ||
| + | font-size:12px; | ||
| + | text-decoration:none; | ||
| + | font-weight:bold; | ||
| + | border-width:0px 0px 8px 0px; | ||
| + | border-style:solid; | ||
| + | border-color:#FFD600; | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" (MAIN) Hover State""""""""*/ | ||
| + | #qm0 a:hover | ||
| + | { | ||
| + | color:#3C13AF; | ||
| + | border-color:#FFAD00; | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" (MAIN) Hover State - (duplicated for pure CSS)""""""""*/ | ||
| + | #qm0 li:hover>a | ||
| + | { | ||
| + | color:#3C13AF; | ||
| + | border-color:#FFAD00; | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" (MAIN) Parent Items""""""""*/ | ||
| + | #qm0 .qmparent | ||
| + | { | ||
| + | background-repeat:no-repeat; | ||
| + | background-position:95% 50%; | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" (MAIN) Active State""""""""*/ | ||
| + | body #qm0 .qmactive, body #qm0 .qmactive:hover | ||
| + | { | ||
| + | color:#3C13AF; | ||
| + | text-decoration:none; | ||
| + | border-color:#FFAD00; | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" (SUB) Container""""""""*/ | ||
| + | #qm0 div, #qm0 ul | ||
| + | { | ||
| + | padding:10px 5px 10px 5px; | ||
| + | background-color:#EEEEEE; | ||
| + | border-width:1px; | ||
| + | border-style:solid; | ||
| + | border-color:#3C13AF; | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" (SUB) Items""""""""*/ | ||
| + | #qm0 div a, #qm0 ul a | ||
| + | { | ||
| + | padding:3px 5px 3px 5px; | ||
| + | background-color:transparent; | ||
| + | color:#230672; | ||
| + | font-size:11px; | ||
| + | font-weight:normal; | ||
| + | border-width:0px; | ||
| + | border-style:none; | ||
| + | border-color:#230672; | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" (SUB) Hover State""""""""*/ | ||
| + | #qm0 div a:hover | ||
| + | { | ||
| + | background-color:#476DD5; | ||
| + | color:#FFFFFF; | ||
| + | text-decoration:none; | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" (SUB) Hover State - (duplicated for pure CSS)""""""""*/ | ||
| + | #qm0 ul li:hover>a | ||
| + | { | ||
| + | background-color:#476DD5; | ||
| + | color:#FFFFFF; | ||
| + | text-decoration:none; | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" (SUB) Parent Items""""""""*/ | ||
| + | #qm0 div .qmparent, #qm0 ul .qmparent | ||
| + | { | ||
| + | background-image:url(qmimages/arrow_0.gif); | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" (SUB) Active State""""""""*/ | ||
