Team:NYMU-Taipei/Project/Lab Note
From 2009.igem.org
(→Removal Design) |
(→Removal Design) |
||
| Line 98: | Line 98: | ||
=== Removal for Binding Viruses === | === Removal for Binding Viruses === | ||
| - | Mentioned before at [[Team:NYMU-Taipei/Project/ | + | Mentioned before at [[Team:NYMU-Taipei/Project/signal transduction|Signal]], an output promoter called ompC promoter will be inactivate through the way of the signal transduction of binding with viruses. Obviously, if we use a promoter inhibited by the ompC promoter output protein , when ompC inactivated, the gene we want to express will tun on. Therefore, we come out this design: |
[[image:NYMU 5.jpg]](this picture must be changed) | [[image:NYMU 5.jpg]](this picture must be changed) | ||
Revision as of 20:37, 21 October 2009
| Home | Project Overview: | Chassis | Receptors | Removal | Experiments and Parts | F.A.Q | About Us |
Motivation
Our engineered E. coli (ViroCatcher) is designed to trap the viruses and successfully prevents viruses spreading out in our bodies. If we remove ViroCatcher, viruses will be cleaned up together as well with our ViroCatcher. To remove viruses, we must work out a method for E. coli removal.
For removing ViroCatcher, we have two choices. One is to let it suicide, and the other is to let it be removed by our immune cells (like how dead cells are removed). Before removal, we must be sure that the intended work is done, and viruses must be removed at the same time. Therefore, we designed a removal gene and included it in ViroCatcher. If ViroCatcher gets the sinal of binding with viral proteins, it will turn on the removal gene and be removed.
Furthermore, if ViroCatcher did not catch anything, we still have to design a mechanism to let our ViroCatcher express the removal gene and remove itself. Otherwise, bacteria will fill our blood vessel and block our blood flow.
Selecting the Removal Gene
We had considered the following three removal genes. In the end, we chose to use the expression of LPS.
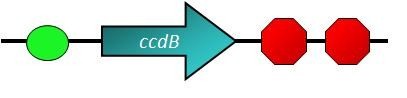
Cell Suicide Generator
- The CcdB protein, constitutively expressed by P1010, is lethal to most of the BioBrick cell strains, only DB3.1 is resistant[5]. A hybrid promoter controls the production of LuxR and ccdB. The promoter is activated by HSL-LuxR complex and repressed by P22 c2. The LuxR coding region is preceded by a strong RBS and followed by a bad terminator (60% efficiency). This means there is read through into the ccdB coding region which can be translated from a very weak RBS. If the P22 c2 repressor is absent, there should be a strong enough LuxR background to allow autoactivation if HSL is added. The amound of ccdB should be sublethal as long as there is no significant activation[6].
The most simple way to get rid of the E. coli is to let it suicide, however, there are many viruses that have been caught on the surface that we have to pay attention to. If ViroCatcher dies or explodes, the trapped viruses will spread into our bloodstream again. Cell Suicide Generator seems not to be a good choice.
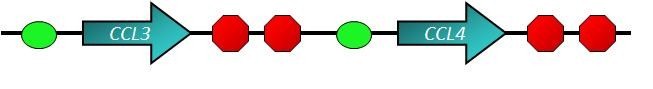
Macrophage Inflammatory Proteins
- Macrophage Inflammatory Proteins (MIP) belong to the family of chemotactic cytokines known as chemokines. In humans, there are two major forms, MIP-1α and MIP-1β that are now officially named CCL3 and CCL4 respectively. Both are major factors produced by macrophages after they are stimulated with bacterial endotoxins[1]. They activate human granulocytes (neutrophils, eosinophils and basophils) which can lead to acute neutrophilic inflammation. They also induce the synthesis and release other pro-inflammatory cytokines such as interleukin 1 (IL-1), IL-6 and TNF-α from fibroblasts and macrophages. The genes for CCL3 and CCL4 are both located on human chromosome 17[2].
- Chemokine (C-C motif) ligand 3, also known as CCL3, is a human gene. Macrophage inflammatory protein-1 is a so-called monokine that is involved in the acute inflammatory state in the recruitment and activation of polymorphonuclear leukocytes (Wolpe et al., 1988). Sherry et al. (1988) demonstrated 2 protein components of MIP1, called by them alpha and beta[supplied by OMIM][1].
