Team:Lethbridge/Project
From 2009.igem.org
(→Project Details) |
(→Project Details) |
||
| Line 295: | Line 295: | ||
== Project Details== | == Project Details== | ||
| - | [[Image:YFP_CFP_Lumazine.png|right| | + | [[Image:YFP_CFP_Lumazine.png|right|275px]] |
Many of the metabolic processes in eukaryotic cells are localized to various subcellular compartments. For example, transcription of mRNA from DNA is localized in the nucleus; post-translational modification and folding of proteins occurs in the endoplasmic reticulum (Voet et al., 2004). This essentially segregates and regulates these processes. However, bacteria lack distinct organelles, and as such, many metabolic processes intermingle. Engineering an artificial organelle, capable of containing metabolic proteins, in bacteria would thus represent a fundamental advance in biotechnology. Towards this end, the University of Lethbridge iGEM team is working towards producing microcompartments from the lumazine synthase gene, which forms 60 subunit icosahedral capsids (Seebeck et al., 2006). | Many of the metabolic processes in eukaryotic cells are localized to various subcellular compartments. For example, transcription of mRNA from DNA is localized in the nucleus; post-translational modification and folding of proteins occurs in the endoplasmic reticulum (Voet et al., 2004). This essentially segregates and regulates these processes. However, bacteria lack distinct organelles, and as such, many metabolic processes intermingle. Engineering an artificial organelle, capable of containing metabolic proteins, in bacteria would thus represent a fundamental advance in biotechnology. Towards this end, the University of Lethbridge iGEM team is working towards producing microcompartments from the lumazine synthase gene, which forms 60 subunit icosahedral capsids (Seebeck et al., 2006). | ||
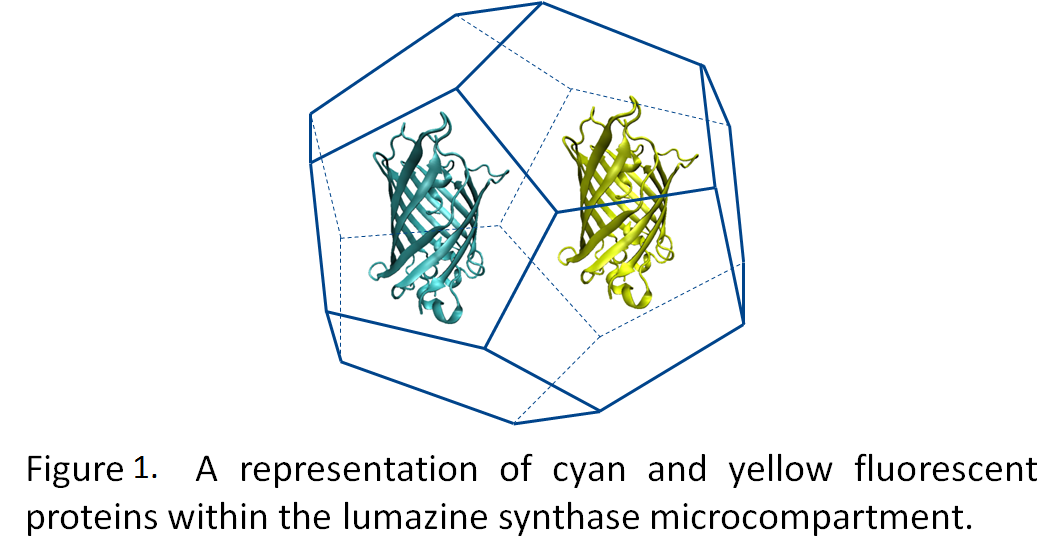
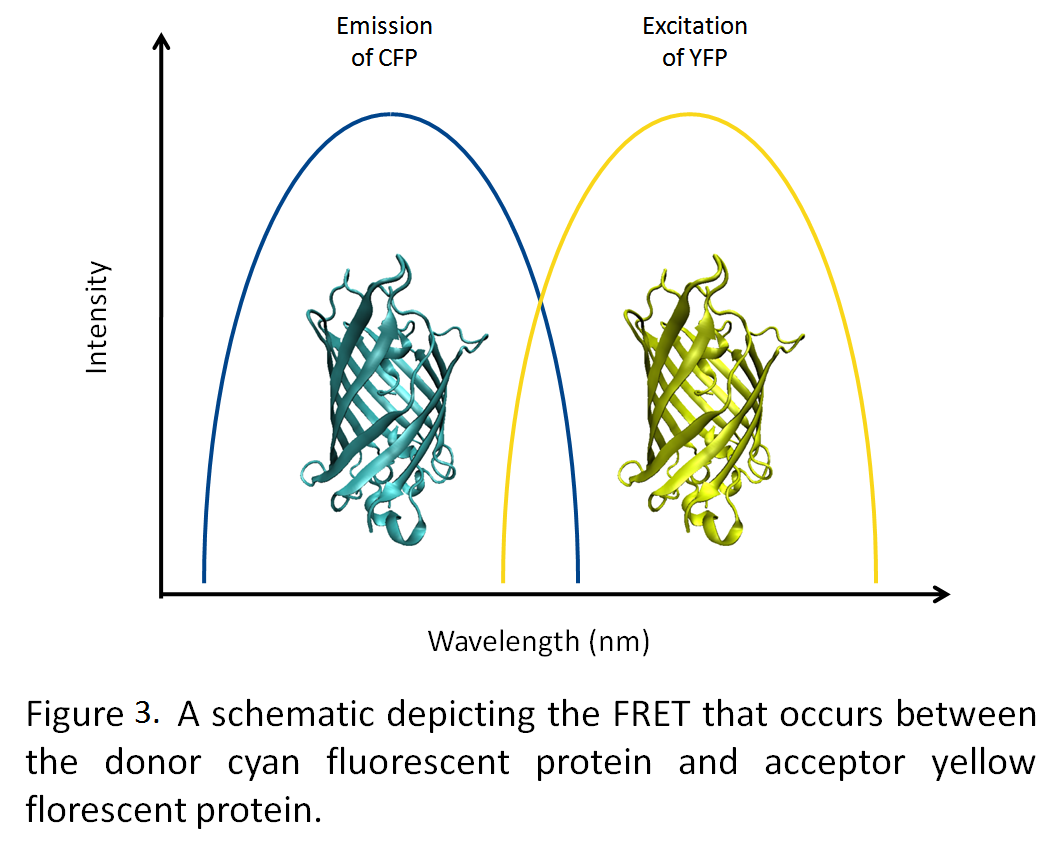
| - | [[Image:YFP_CFP_fret.png|left| | + | [[Image:YFP_CFP_fret.png|left|275px]]By generating a highly negative interior to the microcompartment, we hope to be able to target fluorescent proteins tagged with positively charged termini to the interior of the microcompartment (Figure 1). Utilizing fluorescence resonance energy transfer (FRET) between cyan and yellow fluorescent proteins, we will demonstrate the co-localization of these two proteins into the microcompartment (Figure 2). |