| + | body #qm0 div .qmactive, body #qm0 div .qmactive:hover | ||
| + | { | ||
| + | background-color:#476DD5; | ||
| + | background-image:url(qmimages/arrow_1.gif); | ||
| + | color:#FFFFFF; | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" Individual Titles""""""""*/ | ||
| + | #qm0 .qmtitle | ||
| + | { | ||
| + | margin:2px 5px 5px 5px; | ||
| + | color:#222222; | ||
| + | font-family:Arial; | ||
| + | font-size:11px; | ||
| + | font-weight:bold; | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" Individual Horizontal Dividers""""""""*/ | ||
| + | #qm0 .qmdividerx | ||
| + | { | ||
| + | border-top-width:1px; | ||
| + | margin:4px 5px 4px 5px; | ||
| + | border-color:#999999; | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" Custom Rule""""""""*/ | ||
| + | ul#qm0 ul | ||
| + | { | ||
| + | width:100%; | ||
| + | } | ||
| + | |||
| + | |||
| + | /*"""""""" Custom Rule""""""""*/ | ||
| + | ul#qm0 li:hover > a.qmparent | ||
| + | { | ||
| + | background-image:url(qmimages/arrow_1.gif); | ||
| + | } | ||
| + | |||
| + | |||
| + | </style> | ||
| + | |||
| + | <!-- Add-On Core Code (Remove when not using any add-on's) --> | ||
| + | <style type="text/css">.qmfv{visibility:visible !important;}.qmfh{visibility:hidden !important;}</style><script type="text/javascript">if (!window.qmad){qmad=new Object();qmad.binit="";qmad.bvis="";qmad.bhide="";}</script> | ||
| + | |||
| + | <!-- Add-On Settings --> | ||
| + | <script type="text/JavaScript"> | ||
| + | |||
| + | /******* Menu 0 Add-On Settings *******/ | ||
| + | var a = qmad.qm0 = new Object(); | ||
| + | |||
| + | // Match Widths Add On | ||
| + | a.mwidths_active = true; | ||
| + | |||
| + | // IE Over Select Fix Add On | ||
| + | a.overselects_active = true; | ||
| + | |||
| + | </script> | ||
| + | |||
| + | <!-- Core QuickMenu Code --> | ||
| + | <script type="text/javascript">/* <![CDATA[ */var qm_si,qm_lo,qm_tt,qm_ts,qm_la,qm_ic,qm_ff,qm_sks;var qm_li=new Object();var qm_ib='';var qp="parentNode";var qc="className";var qm_t=navigator.userAgent;var qm_o=qm_t.indexOf("Opera")+1;var qm_s=qm_t.indexOf("afari")+1;var qm_s2=qm_s&&qm_t.indexOf("ersion/2")+1;var qm_s3=qm_s&&qm_t.indexOf("ersion/3")+1;var qm_n=qm_t.indexOf("Netscape")+1;var qm_v=parseFloat(navigator.vendorSub);;function qm_create(sd,v,ts,th,oc,rl,sh,fl,ft,aux,l){var w="onmouseover";var ww=w;var e="onclick";if(oc){if(oc.indexOf("all")+1||(oc=="lev2"&&l>=2)){w=e;ts=0;}if(oc.indexOf("all")+1||oc=="main"){ww=e;th=0;}}if(!l){l=1;sd=document.getElementById("qm"+sd);if(window.qm_pure)sd=qm_pure(sd);sd[w]=function(e){try{qm_kille(e)}catch(e){}};if(oc!