- CCL4 is a CC chemokine with specificity for CCR5 receptors. It is a chemoattractant for natural killer cells, monocytes and a variety of other immune cell [2]. CCL4 is a major HIV-suppressive factor produced by CD8+ T cells [3]. Perforin-low memory CD8+ T cells that normally synthesize MIP-1-beta [4].
- The main function of chemokines is the induction of cell migration. Cells will move toward the direction of increment of continuous chemokine concentration gradient. In other words, cells migrate toward the source of chemokine. For example, the induction of lymphocyte to the lymphatic node is due to chemokines. Apply this thinking to our design, we want to use CCL3 and CCL4, or chemokines for macrophages, to assemble macrophages around ViroCatcher. It will raise the possibility that macrophages recognize the proteins of viruses and swallow the E. coli whole.
But there's a problem. CCL3 and CCL4 is two kinds of ligands of receptors of antibody. If we use this design, ViroCatchers will bind with each other, aggregate together, and block our blood vessels. So unfortunately, this design still cannot be used as the removal circuit.
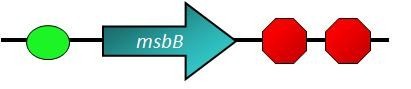
Expression of LPS
- In the Chassis part of our project, we have mentioned that ViroCatcher is a LPS[7] knock-out stain, in order to avoid the detection by the immune system. Therefore, if we re-express LPS, our ViroCatcher will have the ability to attract immune cells and let immune system macrophage remove ViroCatcher itself along with the viruses bound to the surface[8][9]. To express LPS again in ViroCatcher, we just have to turn on msbB gene.
Homo sapiens chemokine (C-C motif) ligand 3, mRNA (cDNA clone MGC:198546 IMAGE:9054485), complete cds[http://www.ncbi.nlm.nih.gov/nuccore/219521699]
1 acactcgagc ccacattccg tcacctgctc agaatcatgc aggtctccac tgctgccctt
61 gctgtcctcc tctgcaccat ggctctctgc aaccagttct ctgcatcact tgctgctgac
121 acgccgaccg cctgctgctt cagctacacc tcccggcaga ttccacagaa tttcatagct
181 gactactttg agacgagcag ccagtgctcc aagcctggtg tcatcttcct aaccaagcga
241 agccggcagg tctgtgctga ccccagtgag gagtgggtcc agaaatatgt cagcgacctg
301 gagctgagtg cctgaggggt ccagaagctt cgaggcccag cgacctcggt gggcccagtg
361 gggaggagca ggagcctgag ccttgggaac atgcgtgtga cctccacagc tacctcttct
421 atggactggt tgttgccaaa cagccacact gtgggactct tcttaactta aattttaatt
481 tatttatact atttagtttt tgtaatttat tttcgatttc acagtgtgtt tgtgattgtt
541 tgctctgaga gttccccctg tcccctc
Homo sapiens chemokine (C-C motif) ligand 4, mRNA (cDNA clone MGC:126026 IMAGE:40032172), complete cds [http://www.ncbi.nlm.nih.gov/nuccore/74355495]
1 cacagctggg ttctgaagct tctgagttct gcagcctcac ctctgagaaa acctcttttc
61 caccaatacc atgaagctct gcgtgactgt cctgtctctc ctcatgctag tagctgcctt
121 ctgctctcca gcgctctcag caccaatggg ctcagaccct cccaccgcct gctgcttttc
181 ttacaccgcg aggaagcttc ctcgcaactt tgtggtagat tactatgaga ccagcagcct
241 ctgctcccag ccagctgtgg tattccaaac caaaagaagc aagcaagtct gtgctgatcc
301 cagtgagacc tgggtccagg agtacgtgta tgacctggaa ctgaactgag ctgctcagag
361 acaggaagtc ttcagggaag gtcacctgag cccggatgct tctccatgag acacatctcc
