Team:Aberdeen Scotland/parameters/invest 5
From 2009.igem.org
| Line 1: | Line 1: | ||
{{:Team:Aberdeen_Scotland/css}} | {{:Team:Aberdeen_Scotland/css}} | ||
{{:Team:Aberdeen_Scotland/header2}} | {{:Team:Aberdeen_Scotland/header2}} | ||
| - | + | = Final Solution = | |
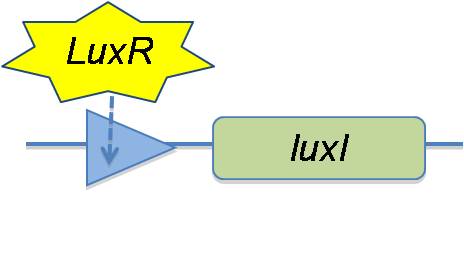
In the V.fischeri; where quorum sensing occurs naturally, the production of LuxI is on a very low level before quorum sensing occurs. This means that the HSL concentration is low in the cell, and thus is more likely to diffuse out of the cell rather than combining with LuxR. This results in a low level of HSL-LuxR which is never at a high enough concentration to activate the lux-box. Mimicking the natural system, we suggest putting the LuxI gene on a different promoter additionally regulating LuxR and that the promoter for the LuxI gene by a lux-box. | In the V.fischeri; where quorum sensing occurs naturally, the production of LuxI is on a very low level before quorum sensing occurs. This means that the HSL concentration is low in the cell, and thus is more likely to diffuse out of the cell rather than combining with LuxR. This results in a low level of HSL-LuxR which is never at a high enough concentration to activate the lux-box. Mimicking the natural system, we suggest putting the LuxI gene on a different promoter additionally regulating LuxR and that the promoter for the LuxI gene by a lux-box. | ||
| Line 7: | Line 7: | ||
[[Image:Qs_invest_4.jpg|center|300px]] | [[Image:Qs_invest_4.jpg|center|300px]] | ||
| - | In the natural system, it is suggested that the lux box constitutively transcripts LuxI at a rate of 1.5*10<sup>-4</sup> pops and a maximal transcription 2.0*10<sup>-3</sup> pops[1]. Using these values for our system including an amplifying loop for LuxI, we are left with: | + | The model we now look at is the final step in our progression. In the natural system, it is suggested that the lux box constitutively transcripts LuxI at a rate of 1.5*10<sup>-4</sup> pops and a maximal transcription 2.0*10<sup>-3</sup> pops[1]. Using these values for our system including an amplifying loop for LuxI, we are left with: |
[[Image:Qs_invest_5.jpg|center|700px]] | [[Image:Qs_invest_5.jpg|center|700px]] | ||
| Line 32: | Line 32: | ||
= The Amended Model = | = The Amended Model = | ||
| - | + | We next investigate the robustness of our system by performing a sensitivity analysis on K<sub>CI</sub> and K<sub>TetR</sub>. We see in the next few plots that the values of K<sub>CI</sub> and K<sub>TetR</sub> are very robust in this model: | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
[[Image:Amended Model 1.jpg|center|700px]] | [[Image:Amended Model 1.jpg|center|700px]] | ||
Revision as of 10:10, 17 August 2009
University of Aberdeen - Pico Plumber
Contents |
Final Solution
In the V.fischeri; where quorum sensing occurs naturally, the production of LuxI is on a very low level before quorum sensing occurs. This means that the HSL concentration is low in the cell, and thus is more likely to diffuse out of the cell rather than combining with LuxR. This results in a low level of HSL-LuxR which is never at a high enough concentration to activate the lux-box. Mimicking the natural system, we suggest putting the LuxI gene on a different promoter additionally regulating LuxR and that the promoter for the LuxI gene by a lux-box.