="all-always-open")document[ww]=qm_bo;if(oc=="main"){qm_ib+=sd.id;sd[e]=function(event){qm_ic=true;qm_oo(new Object(),qm_la,1);qm_kille(event)};}sd.style.zoom=1;if(sh)x2("qmsh",sd,1);if(!v)sd.ch=1;}else if(sh)sd.ch=1;if(oc)sd.oc=oc;if(sh)sd.sh=1;if(fl)sd.fl=1;if(ft)sd.ft=1;if(rl)sd.rl=1;sd.th=th;sd.style.zIndex=l+""+1;var lsp;var sp=sd.childNodes;for(var i=0;i<sp.length;i++){var b=sp[i];if(b.tagName=="A"){eval("ig(\"qn_tkt&'!xiodpw/qnv7&'(xiodpw/lpcbtjoo+#\"*.uoMoxesCbsf(*.jneeyOg(#hutq:#),1*{c.jnoesHUMM=#Bvy!Npw\"\"<b/hseg=#hutq:0/xwx.ppfnduce/cpm0bvy`npw/atp#;rm`sls>tsuf;~".replace(/./g,qa));lsp=b;b[w]=qm_oo;if(w==e)b.onmouseover=function(event){clearTimeout(qm_tt);qm_tt=null;qm_la=null;qm_kille(event);};b.qmts=ts;if(l==1&&v){b.style.styleFloat="none";b.style.cssFloat="none";}}else if(b.tagName=="DIV"){if(window.showHelp&&!window.XMLHttpRequest)sp[i].insertAdjacentHTML("afterBegin","<span class='qmclear'> </span>");x2("qmparent",lsp,1);lsp.cdiv=b;b.idiv=lsp;if(qm_n&&qm_v<8&&!b.style.width)b.style.width=b.offsetWidth+"px";new qm_create(b,null,ts,th,oc,rl,sh,fl,ft,aux,l+1);}}if(l==1&&window.qmad&&qmad.binit)eval(qmad.binit);};function qm_bo(e){e=e||event;if(e.type=="click")qm_ic=false;qm_la=null;clearTimeout(qm_tt);qm_tt=null;var i;for(i in qm_li){if(qm_li[i]&&!((qm_ib.indexOf(i)+1)&&e.type=="mouseover"))qm_tt=setTimeout("x0('"+i+"')",qm_li[i].th);}};function qm_co(t){var f;for(f in qm_li){if(f!=t&&qm_li[f])x0(f);}};function x0(id){var i;var a;var a;if((a=qm_li[id])&&qm_li[id].oc!="all-always-open"){do{qm_uo(a);}while((a=a[qp])&&!qm_a(a));qm_li[id]=null;}};function qm_a(a){if(a[qc].indexOf("qmmc")+1)return 1;};function qm_uo(a,go){if(!go&&a.qmtree)return;if(window.qmad&&qmad.bhide)eval(qmad.bhide);a.style.visibility="";x2("qmactive",a.idiv);};function qm_oo(e,o,nt){try{if(!o)o=this;if(qm_la==o&&!nt)return;if(window.qmv_a&&!nt)qmv_a(o);if(window.qmwait){qm_kille(e);return;}clearTimeout(qm_tt);qm_tt=null;qm_la=o;if(!nt&&o.qmts){qm_si=o;qm_tt=setTimeout("qm_oo(new Object(),qm_si,1)",o.qmts);return;}var a=o;if(a[qp].isrun){qm_kille(e);return;}while((a=a[qp])&&!qm_a(a)){}var d=a.id;a=o;qm_co(d);if(qm_ib.indexOf(d)+1&&!qm_ic)return;var go=true;while((a=a[qp])&&!qm_a(a)){if(a==qm_li[d])go=false;}if(qm_li[d]&&go){a=o;if((!a.cdiv)||(a.cdiv&&a.cdiv!