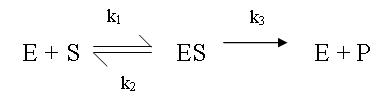Team:Imperial College London/Drylab/Enzyme/Analysis
From 2009.igem.org
(Difference between revisions)
| Line 12: | Line 12: | ||
**No allosteric regulations from either the product or the substrate. | **No allosteric regulations from either the product or the substrate. | ||
**There is no product inhibition of the enzyme. | **There is no product inhibition of the enzyme. | ||
| + | |||
*Total [E] does not change with regards to time. Enzyme will not be used up nor degraded over time, thus [E] at the beginning of the reaction will be the same as [E] at the end of reaction. | *Total [E] does not change with regards to time. Enzyme will not be used up nor degraded over time, thus [E] at the beginning of the reaction will be the same as [E] at the end of reaction. | ||
| Line 18: | Line 19: | ||
*For which | *For which | ||
| - | + | [[Image:ek1.jpg]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
, k1>>k2 | , k1>>k2 | ||
| - | + | <br> | |
<b>Law of mass action assumptions:</b> | <b>Law of mass action assumptions:</b> | ||
*Free diffusion | *Free diffusion | ||
| Line 37: | Line 30: | ||
<b>Michaelis-Menten assumption:</b> | <b>Michaelis-Menten assumption:</b> | ||
*There is a quasi-steady state of [ES] , ie: , during which the enzyme is used for the reaction. This means that the rate of formation of ES complex is equal to the rate of dissociation of ES complex. | *There is a quasi-steady state of [ES] , ie: , during which the enzyme is used for the reaction. This means that the rate of formation of ES complex is equal to the rate of dissociation of ES complex. | ||
| - | * | + | * |
| - | + | ||
| - | + | ||
| - | + | ||
*from the above assumptions, we know that | *from the above assumptions, we know that | ||
{{Imperial/09/TemplateBottom}} | {{Imperial/09/TemplateBottom}} | ||
Revision as of 04:42, 22 September 2009

- Overview
- The model
- Simulations
Assumptions
Enzymatic assumptions:
- The enzyme is specific only for the substrate and not for any other chemicals.
- Only one enzyme, our enzyme of interest is present and participating in the reaction.
- There is negligible formation of product without the enzyme.
- The rate of enzymatic activity remains constant over time because:
- There is no co-operativity of the system. Binding of substrate to one enzyme binding site doesn't influence the affinity or activity of an adjacent site.
- No allosteric regulations from either the product or the substrate.
- There is no product inhibition of the enzyme.
- Total [E] does not change with regards to time. Enzyme will not be used up nor degraded over time, thus [E] at the beginning of the reaction will be the same as [E] at the end of reaction.
- The reaction catalysed is irreversible
- [S] >> [E], such that the free concentration of substrate is very close to the concentration I added. This also ensures a constant substrate concentration throughout the assay. This allows easy determination of [E].
- For which
, k1>>k2
Law of mass action assumptions:
- Free diffusion
- Free unrestricted molecular motion
Michaelis-Menten assumption:
- There is a quasi-steady state of [ES] , ie: , during which the enzyme is used for the reaction. This means that the rate of formation of ES complex is equal to the rate of dissociation of ES complex.
- from the above assumptions, we know that
 "
"