Team:Imperial College London/Stomach
From 2009.igem.org
JamesField (Talk | contribs) (→Duodenum) |
JamesField (Talk | contribs) (→Human Digestive Proteases:) |
||
| Line 2: | Line 2: | ||
=<b>Human Digestive Proteases:</b>= | =<b>Human Digestive Proteases:</b>= | ||
| + | |||
| + | [[Image:Digestivesystem.jpg]] | ||
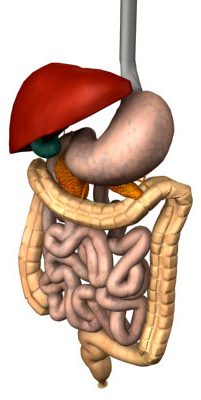
The stomach serves to break large proteins into peptides. These peptides are then broken down into amino acids once they enter the duodenum. | The stomach serves to break large proteins into peptides. These peptides are then broken down into amino acids once they enter the duodenum. | ||
Revision as of 12:38, 2 October 2009

Contents |
Human Digestive Proteases:
The stomach serves to break large proteins into peptides. These peptides are then broken down into amino acids once they enter the duodenum.
- Pepsin = stomach
- Trypsin = duodenum
- Chymotrypsin
- Carboxypeptidase
Stomach
The stomach is the first point at which polypeptides are broken down. The low pH of the stomach causes enzymes to denature, opening them up to attack from proteases such as pepsin.
Pepsin
Pepsin is a powerful protease that cleaves at the N-terminus after aromatic amino acids such as phenylalanine, tryptophan, and tyrosine. The optimum pH of pepsin is from 1.5 to 2, at a pH of above 5 denaturation occurs. This means that pepsin is only functional in the acidic environment of the stomach.
One of the enzymes that we are showcasing is phenylalanine hydroxylase (PAH) (as a treatment for PKU). If this enzyme was simply swallowed in an unencapsulated form it would be cleaved in 146 places by pepsin in the stomach.
Click on the perform button to see a simulated digestion of PAH by pepsin. Alternativly, paste in your own sequence to see if it would be a good candidate for encapsulation.
Duodenum
Endopeptidases (trypsin, chymotryopsin, elastase) Exopeptidases (carboxypeptidases A & B) Enteropeptidase
Trypsin
- Trypsin is found in the duodenum and serves to hydrolyse peptides into amino acids.
- Trypsin has an optimal operating pH of ~8 and an optimum temperature of 37°C.
- Trypsin breaks down the milk protein casein. For this reason, milk protein would be a good protein to use for secondary encapsulation.
Chymotryopsin
Elastase
Enterokinase
Carboxypeptidases A & B
 "
"