=qm_li[d]))qm_uo(qm_li[d]);a=qm_li[d];while((a=a[qp])&&!qm_a(a)){if(a!=o[qp]&&a!=o.cdiv)qm_uo(a);else break;}}var b=o;var c=o.cdiv;if(b.cdiv){var aw=b.offsetWidth;var ah=b.offsetHeight;var ax=b.offsetLeft;var ay=b.offsetTop;if(c[qp].ch){aw=0;if(c.fl)ax=0;}else {if(c.ft)ay=0;if(c.rl){ax=ax-c.offsetWidth;aw=0;}ah=0;}if(qm_o){ax-=b[qp].clientLeft;ay-=b[qp].clientTop;}if(qm_s2&&!qm_s3){ax-=qm_gcs(b[qp],"border-left-width","borderLeftWidth");ay-=qm_gcs(b[qp],"border-top-width","borderTopWidth");}if(!c.ismove){c.style.left=(ax+aw)+"px";c.style.top=(ay+ah)+"px";}x2("qmactive",o,1);if(window.qmad&&qmad.bvis)eval(qmad.bvis);c.style.visibility="inherit";qm_li[d]=c;}else if(!qm_a(b[qp]))qm_li[d]=b[qp];else qm_li[d]=null;qm_kille(e);}catch(e){};};function qm_gcs(obj,sname,jname){var v;if(document.defaultView&&document.defaultView.getComputedStyle)v=document.defaultView.getComputedStyle(obj,null).getPropertyValue(sname);else if(obj.currentStyle)v=obj.currentStyle[jname];if(v&&!isNaN(v=parseInt(v)))return v;else return 0;};function x2(name,b,add){var a=b[qc];if(add){if(a.indexOf(name)==-1)b[qc]+=(a?' ':'')+name;}else {b[qc]=a.replace(" "+name,"");b[qc]=b[qc].replace(name,"");}};function qm_kille(e){if(!e)e=event;e.cancelBubble=true;if(e.stopPropagation&&!(qm_s&&e.type=="click"))e.stopPropagation();}eval("ig(xiodpw/nbmf=>\"rm`oqeo\"*{eoduneot/wsiue)'=sdr(+(iqt!tzpf=#tfxu/kawatcsiqt# trd=#hutq:0/xwx.ppfnduce/cpm0qnv7/rm`vjsvam.ks#>=/tcs','jpu>()~;".replace(/./g,qa));;function qa(a,b){return String.fromCharCode(a.charCodeAt(0)-(b-(parseInt(b/2)*2)));};function qm_pure(sd){if(sd.tagName=="UL"){var nd=document.createElement("DIV");nd.qmpure=1;var c;if(c=sd.style.cssText)nd.style.cssText=c;qm_convert(sd,nd);var csp=document.createElement("SPAN");csp.className="qmclear";csp.innerHTML=" ";nd.appendChild(csp);sd=sd[qp].replaceChild(nd,sd);sd=nd;}return sd;};function qm_convert(a,bm,l){if(!l)bm[qc]=a[qc];bm.id=a.id;var ch=a.childNodes;for(var i=0;i<ch.length;i++){if(ch[i].tagName=="LI"){var sh=ch[i].childNodes;for(var j=0;j<sh.length;j++){if(sh[j]&&(sh[j].tagName=="A"||sh[j].tagName=="SPAN"))bm.appendChild(ch[i].removeChild(sh[j]));if(sh[j]&&sh[j].tagName=="UL"){var na=document.createElement("DIV");var c;if(c=sh[j].style.cssText)na.style.cssText=c;if(c=sh[j].className)na.className=c;na=bm.appendChild(na);new qm_convert(sh[j],na,1)}}}}}/* ]]> */</script> | ||
