Team:Imperial College London/Temporal Control/Autoinduction
From 2009.igem.org
(→Temporal Control: Autoinduction) |
(→Temporal Control: Autoinduction) |
||
| Line 33: | Line 33: | ||
<br> | <br> | ||
<br> | <br> | ||
| - | <html><center><a href="https://2009.igem.org/Team:Imperial_College_London/Temporal_Control/Chemical_Induction"><img style="vertical-align:bottom;" width="20%" src="http://i691.photobucket.com/albums/vv271/dk806/II09_Drylabmaainimage1.png"></a><a href="https://2009.igem.org/Team:Imperial_College_London/Temporal_Control/Chemical_Induction"><img style="vertical-align:bottom;" width="20%" src="http://i691.photobucket.com/albums/vv271/dk806/II09_chemicalinduction.png"></a><a href="https://2009.igem.org/Team:Imperial_College_London/Temporal_Control/Thermoinduction "><img style="vertical-align:bottom;" width="20%" src="http://i691.photobucket.com/albums/vv271/dk806/ | + | <html><center><a href="https://2009.igem.org/Team:Imperial_College_London/Temporal_Control/Chemical_Induction"><img style="vertical-align:bottom;" width="20%" src="http://i691.photobucket.com/albums/vv271/dk806/II09_Drylabmaainimage1.png"></a><a href="https://2009.igem.org/Team:Imperial_College_London/Temporal_Control/Chemical_Induction"><img style="vertical-align:bottom;" width="20%" src="http://i691.photobucket.com/albums/vv271/dk806/II09_chemicalinduction.png"></a><a href="https://2009.igem.org/Team:Imperial_College_London/Temporal_Control/Thermoinduction "><img style="vertical-align:bottom;" width="20%" src="http://i691.photobucket.com/albums/vv271/dk806/II09_Thermoinduction1.png"></a><a href="https://2009.igem.org/Team:Imperial_College_London/Drylab"><img style="vertical-align:bottom;" width="20%" src="http://i691.photobucket.com/albums/vv271/dk806/II09_Wetlabmainimage9.png"></a><a href="https://2009.igem.org/Team:Imperial_College_London/Wetlab"><img style="vertical-align:bottom;" width="20%" src="http://i691.photobucket.com/albums/vv271/dk806/II09_Drylabmainimage6.png"></a></center></html> |
<html><table border="0" style="background-color:transparent;" width="100%"> | <html><table border="0" style="background-color:transparent;" width="100%"> | ||
Revision as of 11:19, 12 October 2009

Contents |
Autoinduction
Overview
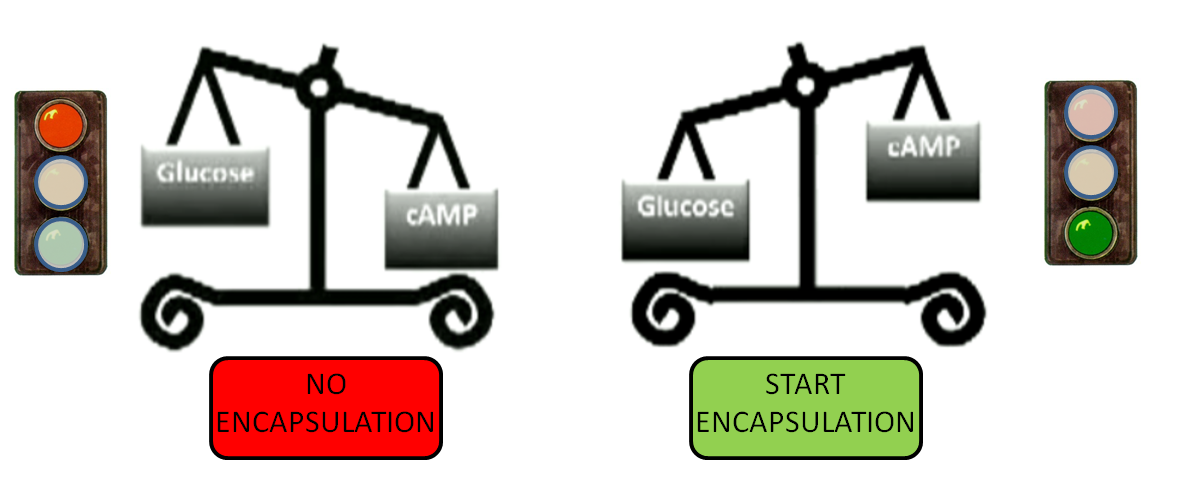
The CRP glucose repressible promoter triggers the encapuslation phase.
- When levels of glucose in the medium are high, cAMP levels are low and encapsulation is repressed
- When the levels of glucose in the medium are low, cAMP levels are high and encapsulation is induced.