421 tccatactca ggactcctct ccgcagttcc tgtcccttct cttaatttaa tcttttttat
481 gtgccgtgtt attgtattag
Escherichia coli K-12 substr. MG1655 msbB gene sequence[http://biocyc.org/ECOLI/sequence-rc?type=GENE&object=EG10614]
1 atgGAAACGA AAAAAAATAA TAGCGAATAC ATTCCTGAGT TTGATAAATC CTTTCGCCAC
61 CCGCGCTACT GGGGAGCATG GCTGGGCGTA GCAGCGATGG CGGGTATCGC TTTAACGCCG
121 CCAAAGTTCC GTGATCCCAT TCTGGCACGG CTGGGACGTT TTGCCGGACG ACTGGGAAAA
181 AGCTCACGCC GTCGTGCGTT AATCAATCTG TCGCTCTGCT TTCCAGAACG TAGTGAAGCT
241 GAACGCGAAG CGATTGTTGA TGAGATGTTT GCCACCGCGC CGCAAGCGAT GGCAATGATG
301 GCTGAGTTGG CAATACGCGG GCCGGAGAAA ATTCAGCCGC GCGTTGACTG GCAAGGGCTG
361 GAGATCATCG AAGAGATGCG GCGTAATAAC GAGAAAGTTA TCTTTCTGGT GCCGCACGGT
421 TGGGCCGTCG ATATTCCTGC CATGCTGATG GCCTCGCAAG GGCAGAAAAT GGCAGCGATG
481 TTCCATAATC AGGGCAACCC GGTTTTTGAT TATGTCTGGA ACACGGTGCG TCGTCGCTTT
541 GGCGGTCGTC TGCATGCGAG AAATGACGGT ATTAAACCAT TCATCCAGTC GGTACGTCAG
601 GGGTACTGGG GATATTATTT ACCCGATCAG GATCATGGCC CAGAGCACAG CGAATTTGTG
661 GATTTCTTTG CCACCTATAA AGCGACGTTG CCCGCGATTG GTCGTTTGAT GAAAGTGTGC
721 CGTGCGCGCG TTGTACCGCT GTTTCCGATT TATGATGGCA AGACGCATCG TCTGACGATT
781 CAGGTGCGCC CACCGATGGA TGATCTGTTA GAGGCGGATG ATCATACGAT TGCGCGGCGG
841 ATGAATGAAG AAGTCGAGAT TTTTGTTGGT CCGCGACCAG AACAATACAC CTGGATACTA
901 AAATTGCTGA AAACTCGCAA ACCGGGCGAA ATCCAGCCGT ATAAGCGCAA AGATCTTTAT
961 CCCATCAAAT AA
*Primer Design gaattcgcggccgcttctag atgGAAACGAAAAAAAATAATAGCGAA 55deg GC:26% length:27bp ctgcagcggccgctactagta TTATTTGATGGGATAAAGATCTTTGC 54deg GC:31% length:26bp
Goals
There are two specific aims for the removal part. One is ViroCatcher binding to viruses and being removed, and the other is self-removal after a specified period of time. Since the gene msbB of LPS is knocked out, if there is an insertion of msbB gene into the plasmid, then msbB gene and the LPS functions will express. Once the LPS is expressed, the immune system is turned on and ViroCatcher is swallowed by macrophage through phagocytosis along with many viruses attached.
If the machine catches nothing, it is still necessary to remove the E. coli machine after a period of time. This period of time should be controlled precisely, otherwise the machine will be removed before it catches any viruses. We compose an oscillator and a toggle switch together as our timer.
Removal Design
Removal for Binding Viruses
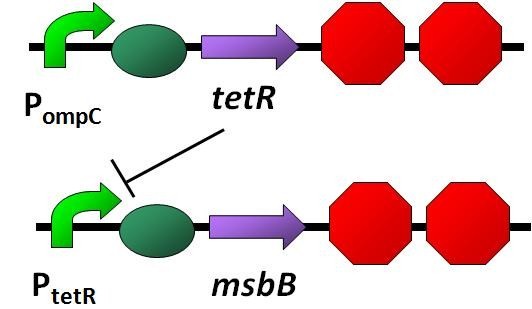
Mentioned before at Signal, an output promoter called ompC promoter will be inactivate through the way of the signal transduction of binding with viruses. Obviously, if we use a promoter inhibited by the ompC promoter output protein , when ompC inactivated, the gene we want to express will tun on. Therefore, we come out this design:
 (this picture must be changed)
(this picture must be changed)
Use the tetR promoter as a switch for the msbB gene. If the receptor catches a virus, TetR protein absent, tetR promoter will turn on msbB gene, then LPS will be expressed and it will attract the immune system. ViroCatcher will be cleaned up by immune cells together with the trapped viruses.