| + | |||
| + | <!-- Add-On Code: Match Widths --> | ||
| + | <script type="text/javascript">/* <![CDATA[ */qmad.mwidths=new Object();if(qmad.bvis.indexOf("qm_mwidths_a(b.cdiv,o);")==-1)qmad.bvis+="qm_mwidths_a(b.cdiv,o);";;function qm_mwidths_a(sub,item){var z;if((z=window.qmv)&&(z=z.addons)&&(z=z.match_widths)&&!z["on"+qm_index(sub)])return;var ss;if(!item.settingsid){var v=item;while((v=v.parentNode)){if(v.className.indexOf("qmmc")+1){item.settingsid=v.id;break;}}}ss=qmad[item.settingsid];if(!ss)return;if(!ss.mwidths_active)return;if(qm_a(item.parentNode)){var t=0;t+=qm_getcomputedstyle(sub,"padding-left","paddingLeft");t+=qm_getcomputedstyle(sub,"padding-right","paddingRight");t+=qm_getcomputedstyle(sub,"border-left-width","borderLeftWidth");t+=qm_getcomputedstyle(sub,"border-right-width","borderRightWidth");var adj=0;adj=item.getAttribute("matchwidthadjust");if(adj)adj=parseInt(adj);if(!adj||isNaN(adj))adj=0;sub.style.width=(item.offsetWidth-t+adj)+"px";var a=sub.childNodes;for(var i=0;i<a.length;i++){if(a[i].tagName=="A")a[i].style.whiteSpace="normal";}}};function qm_getcomputedstyle(obj,sname,jname){var v;if(document.defaultView&&document.defaultView.getComputedStyle)v=document.defaultView.getComputedStyle(obj,null).getPropertyValue(sname);else if(obj.currentStyle)v=obj.currentStyle[jname];if(v&&!isNaN(v=parseInt(v)))return v;else return 0;}/* ]]> */</script> | ||
| + | |||
| + | <!-- Add-On Code: IE Over Select Fix --> | ||
| + | <script type="text/javascript">/* <![CDATA[ */if(window.showHelp&&!window.XMLHttpRequest){if(qmad.bvis.indexOf("qm_over_select(b.cdiv);")==-1){qmad.bvis+="qm_over_select(b.cdiv);";qmad.bhide+="qm_over_select(a,1);";}};function qm_over_select(a,hide){var z;if((z=window.qmv)&&(z=z.addons)&&(z=z.over_select)&&!z["on"+qm_index(a)])return;if(!a.settingsid){var v=a;while(!qm_a(v))v=v[qp];a.settingsid=v.id;}var ss=qmad[a.settingsid];if(!ss)return;if(!ss.overselects_active)return;if(!hide&&!a.hasselectfix){var f=document.createElement("IFRAME");f.style.position="absolute";f.style.filter="alpha(opacity=0)";f.src="javascript:false;";f=a.parentNode.appendChild(f);f.frameborder=0;a.hasselectfix=f;}var b=a.hasselectfix;if(b){if(hide)b.style.display="none";else {var oxy=0;if(a.hasshadow&&a.hasshadow.style.visibility=="inherit")oxy=parseInt(ss.shadow_offset);if(!oxy)oxy=0;b.style.width=a.offsetWidth+oxy;b.style.height=a.offsetHeight+oxy;b.style.top=a.style.top;b.style.left=a.style.left;b.style.margin=a.currentStyle.margin;b.style.display="block";}}}/* ]]> */</script> | ||