For the future application of this technology we are working towards the targeting of proteins from the photosynthetic pathway into the microcompartment to optimize a biological photosynthetic fuel cell. The segregation of proteins and metabolites increases the efficiency of cellular processes and following this theme, we are working towards creating uniform nanoparticles through the segregation of Mms6 protein within Escherichia coli (Figure 3) | For the future application of this technology we are working towards the targeting of proteins from the photosynthetic pathway into the microcompartment to optimize a biological photosynthetic fuel cell. The segregation of proteins and metabolites increases the efficiency of cellular processes and following this theme, we are working towards creating uniform nanoparticles through the segregation of Mms6 protein within Escherichia coli (Figure 3) | ||
| - | [[Image:Nanoparticles.png|right| | + | [[Image:Nanoparticles.png|right|300px]] |
(Prozorov et al., 2007). | (Prozorov et al., 2007). | ||
Revision as of 16:28, 21 October 2009
- Home
- Team
- Project
- Ethics
- Notebook
- Meetings
- Modeling
- Parts
- Collaboration
- Judging
- Extra Information on SynBio
Project Details
Many of the metabolic processes in eukaryotic cells are localized to various subcellular compartments. For example, transcription of mRNA from DNA is localized in the nucleus; post-translational modification and folding of proteins occurs in the endoplasmic reticulum (Voet et al., 2004). This essentially segregates and regulates these processes. However, bacteria lack distinct organelles, and as such, many metabolic processes intermingle. Engineering an artificial organelle, capable of containing metabolic proteins, in bacteria would thus represent a fundamental advance in biotechnology. Towards this end, the University of Lethbridge iGEM team is working towards producing microcompartments from the lumazine synthase gene, which forms 60 subunit icosahedral capsids (Seebeck et al., 2006).
By generating a highly negative interior to the microcompartment, we hope to be able to target fluorescent proteins tagged with positively charged termini to the interior of the microcompartment (Figure 1). Utilizing fluorescence resonance energy transfer (FRET) between cyan and yellow fluorescent proteins, we will demonstrate the co-localization of these two proteins into the microcompartment (Figure 2).For the future application of this technology we are working towards the targeting of proteins from the photosynthetic pathway into the microcompartment to optimize a biological photosynthetic fuel cell. The segregation of proteins and metabolites increases the efficiency of cellular processes and following this theme, we are working towards creating uniform nanoparticles through the segregation of Mms6 protein within Escherichia coli (Figure 3)
(Prozorov et al., 2007).
The Experiments
Results
 "
"