Timed Removal
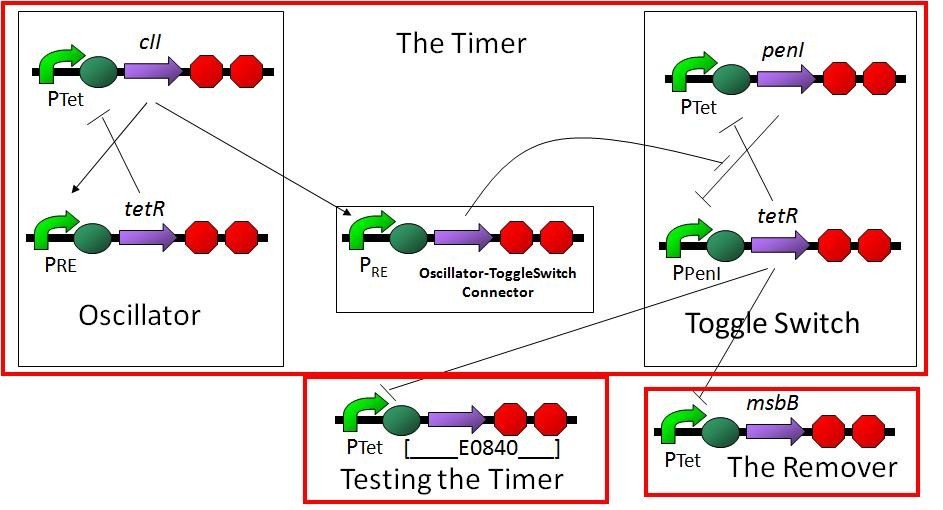
If the receptor does not catch anything, the ompC promoter will not be turned on, and the removal gene will not be expressed, so ViroCatcher cannot be removed when it catches nothing. How to make sure our Viro Catcher is removed from the human body? We worked out this set of design:
This circuit is composed of three parts - oscillator, toggle switch, and remover. Oscillator and toggle switch work together as the timer. The oscillator is consist of two genes. One gene activate the other gene and get a negative feedback. When output proteins of oscillator reach a threshold that preset, then the promoter of toggle switch will be activate, and output protein of toggle switch will suddenly activate the promoter of remover.
The following picture shows how the timer works by modeling:
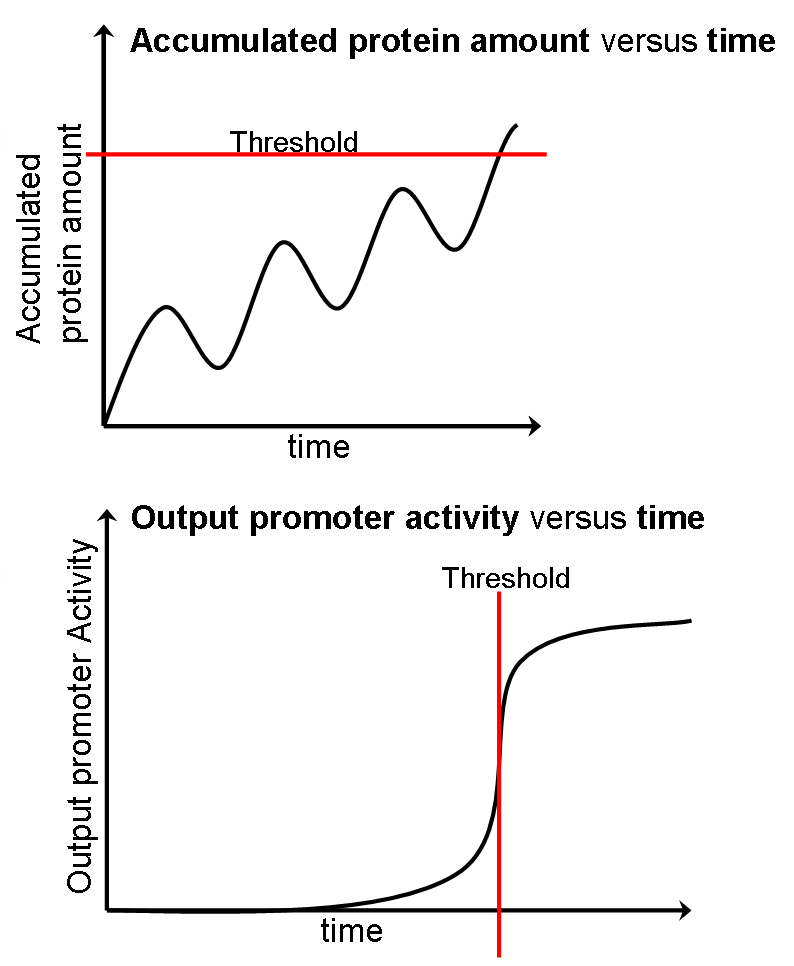
We stop the timer by setting a threshold. As the protein produced by the oscillator promoter accumulates and reaches a certain threshold, the output promoter activity would suddenly change. This output promoter is connected to removal gene msbB. Therefore, at the time that timer stops, LPS expressed and attract macrophages.