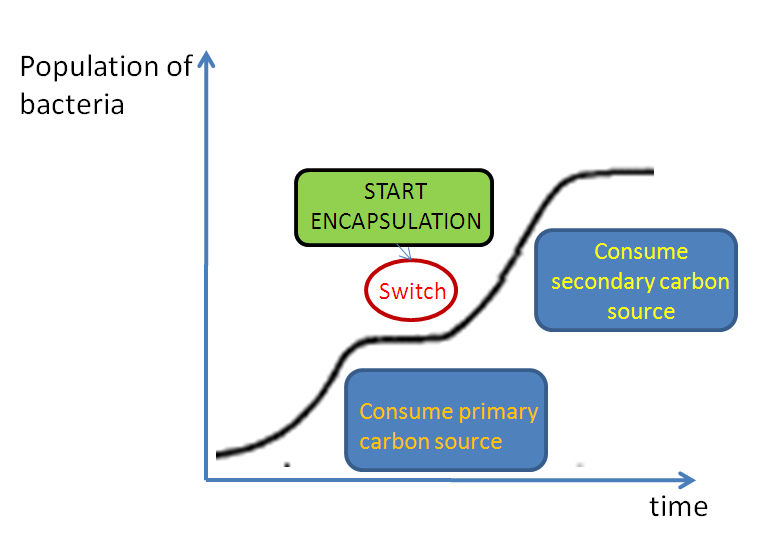
Diauxic growth
Starting the encapsulation and maintaining expression of colanic acid requires energy in the form of carbon sources. Bacteria use up a primary source to grow and trigger encapsulation. Glucose here is the primary source, which in unison with cAMP, is responsible for switching on the CRP promoter and the encapsulation process.A secondary carbon source is also needed to maintain the system once glucose is used up. This phenomenon of switching from a primary source to a secondary source is known as a diauxie.
This switching process occurs automatically, hence we have called it "autoinduction".
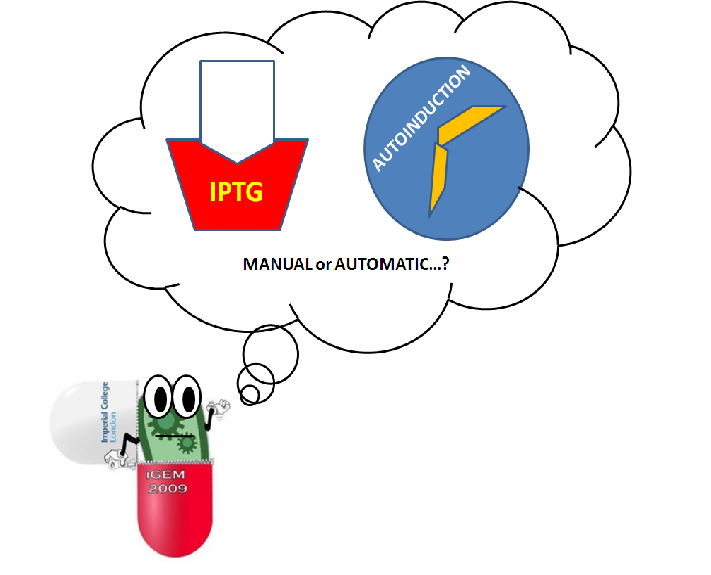
Comparison with IPTG induction
Conventional induction relies on adding small amounts of IPTG into a cell culture. Autoinduction media instead rely on culture conditions for induction. When glucose levels are low due to cellular consumption, the CRP promoter in cells is activated, and induction occurs automatically. Compared to IPTG, current methods of autoinduction allow for higher density without the need to monitor cell growth [1].
For the autoinduction media, it may include one or more carbon sources selected from the group consisting of glucose, lactose, glycerol, rhamnose, arabinose, succinate, fumarate, malate, citrate, acetate, maltose and sorbitol. The secondary carbon source involves the import and consumption of lactose and glycerol [2].
Relevance to our project
We are not using autoinduction for ensuring high protein density as it was originally intended to. Instead, we are using autoinduction for ensuring a time delay before encapsulation occurred(Glucose time delay assay). Therefore, we not only have a simplistic cell growth model to take into account. Instead, we will also have to take note of the cell growth and its changes when protein production is induced. The time duration will no longer be purely overnight, but rather it will be about a whole day (growth to high cell density plus around 6 hours of protein production).
We have tried to optimize the growth conditions. We have decided that the nitrogen source 0.2% Casamino acids is necessary for cell growth [modifying of Endy protocol, Larry], while glycerol should be omitted as it will be taken up similar to a secondary carbon source. Preliminary experimental data has shown that with 0.05% glucose, the switch point occurs after about 7 hours.
References
[1]Unattended high-density cell growth and induction of protein expression with the Overnight Express™ Autoinduction System Anthony Grabski, Mark Mehler, and Don Drott — Novagen [2]Studier et al. Protein production by auto-induction in high-density shaking cultures. Expr Purif. 2005 May;41(1):207-34.
Temporal Control: Autoinduction






 "
"