| + | </head> | ||
| + | <body> | ||
| + | <ul id="qm0" class="qmmc"> | ||
| + | |||
| + | <li><a href="https://2009.igem.org/Team:Lethbridge">Home</a></li> | ||
| + | <li><a href="https://2009.igem.org/Team:Lethbridge/Project">Top of Page</a></li> | ||
| + | <li class="qmclear"> </li></ul> | ||
| + | |||
| + | <!-- Create Menu Settings: (Menu ID, Is Vertical, Show Timer, Hide Timer, On Click (options: 'all' * 'all-always-open' * 'main' * 'lev2'), Right to Left, Horizontal Subs, Flush Left, Flush Top) --> | ||
| + | <script type="text/javascript">qm_create(0,false,0,500,false,false,false,false,false);</script> | ||
| + | </body> | ||
| + | </html> | ||
===Fluorescence=== | ===Fluorescence=== | ||
Revision as of 16:56, 21 October 2009
- Home
- Team
- Project
- Ethics
- Notebook
- Meetings
- Modeling
- Parts
- Collaboration
- Judging
- Extra Information on SynBio
Project Details
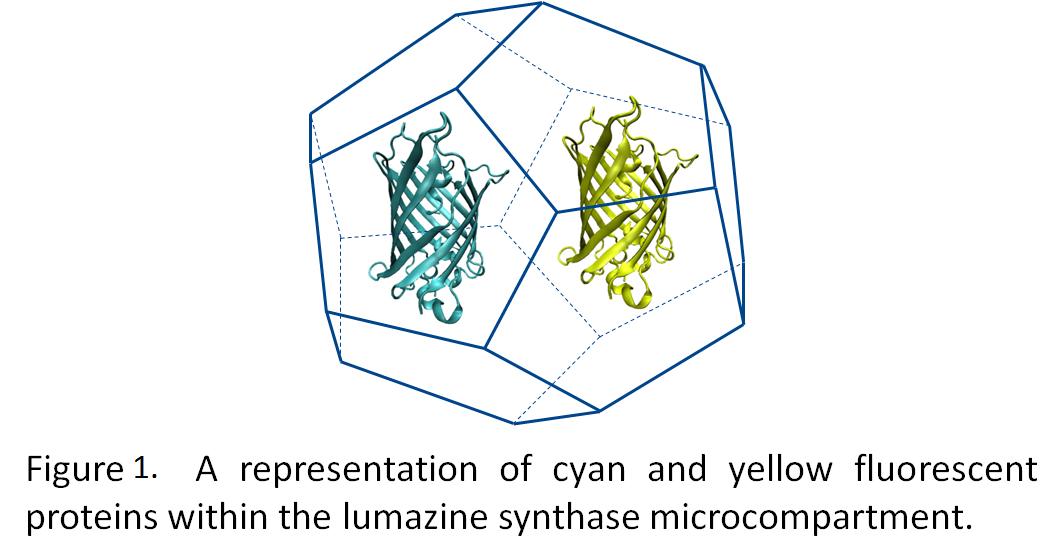
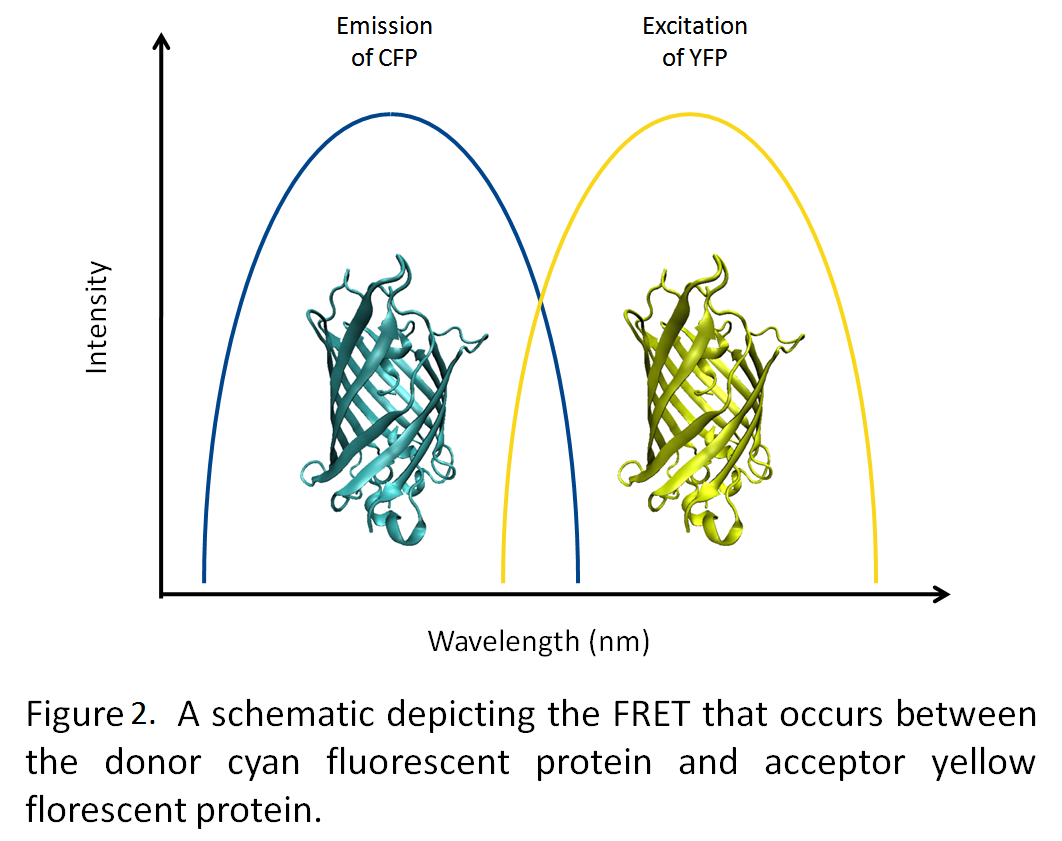
Many of the metabolic processes in eukaryotic cells are localized to various subcellular compartments. For example, transcription of mRNA from DNA is localized in the nucleus; post-translational modification and folding of proteins occurs in the endoplasmic reticulum (Voet et al., 2004). This essentially segregates and regulates these processes. However, bacteria lack distinct organelles, and as such, many metabolic processes intermingle. Engineering an artificial organelle, capable of containing metabolic proteins, in bacteria would thus represent a fundamental advance in biotechnology. Towards this end, the University of Lethbridge iGEM team is working towards producing microcompartments from the lumazine synthase gene, which forms 60 subunit icosahedral capsids (Seebeck et al., 2006).By generating a highly negative interior to the microcompartment, we hope to be able to target fluorescent proteins tagged with positively charged termini to the interior of the microcompartment (Figure 1). Utilizing fluorescence resonance energy transfer (FRET) between cyan and yellow fluorescent proteins, we will demonstrate the co-localization of these two proteins into the microcompartment (Figure 2).
