Abstract
Our project goal is to maximize the photosynthetic productivity for a culture of Rhodobacter sphaeroides under both high and low light intensities in a bioreactor by synthetically regulating the size of the light-harvesting antenna. We chose to undertake such a project in R. sphaeroides due to its well-characterized photosynthetic and genetic system.
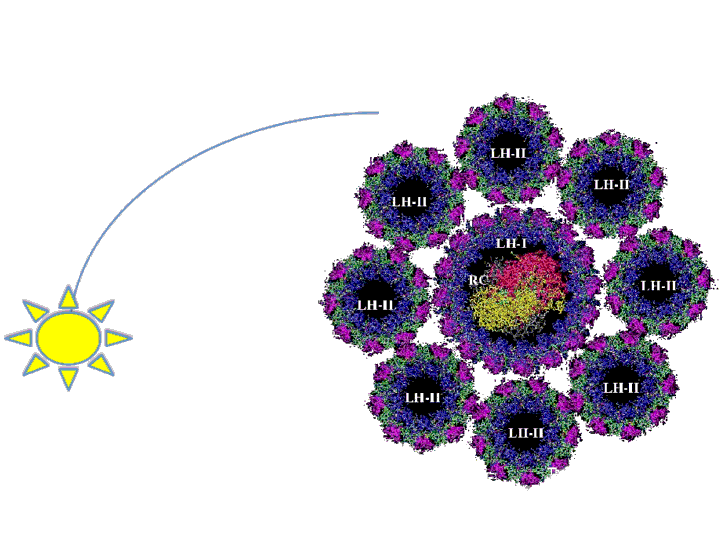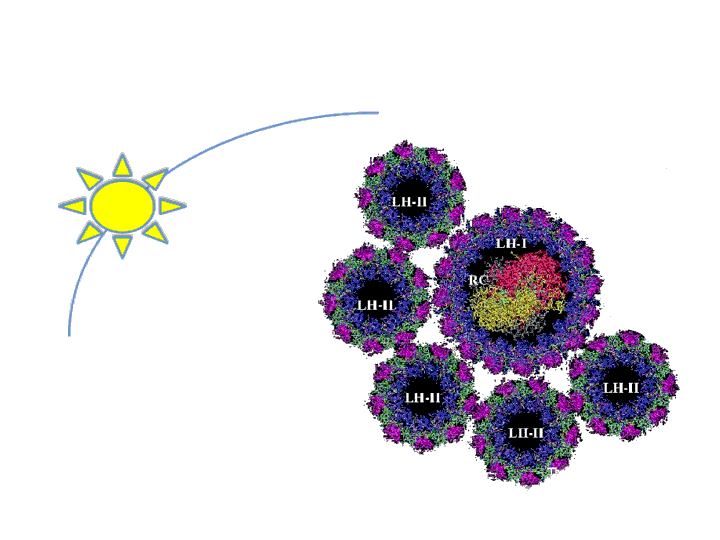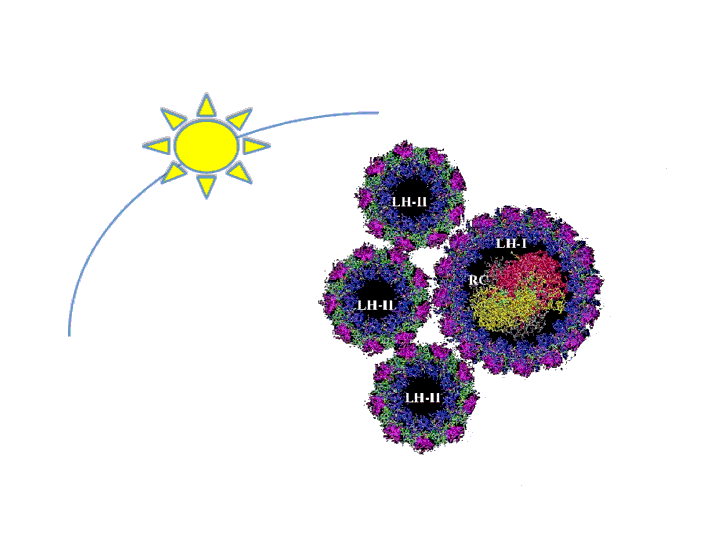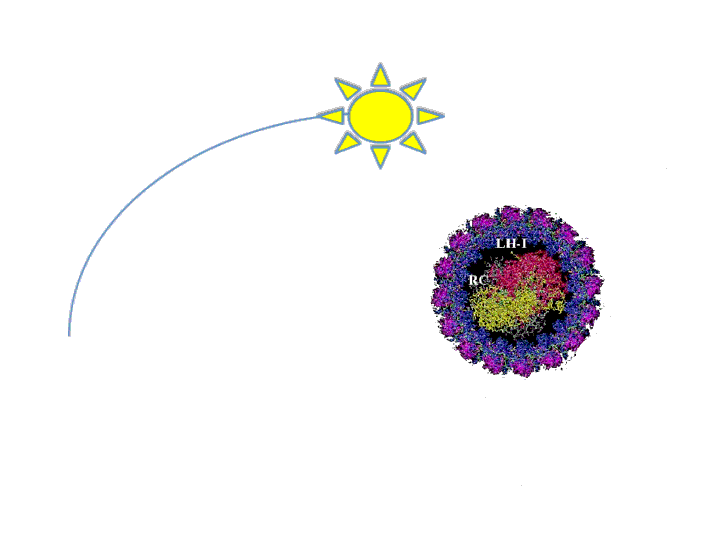
The antenna system functions to expand the spectrum of light available for photosynthesis by absorbing different wavelengths than that of the reaction center. Essentially, it is like the dish around the main receiver of any antenna. Marine bacteria, such as R. sphaeroides, evolved very large antenna complexes to absorb light in a natural environment where there is great competition for photons. As a result, the photosynthetic machinery is saturated at a rather low light intensity in a synthetic non-competitive environment, such as a bioreactor. This causes up to 95% of incidental photons to be dissipated as heat or fluorescence by the bacteria at the exterior of a bioreactor through a process called Non-Photochemical Quenching (NPQ) (Mussgnug et al., 2007). In essence, these photons are being wasted as NPQ reduces light penetration into a bioreactor and starves cells on the interior for photons. One method that has been shown to improve photosynthetic efficiency is the reduction of light-harvesting antenna sizes (Polle et al., 2002, Mussgnug et al., 2007). Though, current approaches to this end rely on genetic knockouts and as such are difficult to precisely control from the perspective of metabolic engineering and synthetic biology. Our intention is to create a more dynamic system to vary antenna size that is dependent on incidental light intensity and that can be readily optimized using synthetic biology principles. This system should result in the bacteria at the exterior of the bioreactor expressing fewer light harvesting antenna proteins than the cells at the interior, reducing NPQ while maintaining a high absorbance of incidental photons.
