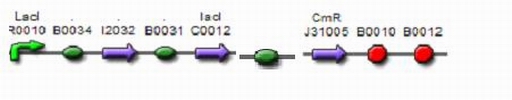
Protocols
From 2009.igem.org
| Line 3: | Line 3: | ||
|[[Home]|[[Project]]|[[Team]] ||bgcolor="yellow"| '''[[Protocols]]'''|| [[Parts]] | [[Modeling]] |[[Gallery]] |[[Notebook]]|[[Ethics]]| | |[[Home]|[[Project]]|[[Team]] ||bgcolor="yellow"| '''[[Protocols]]'''|| [[Parts]] | [[Modeling]] |[[Gallery]] |[[Notebook]]|[[Ethics]]| | ||
|} | |} | ||
| + | |||
| + | == Project == | ||
| + | |||
| + | === Project 1 === | ||
| + | |||
| + | *'''1, the key enzyme for ethanol production''' | ||
| + | There is a pathway both in Saccharomyces Cerevisiae and Zymomonas.Mobilis. | ||
| + | |||
| + | pyruvic acid------> carbon dioxide + acetaldehyde ------>ethanol | ||
| + | |||
| + | *The enzyme which catalysis the first reaction is pyruvic decarboxylase (PDC)and the other enzyme catalyzing the following step is alcohol dehydrogenase(ADH) | ||
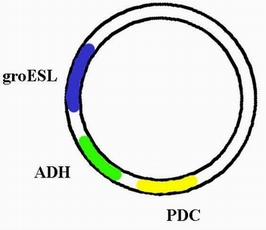
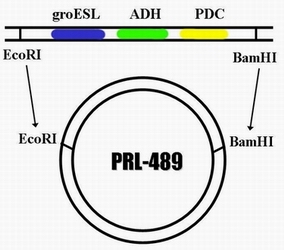
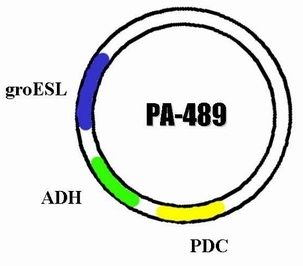
| + | In order to integrate this two enzyme into the genome of cyanobacteria so that it can carry on the ethanol producing pathway during the process of photosynthesis, we decided to construct a plasmid through which the ADH and PDC gene are able to express in cyanobacteria. The basic carrier we use is PRL -489. So after integrating the sequence of ADH and PDC on it , we name the new plasmid PA-489. | ||
| + | |||
| + | *'''2. The construction of PA-489''' | ||
| + | To reach this goal we designed two plans and in each plan two groups are operated parallelly. | ||
| + | |||
| + | '''Plan A''' | ||
| + | |||
| + | '''*Group 1:''' | ||
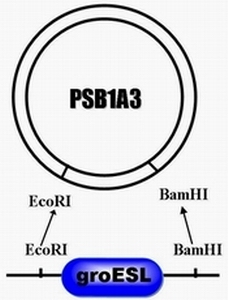
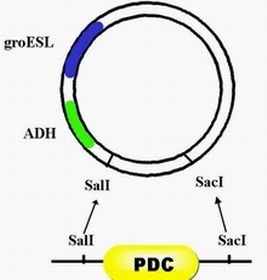
| + | #step 1, there are two enzyme restriction sites on the plasmid PRL-489 which is EColR1 and BamH1,through double digests, excising this piece of segment and replace it with the promoter of groESL ,namely heat shock promoter. | ||
| + | [[Image:tu4.jpg]][[Image:tu5.jpg]] | ||
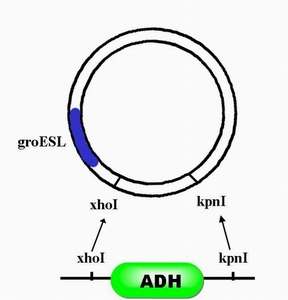
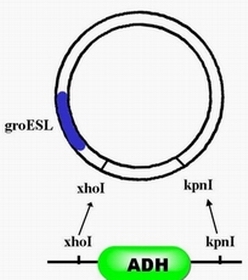
| + | #Step 2, with enzyme restriction sites ,xhoI and kpnI,ADH can be inserted after promoter groESL. | ||
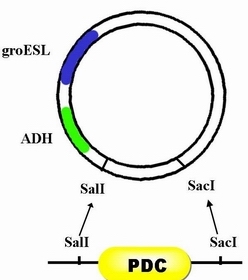
| + | #Step 3, with enzyme restriction sites, SalI and SacI, PDC can be inserted after ADH. | ||
| + | [[Image:tu6.jpg]][[Image:tu7.jpg]][[Image:tu8.jpg]] | ||
| + | *After the three steps the constructed result is our target plasmid. | ||
| + | |||
| + | '''Group 2:''' | ||
| + | All the manipulations are the same with group 1, the only difference is that we use rubisco (rbc) promoter. | ||
| + | |||
| + | '''Plan B''' | ||
| + | *In this plan we utilize another technique called sub-clone. In molecular biology, subcloning is a technique used to move a particular gene of interest from a parent vector to a destination vector in order to further study its functionality. | ||
| + | [[Image:tu9.jpg]] | ||
| + | We need two carriers to construct PA-489 and also do it parallelly. | ||
| + | |||
| + | '''Group 1:''' | ||
| + | *step 1, we choose PSB1A3 as the basic model, with the same enzyme restriction sites EcoR1 and BamH1, heat shock promoter can be subcloned on the plasmid. | ||
| + | [[Image:tu10.jpg]][[Image:tu11.jpg]] | ||
| + | *Step 2, in the same way we subclone ADC after groESL. | ||
| + | *Step 3, with the restriction sites of SalI and SacI, we subclone PDC flowing to ADC. | ||
| + | [[Image:tu12.jpg]][[Image:tu13.jpg]][[Image:tu4.jpg]] | ||
| + | *Step 4, the segment that contain promoter, ADH and PDC is the target sequence, according to the two sites of EcoR1 and BamH1, we digest the segment from PSB1A3 and ligate it onto PRL-489. | ||
| + | [[Image:tu15.jpg]][[Image:tu7.jpg]][[Image:tu8.jpg]] | ||