This setting can control the micromachine to trigger an immune response after a period of time. This period of time is controlled precisely, otherwise ViroCatcher will be removed too fast or too late. Anyway, no matter what kind of situation, our ViroCatcher will be removed.
Experiment Results
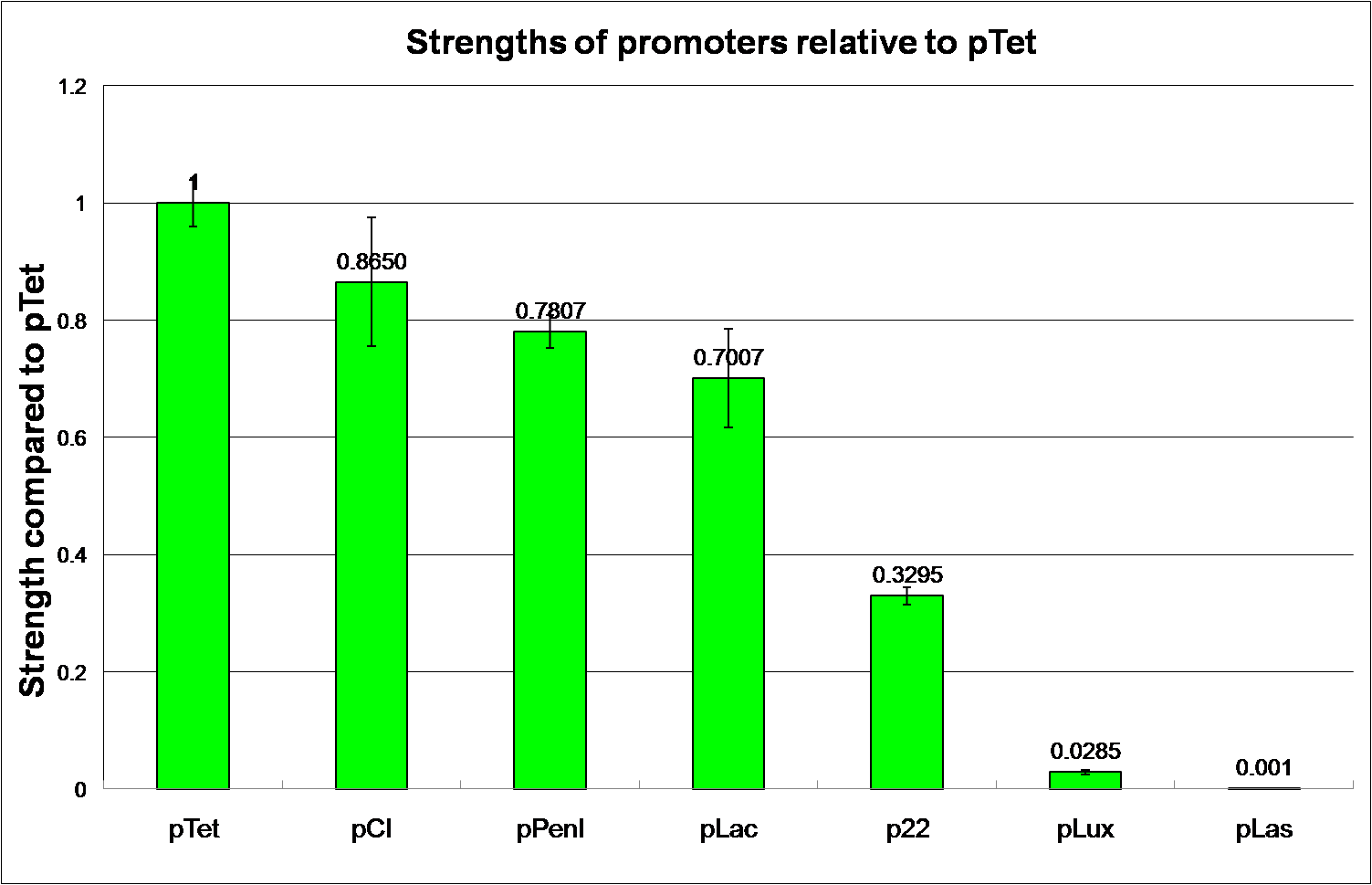
Protocol of Promoter Strength Testing
- extract the Biobrick promoter
- combine it with GFP generator [http://partsregistry.org/Part:BBa_I13504 BBa_I13504] with the promoter and then transform
- colony PCR
- liquid incubation and plasmid extraction
- measure the amount of fluroresence by using an ELISA reader with a 96 well plate