We will focus on altering the quantity of the Light Harvesting Complex 2 (LH2) by regulating the pucB/A genes that code for the two subunits of the complex. LH2 absorbs photons maximally at the wavelength of 842 nm and funnels its energy to LH1 and the reaction center. The ratio of LH2 complexes to LH1 naturally ranges from 3.0 to 6.7 under varying light conditions (Scheuring et al., 2005). We propose that if this ratio of LH2 to LH1 is altered to range from 0-7 or more in response to incidental light intensity, then the photosynthetic efficiency and productivity for a culture of R. sphaeroides may be maximized.
Back To Top
Analysis
full report
Back To Top
Results
results
Back To Top
Conclusion
conclusion
Back To Top
References
- "2007-2008 Catalog & Technical Reference." New England BioLabs Inc. (2007).
- Alon, Uri. Introduction to systems biology and the design principles of biological networks. Boca Raton, FL: Chapman & Hall, 2006.
- Ausubel, Fred et al. Short Protocols in Molecular Biology: Third Edition. Canada: John Wiley and Sons, Inc., 1999.
- "BioBrick Assembly Help." Registry of Standard Biological Parts. 2009. 6 July 2009. <http://partsregistry.org/Main_Page>.
- Bower, James M. Computational Modeling of Genetic and Biochemical Networks (Computational Molecular Biology). New York: M.I.T. PRESS, 2001.
- Braatsch, Stephan et al. "A single Flavoprotein AppA, Integrates Both Redox and Light Signals in Rhodobacter sphaeroides." Molecular Microbiology. Vol. 45.3 (2002): 827-836.
- Brown, Stanley B. et al. "Bile Pigment Synthesis in Plants: Incorporation of Haem into Phycocyanobilin and Phycocyanobiliproteins in Cyanidium Caldarium." Biochemistry. Vol. 194 (1981): 137-147.
- Dragnea, Vladimira et al. "Time-Resolved Spectroscopic Studies of the AppA Blue-Light Receptor BLUF Domain from Rhodobacter sphaeroides Biochemistry. Vol. 44 (2005): 15,978-15,985.