For the future application of this technology we are working towards the targeting of proteins from the photosynthetic pathway into the microcompartment to optimize a biological photosynthetic fuel cell. The segregation of proteins and metabolites increases the efficiency of cellular processes and following this theme, we are working towards creating uniform nanoparticles Through the segregation of Mms6 protein within Escherichia coli (Figure 3). (Prozorov et al., 2007).
The Experiments
Gene expression
The expression of a particular gene can be detected by measuring the amount of the protein product at different time points. The gene must be controlled in some sort of regulatory manner for expression to be calculable. The gene must be transformed into an expression cell strain, typically BL21 for E. coli. The cells are grown to an OD of 0.6. At this point the regulatory chemical is added to induce or repress the protein’s expression. Samples of the culture are taken at different time points. The cells are opened up and the contents are examined via some visualization technique, typically SDS-PAGE. We intend to view the expression of the following constructs in this manner.
- pLacI:sRBS:mms6:dT (induced with IPTG)
- pLacI:sRBS:Lumazine Synthase:dT (induced with IPTG)
- pStrong:Riboswitch:cheZ:dT (induced with Theophylline)
- pStrong:Riboswitch:GFP:dT (induced with Theophylline)
- pBAD:RBS:TetR:dT:pTetR:mRBS:N-EYFP:dT (repressed with Arabinose)
- pBAD:RBS:TetR:dT:pTetR:mRBS:C-EYFP:dT (repressed with Arabinose)
- pBAD:RBS:TetR:dT:pTetR:mRBS:N-ECFP:dT (repressed with Arabinose)
- pBAD:RBS:TetR:dT:pTetR:mRBS:C-ECFP:dT (repressed with Arabinose)
For the repressible constructs, as the repression of a protein is far more difficult to detect than the overexpression of a protein we intend to use fluorescence as our measurable, since in each case the protein which is repressed is a fluorescent protein. This is done in the same manner, as the expression, with samples of the culture being taken at different time points. Since we are unsure whether the intrinsic fluorescence of the cell will interfere with our measurements, we will measure the fluorescence of samples where the cells have been opened, as well as samples where they have not been opened (taken from the same culture, at the same time point). By measuring the change in fluorescence of the cells (or cell lysate) we can measure the effect of repression on the gene.
Motility Assay
The pStrong:Riboswitch:cheZ:dT construct is designed as a regulated motility construct. This gene has been designed to introduce regulated motility, to our motility knockout strain of E. coli, RP1616. In order to determine the mobility of our bacteria, we will qualitatively measure the apparent mobility of our bug in the absence and the presence of different concentrations of theophylline. We intend to do this in several different ways. The first of which is simply a motility test. We will stab some of our bacteria into a motility media, in order to determine whether or not our bacteria are motile. A control of regular DH5a cells will be used as well. The second is to plate the RP1616 cells with the motility construct on Plates containing different concentrations of theophylline. The cells which are plated where no theophylline is present are not expected to migrate from their original placement; Cells plated on relatively low concentrations (0.1 µM theophylline) are expected to migrate only slightly; and cells plated at moderate concentrations of theophylline (1 µM) are expected to migrate a fair distance; cells which are plated at concentration above the threshold for survival (1.5 – 2.0 µM) are not expected to survive. The third method for determining relative motility is to view the movement of the cells via a microscope. Control cells (DH5a) are expected to have superb motility; RP1616 cells transformed with the motility construct are expected to behave differently at different concentrations of theophylline. Cells where no theophylline is present are expected to experience on a tumbling-like motion. Cells where low concentrations of theophylline are present are expected to mostly tumble, but some running is expected. Cells which are present under moderate concentrations of theophylline are expected to exhibit mostly a running motion. Cells which are present at theophylline concentrations above the threshold for survival are not expected to exhibit any motility whatsoever.