- data collection
- using pTet as the standard having the value of 1
- characterize the basal level expression without any inducer or repressor interaction
Oscillator Modeling
Promoter Strength Testing
Reporting Assay of Promoter Strength Testing
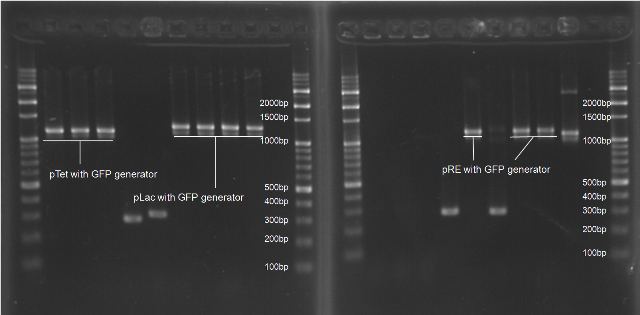
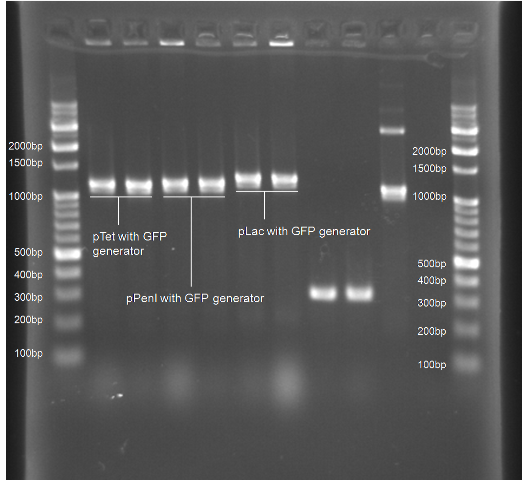
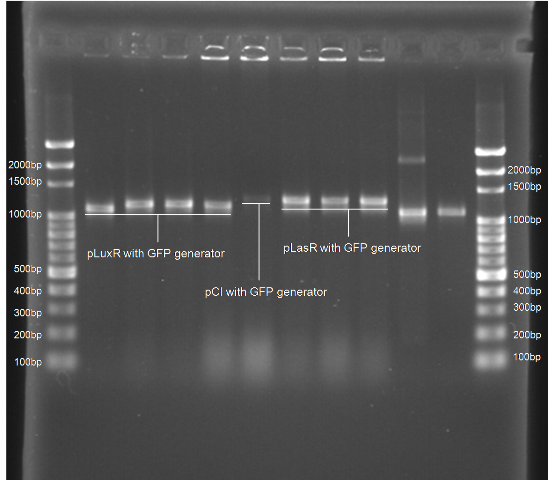
Colony PCR Gel Pictures
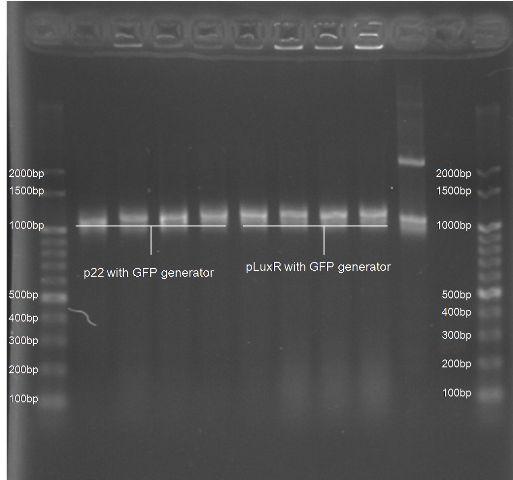 | Figure 4. L1~L4 p22 with generator ([http://partsregistry.org/Part:BBa_K195614 BBa_K195614]) L5~L8 pLuxR with generator ([http://partsregistry.org/Part:BBa_K195615 BBa_K195615]) |