- "Entrez Nucleotide Search." National Center for Biotechnology Information. 2009. 6 June 2009 <http://www.ncbi.nlm.nih.gov/nuccore/49175990?from=3533887&to=3534606&report=gbwithparts>.
- Fowler, Gregory J. S. and C. Neil Hunter. "The Synthesis and Assembly of Functional High and Low Light LH2 Antenna Complexes from Rhodopseudomonas palustris in Rhodobacter sphaeroides." The Journal of Biological Chemistry. Vol. 271.23 (1996): 13,356-13,361.
- Gabrielsen, Mads et al. "Peripheral Complexes of Purple Bacteria." The Purple Phototropic Bacteria. (2009): 135-153.
- Jager, Andreas et al. "The AppA and PpsR Proteins from Rhodobacter sphaeroides Can Establish a Redox-Dependent Signal Chain but Fail to Transmit Blue-Light Signals in Other Bacteria." Journal of Bacteriology. Vol. 189.6 (2007): 2,274-2,282.
- Jones, M. R. et al. "Mutants of Rhodobacter sphaeroides Lacking One or More Pigment-Protein Complexes and Complementation with Reaction-Centre, LH1, and LH2 Genes." Molecular Microbiology. Vol. 6.9 (1992): 1,173-1,184.
- Kondo, Toshihiko et al. "Enhancement of Hydrogen Production by a Photosynthetic Bacterium Mutant with Reduced Pigment." Journal of Bioscience and Bioengineering. Vol. 93.2 (2002): 145-150.
- Lagarias, J. Clark. PCB from Spirulina. Personal Communication. June 2009.
- Laible, Philip D. "Foreign Gene Expression in Photosynthetic Bacteria." The Purple Phototropic Bacteria. (2009): 839-860.
- Lee, Jeong K. and Samuel Kaplan. "Transcriptional Regulation of puc Operon Expression in Rhodobacter sphaeroides." Journal of Biological Chemistry. Vol. 270.35 (1995): 20,453-20,458.
- Levskaya, Anselmetal et al. "Engineering Escherichia coli to See Light." Nature. Vol. 438 (2005): 441-442.
- "Life Sciences Catalog 2009-2010." National Diagnostics. (2009).
- Mizuno, Takeshi and Shoji Mizushima. "Characterization by Deletion and Localized Mutagenesis in Vitro of the Promoter Region of the Escherichia coli OmpC Gene and Importance of the Upstream DNA Domain in Positive Regulation by the OmpR Protein." Journal of Bacteriology. Vol. 168.1 (1986): 86-95.
- Moskvin, Oleg V. et al. "Transcriptome Analysis of the Rhodobacter sphaeroides PspR Regulon: PspR as a Master Regulator of Photosystem Development." Journal of Bacteriology. Vol. 187.6 (2005): 2,148-2,156.
- "NEBCutter V2.0." New England BioLabs. 2009. 6 June 2009 <http://tools.neb.com/NEBcutter2/index.php>.
- "OligoAnalyzer 3.1." Integrated DNA Technologies. 2009. 6 June 2009. <http://www.idtdna.com/analyzer/Applications/OligoAnalyzer/>
- Sambrook, Joseph and David W. Russell. Molecular Cloning: A Laboratory Manual. Vol. 1-3. New York: Cold Springs Harbor Laboratory Press, 2001.
- Shimizu-Sato, Sae et al. "A Light-Switchable Gene Promoter System." Nature Publishing Group. Vol. 20 (2002): 1,041-1044.
- Smith, Harry and M. Geoffrey Holmes. Techniquies in Photomorphogenesis. London: Academic Press, Inc., 1984.
- System modeling in cellular biology from concepts to nuts and bolts. Cambridge, MA: MIT P, 2006.
- Terry, Matthew J. "Biosynthesis and Analysis of Bilins." Heme, Chlorophyll, and Bilins: Methods and Protocols. (2002) 273-291.
- Wang, Wanneng et al. "Heterologous Synthesis and Assembly of Functional LHII Antenna Complexes from Rhodovulum sulfidophilum in Rhodobacter sphaeroides Mutant." Molecular Biology Reports. Vol. 12.3 (2008).
- Yakovlev, A. G. et al. "Light Control Over the Size of an Antenna Unit Building Block as an Efficient Strategy for Light Harvesting in Photosynthesis." Federation of European Biochemical Societies. (2002):129-132.
Back To Top

 "
"