Team:British Columbia/LockandKey
From 2009.igem.org
(→Phase I:) |
m (→Phase II: Validating Lock and Key function) |
||
| Line 27: | Line 27: | ||
===Phase II: Validating Lock and Key function=== | ===Phase II: Validating Lock and Key function=== | ||
| - | + | First we attempted to demonstrate that the lock functioned as expected, i.e. that its presence upstream of a reporter gene prevented the gene from being expressed. | |
====Methods:==== | ====Methods:==== | ||
* assembly of lock, control lock, and control RBS with a constitutive promoter and several different reporter genes | * assembly of lock, control lock, and control RBS with a constitutive promoter and several different reporter genes | ||
Revision as of 02:36, 22 October 2009
Home Team Traffic Light Sensor Lock&Key Jammer [http://partsregistry.org/cgi/partsdb/pgroup.cgi?pgroup=iGEM2009&group=British_Columbia Parts] Safety Sponsors Notebook Bibliography
Contents |
Lock and Key Subproject
SECTION 1: STATEMENT OF RESEARCH PROBLEM
Description of Biological Phenomenon and General Aims of Project
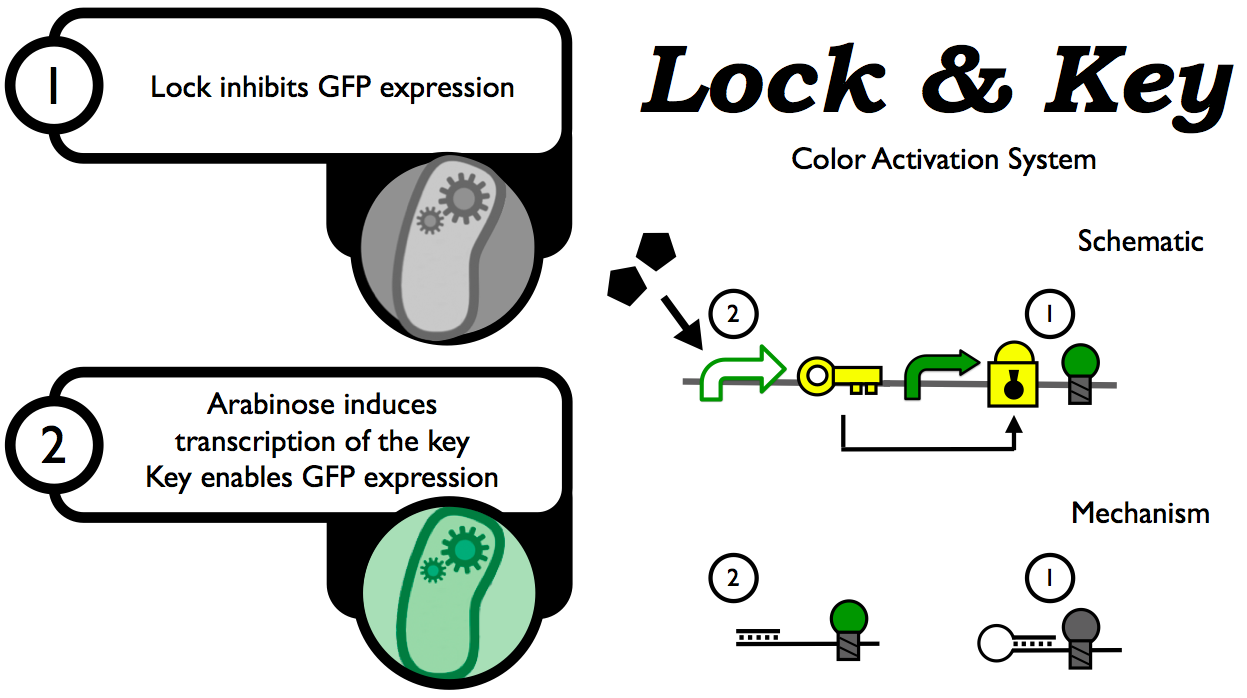
In 2004, Isaacs et al. described a system for regulating gene expression at the RNA level. By inserting an upstream "lock" sequence complementary to the RBS, the RBS becomes unavailable for translation by ribosomes, preventing expression of the target gene. A separately transcribed mRNA, the "key", can interact with a loop region in the locked transcript and preferentially bind the lock sequence, returning the gene to basal expression levels.
Our intention was to adapt this lock and key system to the BioBrick standard, turning it into a modular part that can be used to restrict or allow the transcription of any other part. This would allow us to introduce Boolean logic to our system.
SECTION 2: RESEARCH OBJECTIVES AND SPECIFIC METHODS
Phase I: Creating the Lock and Key as Biobrick parts
Our initial goal was to create a version of Isaacs et al.'s lock and key that was compatible with the BioBrick standard; the main requirements were to add the BioBrick standard prefix and suffix, and remove internal cut sites shared by assembly restriction enzymes (EXSP), while maintaining the locking/unlocking capacity of the various parts. To begin with, we used the lock (aka cr12), key (aka ta12), and control lock (a sequence of similar length to the lock, but comprising random bases that should not have any binding affinity to the RBS) sequences reported in Isaac et al.'s paper.