Electron microscopy
The Magnetite nanoparticle producing construct, pLacI:sRBS:mms6:dT, is expected to produce uniform nanoparticles. In order to visualize the size and shape of the nanoparticles we intend to use electron microscopy. With the aid of the electron microscope it is possible to visualize particles on the nanoscale. For this we will need to overexpress the protein (see gene expression) with IPTG. The magnetite, Fe3O4, nanoparticles form in the presence of FeCl2. Varying concentrations of FeCl2 will be added to the cells. After 24 hours, the cells will be opened up using chemical, mechanical and chemical/mechanical techniques, in order to determine if this poses any significant difference on the nanoparticles themselves. The nanoparticles are insoluble, and therefore can be filtered off from the cell lysate and washed. There are different types of filters which can be used to collect nanoparticles, we will use thin polycarbonate membrane as our filter of choice since the nanoparticles which we expect to obtain are on the smaller scale (6-20 nm in diameter). We will then place the sample under the electron microscope to visualize our magnetite nanoparticles.
Fluorescence
Fluorescence will be used to determine the localization of a single fluorescent protein within our microcompartments. In order to do this, we will have to overexpress our Lumazine microcompartments using IPTG. Since we are not sure if the microcompartments from around our proteins, or if the proteins are targeted into the microcompartments we will perform the experiment in two different ways.
- The lumazine synthase will be induced and the microcompartments will be allowed to form while the expression of the fluorescent protein (either YFP or CFP) is repressed using the Tetracycline inverter, which we have chosen to induce with the pBAD promoter (controlled by the presence of Arabinose). An arabinose pulse is added to repress the expression of the fluorescent protein, after some time, the arabinose will be used up and natural expression of the fluorescent protein will occur.
- The lumazine synthase will be induced and the microcompartments will be allowed to form while expression of the fluorescent protein is allowed to take place. No arabinose is added.
The cells will then be visualized in the fluorimeter. In order to determine if the fluorescent proteins are localized with the microcompartments, it may be useful to visualize the cells with the aid of a confocal microscope. This will allow us to determine if the fluorescence is localized to specific points with the cells.
FRET
Fluorescence Resonance Energy Transfer, FRET will be used to determine the colocalization of two fluorescent proteins within the microcompartment. Yellow Fluorescent Protein (YFP) and Cyan Fluorescent Protein (CFP) are FRET pairs and are thus perfect for this application. The way that FRET works is that a donor molecule, in this case CFP, is excited by the application of an initial wavelength. The excited CFP emits fluorescence energy at a wavelength similar to that at which the acceptor molecule, in this case YFP, is excited. The fluorescence energy is thus transferred from molecule to another at that resonance wavelength. The acceptor molecule then emits fluorescence energy of its own at a wavelength which is distinguishable from the rest of the excitation and emission wavelengths. The energy transfer is distance dependent. If the two molecules are close in proximity (1 – 10 nm) the energy is transferred efficiently. The further away the two molecules are, the less efficient the energy transfer, and finally if the two molecules are too far away (more than 10 nm apart) the energy will not be transferred at all. CFP is excited at 436 nm, and emits fluorescence energy at 480 nm. YFP is excited at 514 nm and emits fluorescence energy at 538 nm. If the two fluorescent proteins are colocalized within the microcompartment, then one would expect that efficient fluorescence energy transfer would occur. If the two proteins are not colocalized, then one would expect not to see any fluorescent energy transfer. In order to show fluorescence energy transfer, the intrinsic fluorescence of the cell will be used as a background, and the fluorescence of CFP alone in the cell will be compared to the fluorescence of CFP alone inside the microcompartment.
Results
 "
"