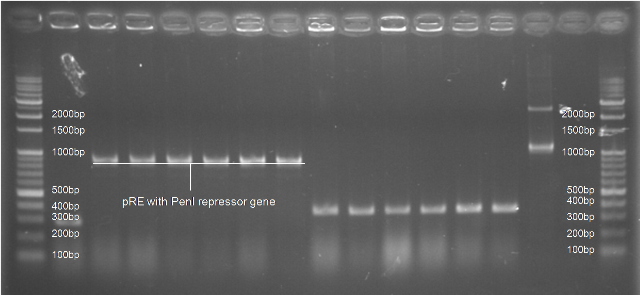 | Figure 5. L1~L6 pRE with PenI repressor gene ([http://partsregistry.org/Part:BBa_K195621 BBa_K195621]) |
Graph of Experiments
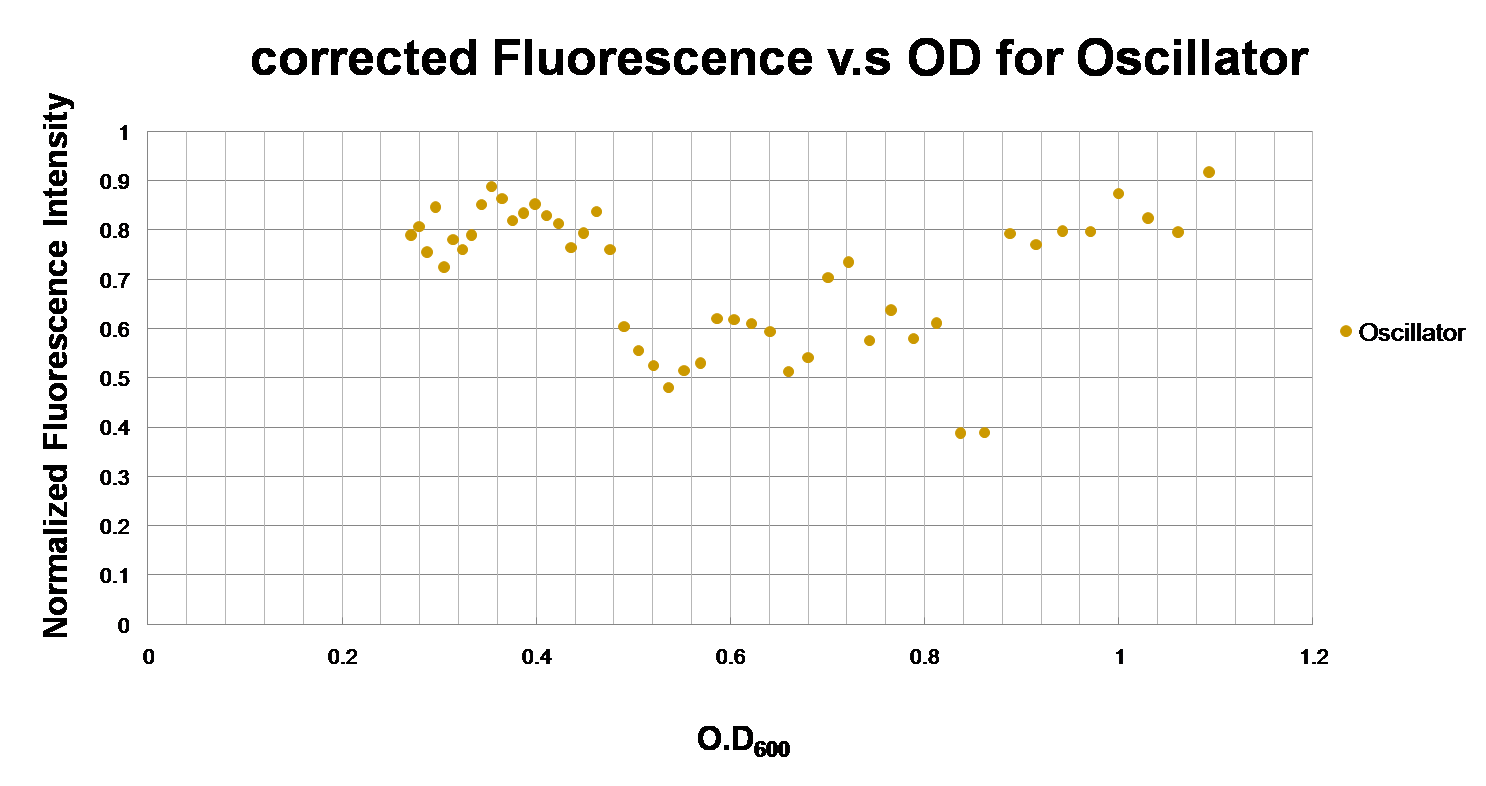
 | Figure 2. This is the component testing with the activator CII at the downstream of pLac inducing pRE to express GFP. |
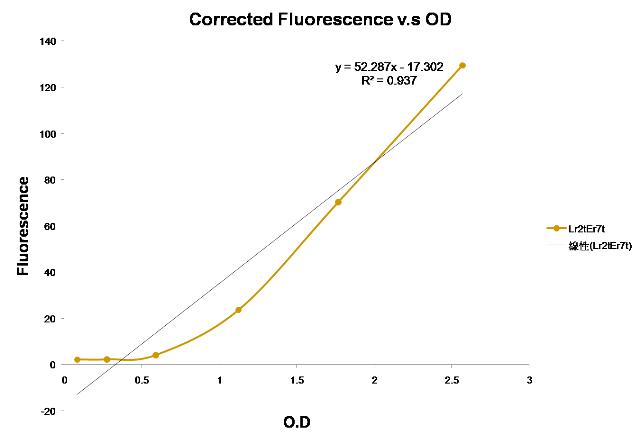
 | Figure 3. This is the measurement by using an ELISA reader, graphing Fluorescence versus O.D of the Time Delay device(I) ([http://partsregistry.org/Part:BBa_K195622 BBa_K195622]) |
msbB gene PCR Results
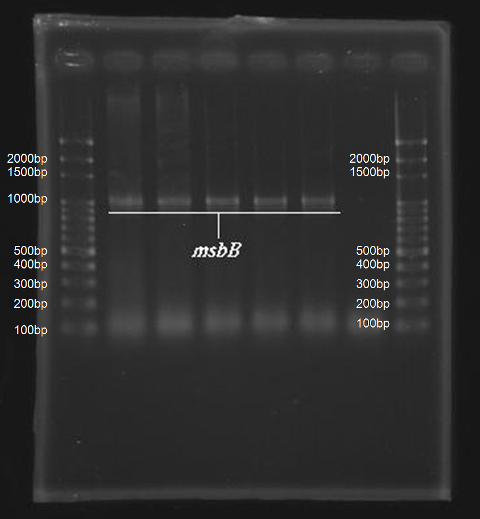 | Figure 1. This gel picture shows the result of PCR by L1~L5 is the length of msbB gene. L6 is negative control. |
References
[3] File:Macrophage activation switching - an asset for the resolution of inflammation.pdf
[4] [http://www.springerlink.com/content/g262813537x0t254/ The effect of various inflammatory agents on the phagocytosis and cytokine profile of mouse and rat macrophages]
[5] [http://partsregistry.org/wiki/index.php/Part:BBa_P1010/ Part:BBa_P1010]
[6] [http://partsregistry.org/wiki/index.php/Part:BBa_K145230/ Part:BBa_K145230]
[7] [http://en.wikipedia.org/wiki/Lipopolysaccharide#Lipid_A/ LPS]
[8] File:NYMU LPS濃度對於細胞CD14的表現的影響.pdf
[9] File:Ginger extract inhibits LPS induced macrophage activation and function.pdf
 "
"