Methods:
- Modification of the sequence described in the Isaacs et al. paper in order to prevent unwanted binding of BioBrick "scars"
- Adaptation of the lock, control lock, and key to the BioBrick standard
- Construct the biobrick plasmid by:
- synthesis of oligos from IDT
- second-strand synthesis via PCR
- digestion of prefix/suffix
- ligation into construction plasmid
Phase II: Validating Lock and Key function
First we attempted to demonstrate that the lock functioned as expected, i.e. that its presence upstream of a reporter gene prevented the gene from being expressed.
Methods:
- assembly of lock, control lock, and control RBS with a constitutive promoter and several different reporter genes
- qualitative evaluation (by eye: fluorescent reporters and lacZ)
- quantitative evaluation (beta-galactosidase timecourse)
- inoculated 3 separate tubes wih 3 different streak plate colonies for each of control and lock overnight into LB-Chlor. 16 hours after, diluted to A600=0.1 in 20mL LB-Chlor + IPTG, set this to t = 0.
- Every hour, measured OD600 of culture, then removed 2x50uL and kept in permeabilization solution (stable for several hours)
- Every four hours, added substrate to samples, allowed to develop until visibly yellow, then added stop solution. Samples were then spun down (to avoid particulate matter) and A420 measured on Haynes lab spectrophotometer.
SECTION 3: SUMMARY OF RESULTS
Phase I:
Using an RNA secondary structure prediction program, we found that the Biobrick scars were complementary to each other, raising the possibility that they would prevent the key from unlocking the lock. The 5' and 3' flanking sequences of the lock were shortened by 6 nucleotides and the sequence was altered, respectively, to mitigate this problem (see figure 1). One nucleotide was altered so as to keep the number of bulges at three.

Figure 1. Proposed modifications to Isaacs Lock to render it compatible with the BioBrick standard.
Isaacs’: GAATTCTACCATTCACCTCTTGGATTTGGGTATTAAAGAGGAGAAAGGUACC UBC iGEM:TACTAGAGAAGCGCAGTAATTCACCTCTTGGATTTGGGTATCAAAGAGGAGAAATACTAG
Figure 2: Sequence alignment of lock sequence pre- and post-modification, with changes highlighted.
On August 1st, Eric reported the successful insertion of the lock, control lock, and key into a construction plasmid (sequence verified: Order # 22108).
Phase II:
After assembling the lock, control lock, and a control RBS (B0034) with our strongest constitutive promoter (J23100) and various reporter genes, we observed:
- no naked-eye-visible fluorescence from either lock or control lock assembled with RFP, YFP, or GFP_LVA (vs. visible fluorescence from the control RBS with all of these genes)
- blue colonies on IPTG and X-gal from both the lock and the control lock assembled with lacZ
Hypothesizing that the lock might exhibit a delayed rate of lacZ gene expression, even if it reached the same eventual maximum as the control, a timecourse was performed to measure lacZ expression by quantifying beta-galactosidase activity on a colorigenic substrate. However, no significant difference was observed between the lock and control over 16 hours of induction (see figure 3).

Figure 3. Beta-galactosidase activity of lock and control cultures over time. Each point represents the mean of 3 biological replicates (which are in turn the mean of two experimental replicates). Miller Units represent A420 absorbance normalized to OD600.
SECTION 4: SUMMARY OF PROPOSED FUTURE EXPERIMENTS AND TIMELINE OF COMPLETION
With the results thus far, there is no conclusive evidence to suggest that the lock as designed is performing its expected role, i.e. repressing gene expression. It is within the realm of possibility that the modifications necessary for adapting the lock to a Biobrick have also abrogated the lock's (quite sensitive) activity. Should this be the case, options are somewhat limited in the time remaining; we could attempt mutagenesis of the current lock with the hopes of introducing locking activity, but we would essentially be making a not-very-educated guess as to what might work (Even Isaacs et al. were unable to improve the lock's efficacy via rational design).
Before deciding whether to abandon the lock + key project or not, I believe it would be beneficial to sequence verify the constructs used in the timecourse experiment (they are streak colonies of transformants of a sequence verified miniprep, so should be correct, but there is the possibility of point mutations/mixups with the preps). Should the sequences be correct, here are some possible next steps:
1. Mutagenize the lock back into the Isaacs original form,
- Rationale: We believe that our modifications are what made the lock stop working, we will hope that the complementarity of the scars has a negligible effect on activity
- Time needed: a lot of time - at least 2 weeks
- Possible issues: rationale is doubtful/disingenuous - the effect of the scars would be to prevent unlocking by the key, which we probably won’t be able to test in time for the jamboree even if we do get what seems to be a working lock
2. Mutagenize the lock with somewhat randomized primers, using lacZ to screen
- Rationale: We believe that our modifications have made the lock stop working, but we don’t know how to make it work again; let chance discover it for us
- Time needed: a lot of time - at least 2 weeks
- Possible issues: will take a lot of time, no guarantee that any lock we discover this way is actually unlockable
3. Attempt a more thorough timecourse, (increase duration/# timepoints, keep reaction lengths closer together)
- Rationale: we believe that the lock is working but we just haven't measured it accurately
- Time needed: approx. 2-3 days work, with 1-2 people helping
- Possible issues: doubt that the lock is actually working
4. Try using a different reporter (full lacZ gene, fluorescent reporters + flow cytometry, others?)
- Rationale: we believe that the lock is working but we just haven't measured it accurately
- Time needed: 1 assembly [approx. half a week], then evaluation (time needed depends on reporter used)
- Possible issues: will probably take longer than we expect, doubt that the lock is actually working
 "
"