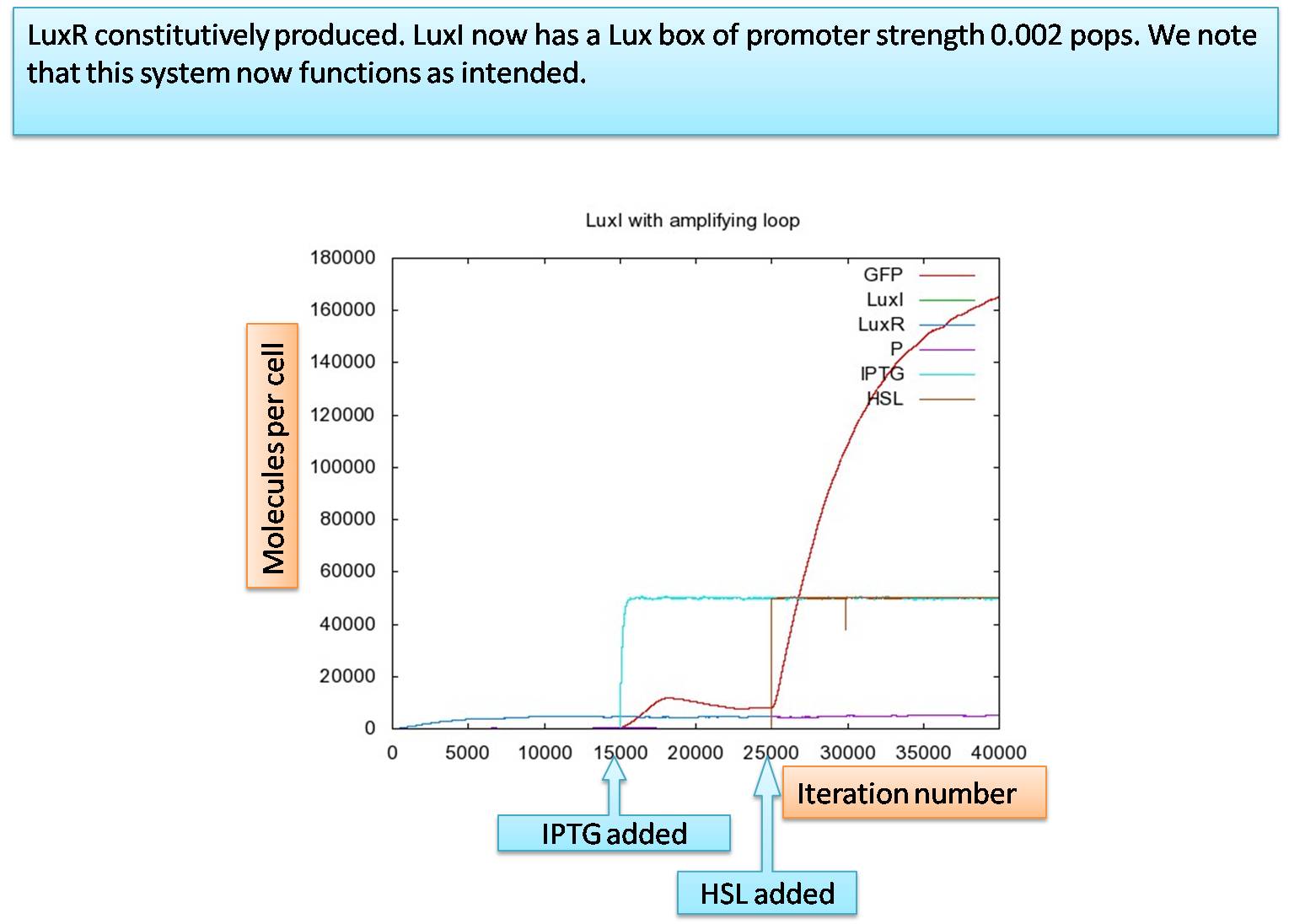
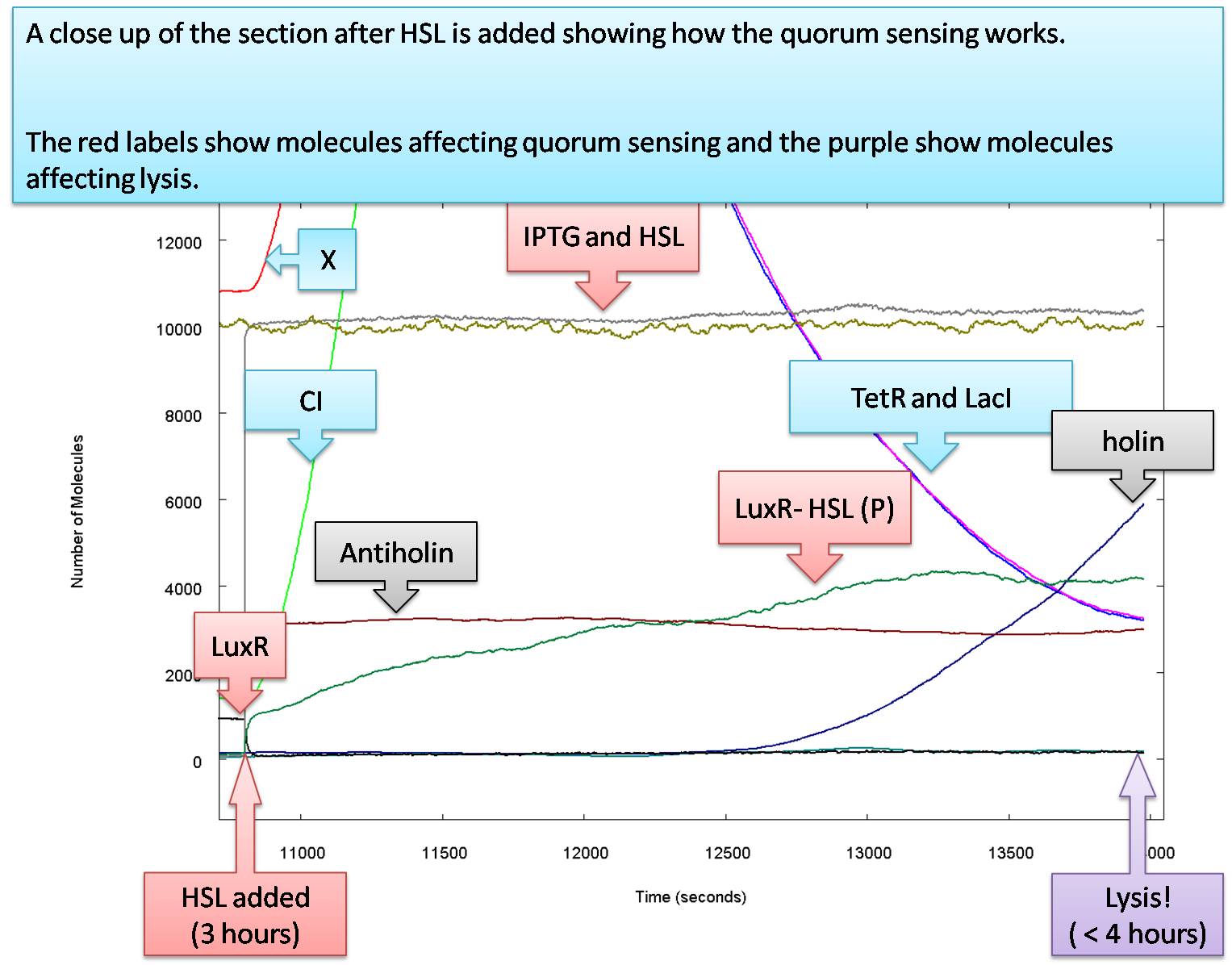
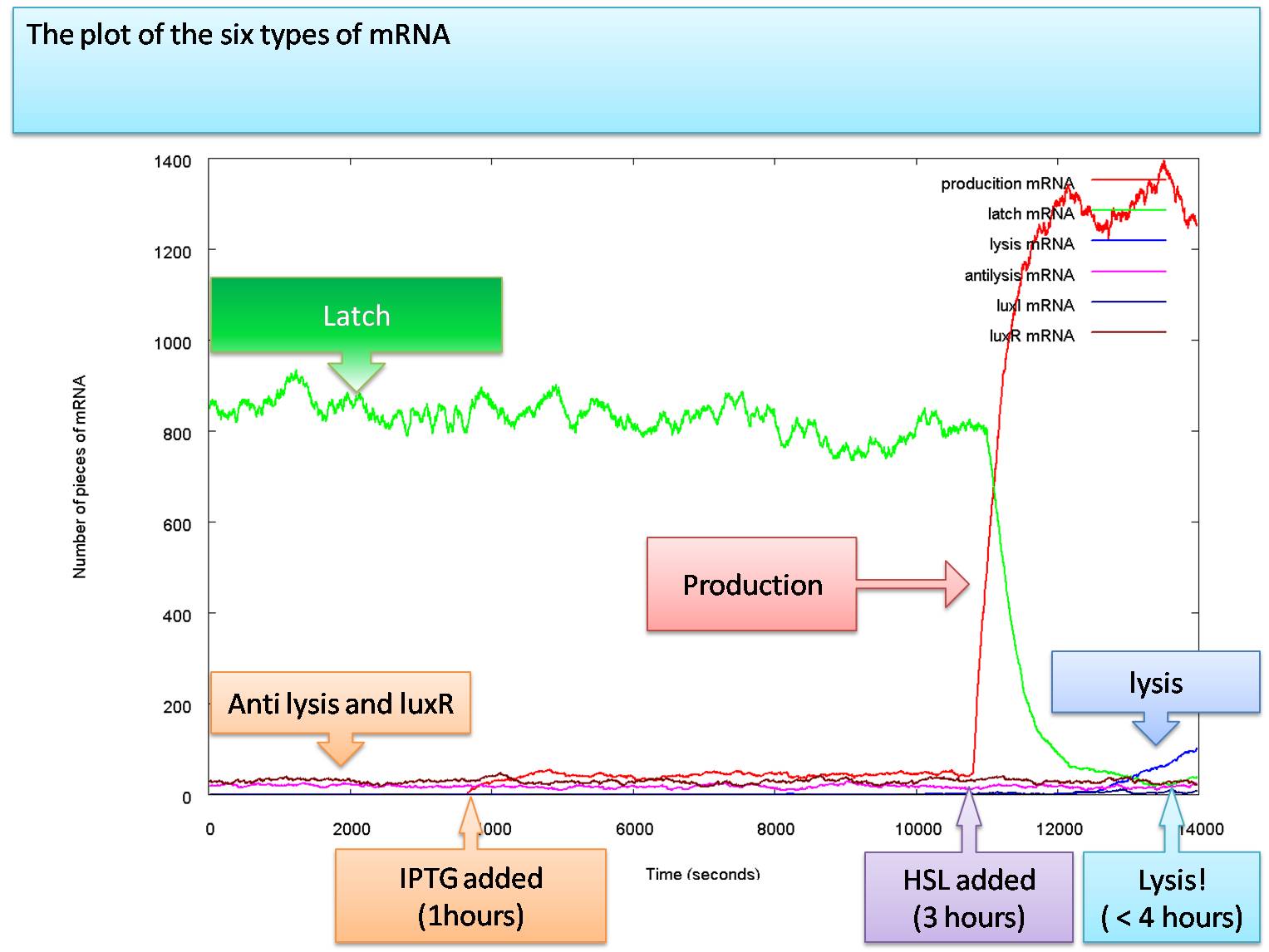
The model we now look at is the final step in our progression. In the natural system, it is suggested that the lux box constitutively transcripts LuxI at a rate of 1.5*10-4 pops and a maximal transcription 2.0*10-3 pops[1]. Using these values for our system including an amplifying loop for LuxI, we are left with:
The graph shows again an increase of GFP when IPTG enters the cell. However,in the beginning the amount of HSL produced in cell is less than the critical concentration required to activate the lux-box. When HSL is present, the increase in the production of GFP becomes even more pronounced. However, the idea behind the amplifying loop is a latch effect. The system should stay on whenever there is no or very weak HSL present form the outside. Testing this effect by turning off HSL from outside at the 35000 iteration step leaves us with the following graph:
In the presence of IPTG, GFP is produced at a lower level. When HSL enters the cell maximal production is reached. However, as soon as there is no HSL outside the cell, the amount of HSL-LuxR complex decreases indicating a too weak maximal production of LuxI. Thus, the maximal production rate for the promoter in front of LuxI is not strong enough.
However, by changing the maximal production of LuxI to 0.0025 pops it is clearly visible that we loose thehave AND-gate behaviour again.
The latching loop is reached when the constitutive production is set to 0.00001 pops and if the maximal production of the LuxI promoter is set to 0.0035 pops.
Concluding, while LuxR is produced constantly on a medium strong promoter we would need LuxI being formed on a very low constitutive level and in the presents of HSL the production needs to be amplified to a medium strong transcription level.
The Amended Model
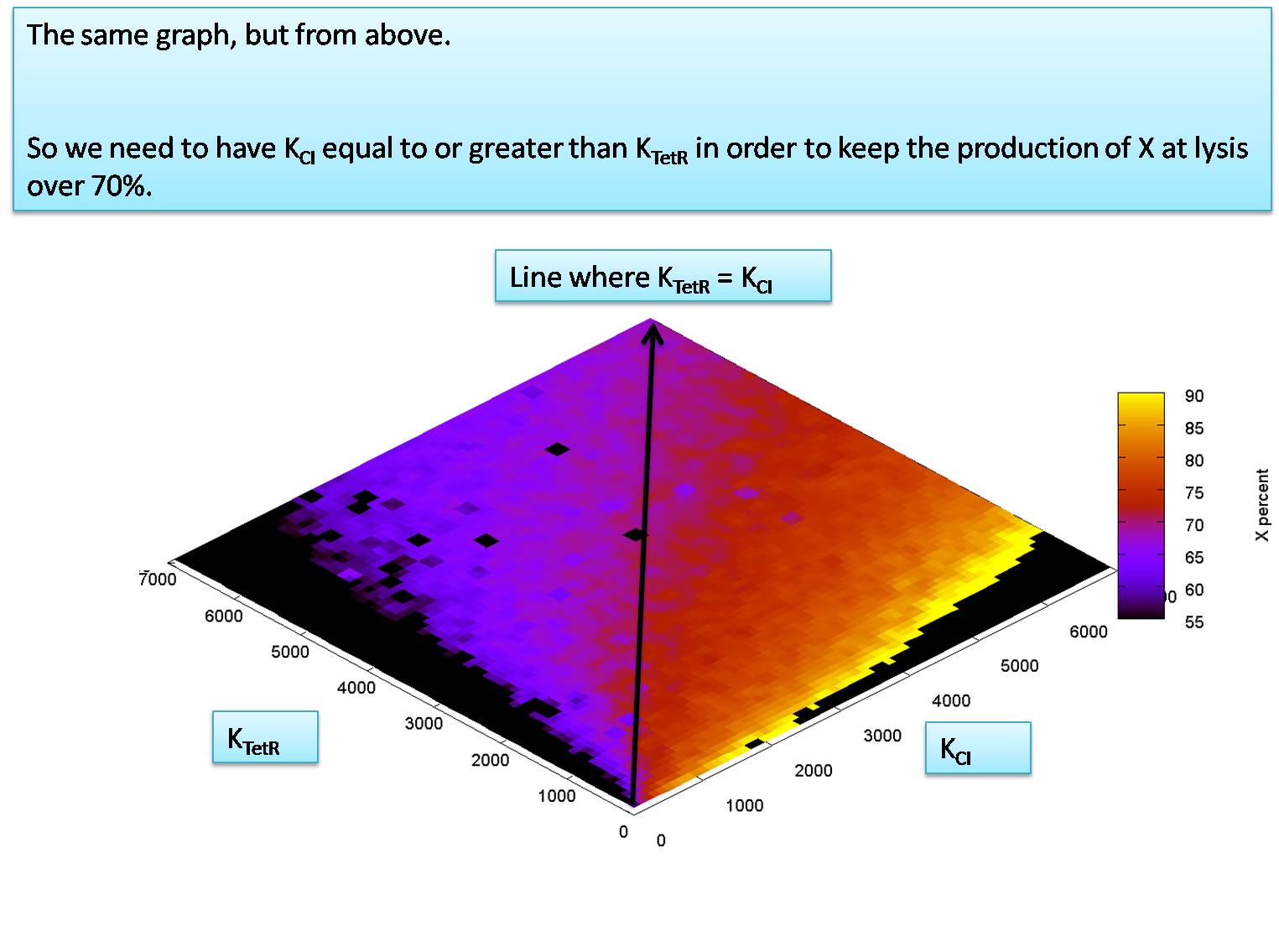
We next investigate the robustness of our system by performing a sensitivity analysis on KCI and KTetR. We see in the next few plots that the values of KCI and KTetR are very robust in this model:
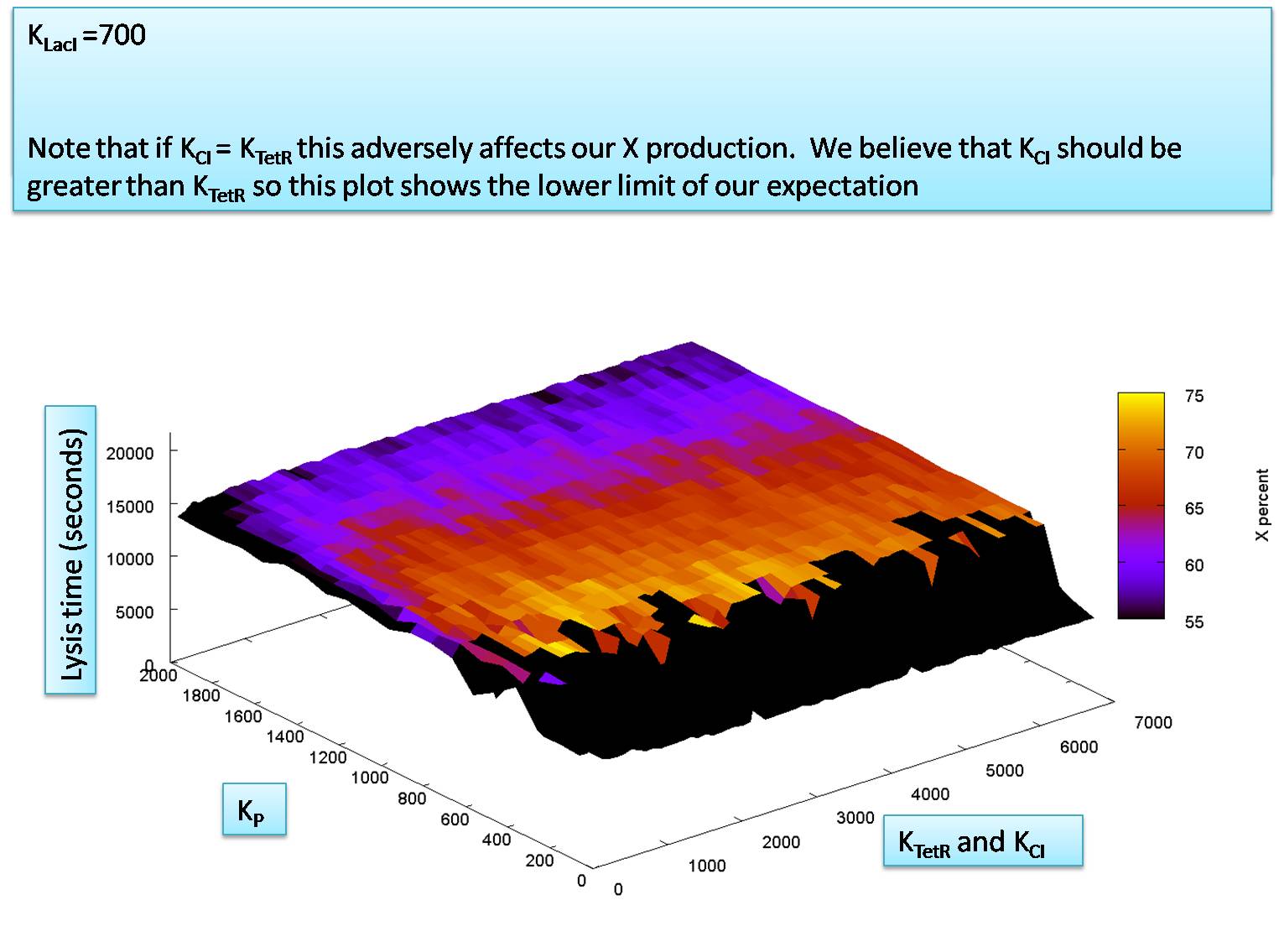
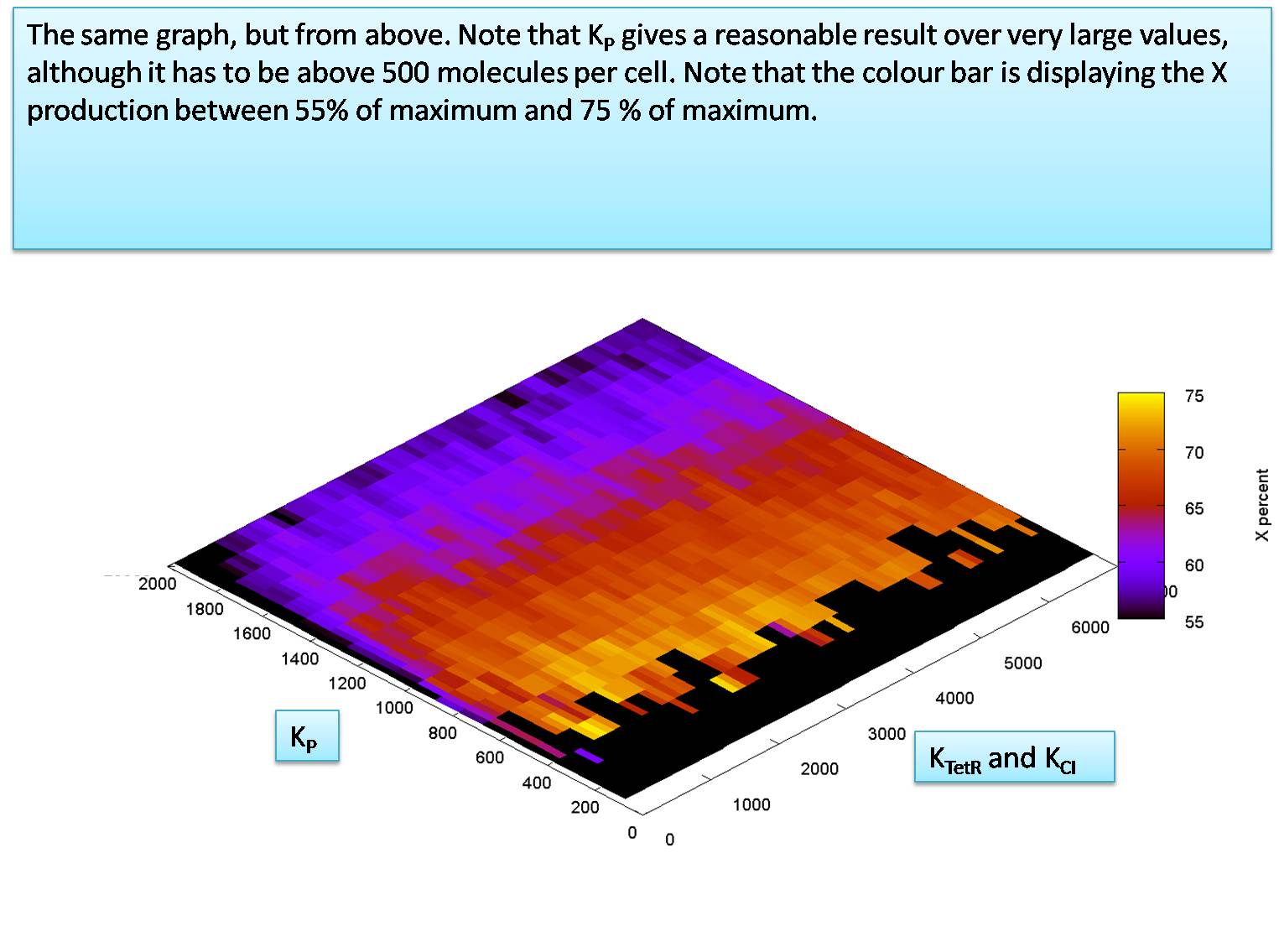
We know from the literature that KCI is expected to be greater than KTetR. This means that we can assume that in a worst case scenario KCI might be equal to KTetR. We use this worst case assumption now to check the relationship between KCI (= KtetR) VS. KP. This is what we find:
So our system is also very robust for different values of KP.
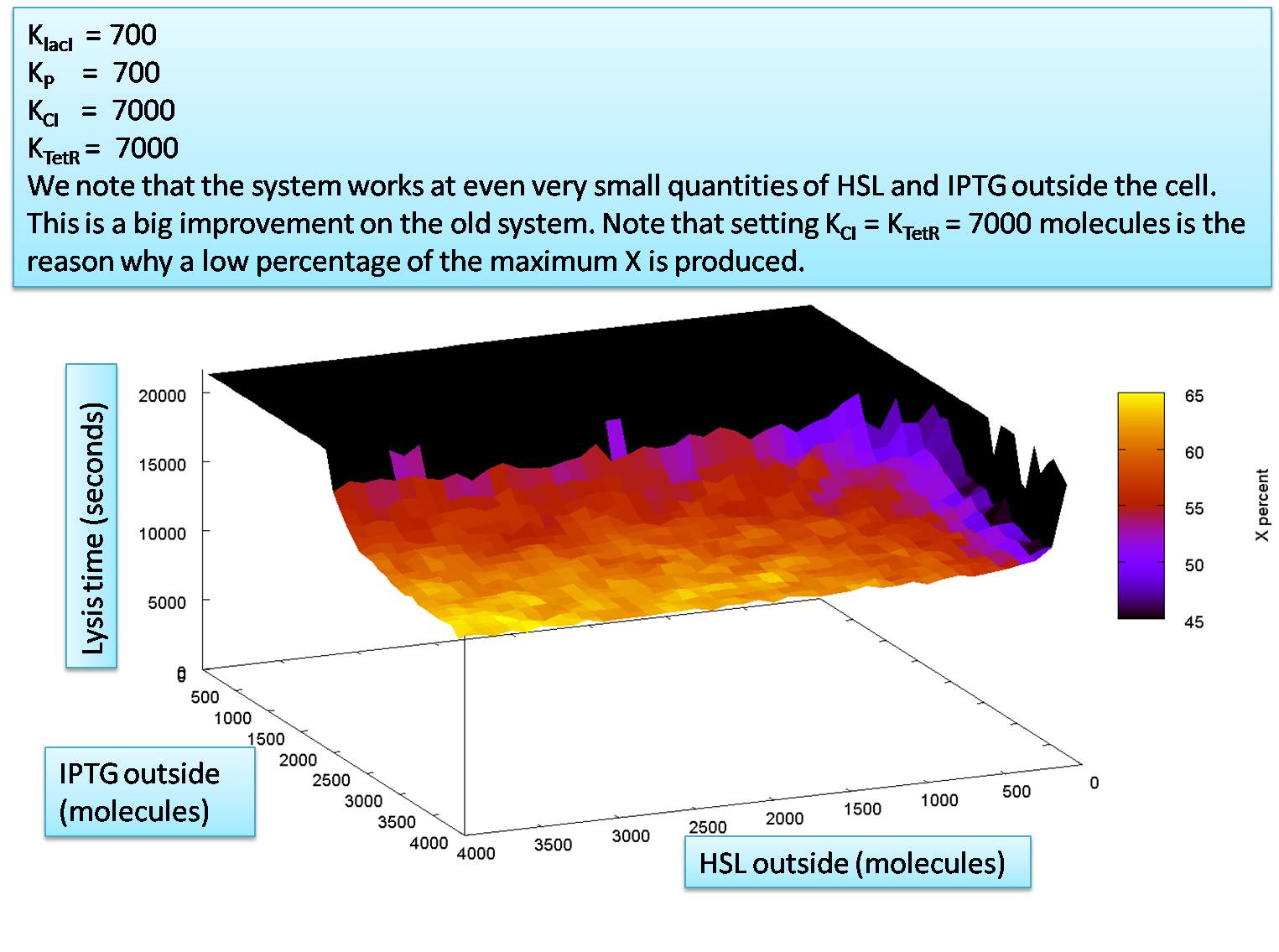
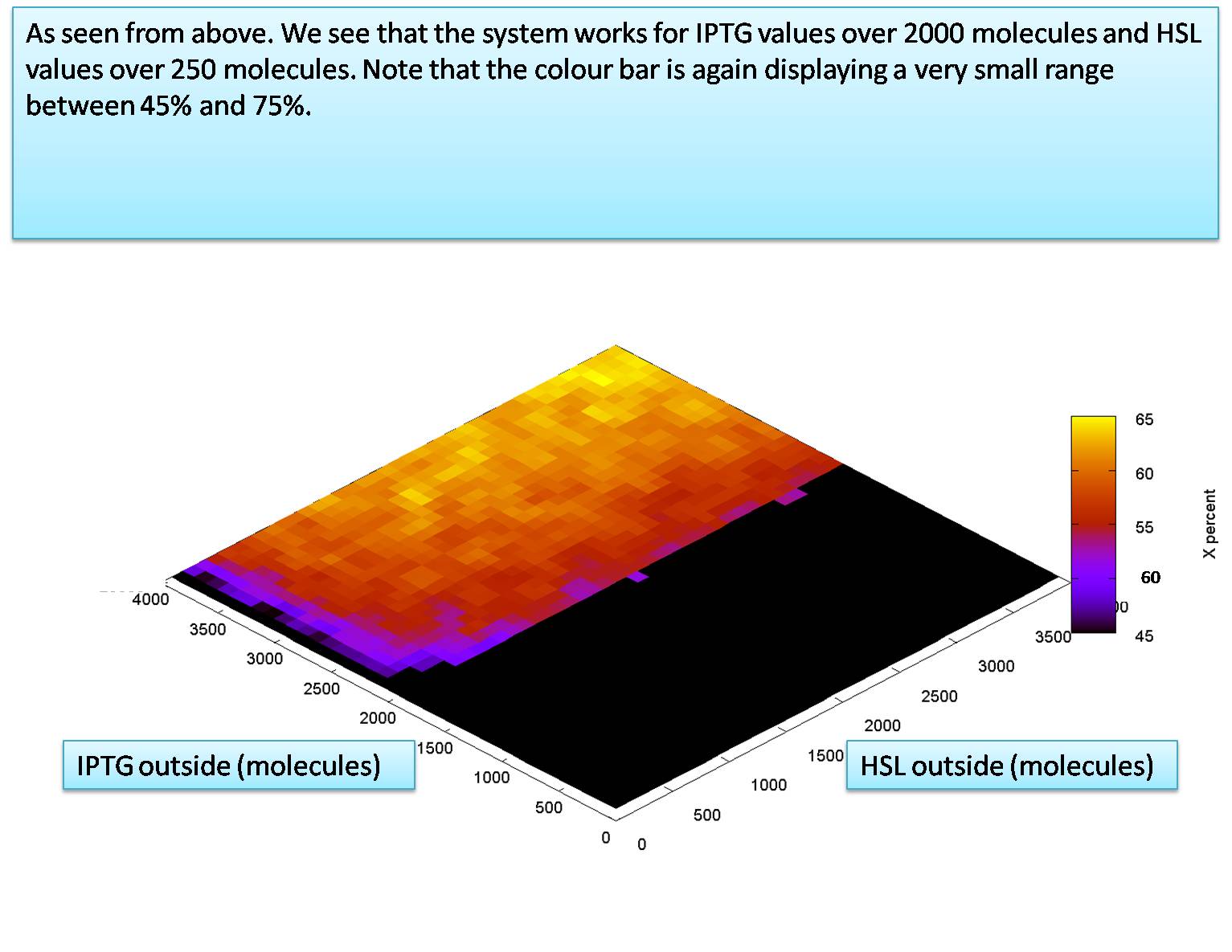
The required levels of HSL outside and IPTG outside
The only area left to explore is how the levels of the input molecules affect the output. We do this here in the same manner as was done for previous versions of the system.
Conclusion
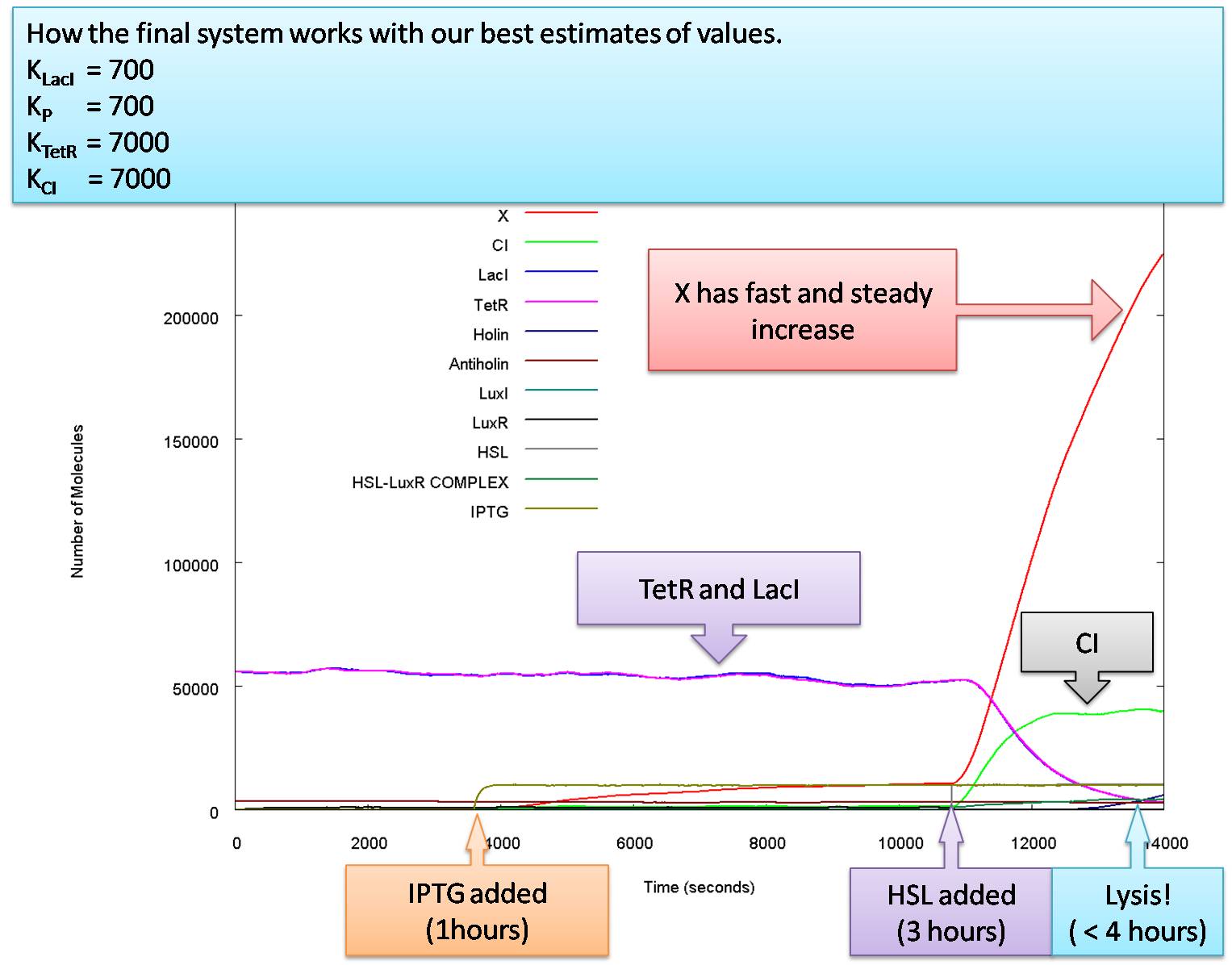
Here is how our system functions with the parameters we believe to be correct:
We can conclude that this system is very robust for K CI, KTetR, KlacI and KP. It can be activated by lower levels of HSL and IPGT than its predecessors and consistently produces large quantities of the glue molecule, molecule X. We have also shown that it is capable of latching behaviour for quorum sensing, given the right promoter strengths on the luxI feedback lux box.
Conclusion
The original design for the system triggers itself because of a higher production of LuxI than in the natural system. Having LuxI and LuxR both on the same amplifying loop results in a shortage of LuxR that can combine to HSL and start full production of the AND – gate as we can see here
In conclusion, changing the system to have LuxI on an amplifying loop will change the behaviour such that we have an AND-gate to trigger glue production and this is explored in detail here
Bibliography
(1) Goryachev. Systems analysis of a quorum sensing network: Design constraints imposed by the functional requirements, network topology and kinetic constants. BioSystems 2006;83(2-3 SPEC. ISS.):178.
(2) James. Luminescence control in the marine bacterium Vibrio fischeri: An analysis of the dynamics of lux regulation. J.Mol.Biol. 2000;296(4):1127.
(3) Ward JP. Mathematical modelling of quorum sensing in bacteria. IMA. 2001;18:263-292.
(4) Link to stochastic Equations
 "
"