| + | As a result, we will be able to construct the plasmid same as we design in plan A. | ||
| + | *'''3. Tri-parental transformation''' | ||
| + | *After successfully constructing plasmid PA-489. The following is about expression. However the problem we are facing is that how the ethanol production pathway, existing in eukaryote, express in cyanobacteria which is a prokaryote? | ||
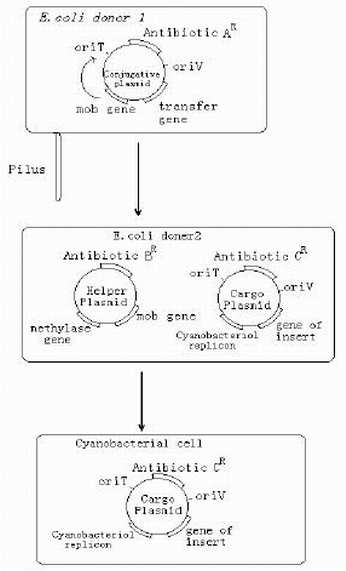
| + | *Firstly,integrating PA-489 into E-Coli with large-scale culture and screening,then we select the final invert E-Coli. Via tri-parental transformation,the plasmid will be able to transferred from E-Coli to cyanobacteria and express stably. | ||
| + | *Triparental mating is a form of Bacterial conjugation where a conjugative plasmid present in one bacterial strain assists the transfer of a mobilizable plasmid present in a second bacterial strain into a third bacterial strain. Plasmids are introduced into bacteria for such purposes as transformation, cloning, or transposon mutagenesis. Triparental matings can help overcome some of the barriers to efficient plasmid mobilization. For instance, if the conjugative plasmid and the mobilizable plasmid are members of the same incompatibility group they do not need to stably coexist in the second bacterial strain for the mobilizable plasmid to be transferred. | ||
| + | [[Image:tu16.jpg]] | ||
| + | *'''4, promoter''' | ||
| + | After construction the ethanol pathway into cyanobacteria,a suitable and effective promoter is needed to control the whole process. Inspired by the PET system, we hopes to build a cyanobacteria model which use T7 promoter to induse and control the express of target gene so that it can not only generate ethanol but also other bio-products. | ||
| + | |||
| + | '''4.1 PET System''' | ||
| + | |||
| + | *One expression system that was developed in 1986 by W. F. Studier and B. A. Moffatt, who created an RNA polymerase expression system which was highly selective for bacteriophage T7 RNA polymerase. The initial system involved two different methods of maintaining T7 RNA polymerase into the cell - in one method, a lambda bacteriophage was used to insert the gene which codes for T7 RNA polymerase, and in the other, the gene for T7 RNA polymerase was inserted into the host chromosome (Studier et al, 1986). This expression system has become known as the pET Expression System, and is now widely used because of its ability to mass-produce proteins, the specificity involved in the T7 promoter which only binds T7 RNA polymerase, and also the design of the system which allows for the easy manipulation of how much of the desired protein is expressed and when that expression occurs. | ||
| + | *A pET vector is a bacterial plasmid designed to enable the quick production of a large quantity of any desired protein when activated. This plasmid (pictured below) contains several important elements - a lacI gene which codes for the lac repressor protein, a T7 promoter which is specific to only T7 RNA polymerase (not bacterial RNA polymerase) and also does not occur anywhere in the prokaryotic genome, a lac operator which can block transcription, a polylinker, an f1 origin of replication (so that a single-stranded plasmid can be produced when co-infected with M13 helper phage), an ampicillin resistance gene, and a ColE1 origin of replication. | ||
| + | To start the process, your favorite gene (YFG) is cloned into a pET plasmid at the polylinker site. Both the T7 promoter and the lac operator are located 5' to YFG. When the T7 RNA polymerase is present and the lac operator is not repressed, the transcription of YFG proceeds rapidly. Because T7 is a viral promoter, it transcribes rapidly and profusely for as long as the T7 RNA polymerase is present (Campbell, 2003). The expression of your favorite protein (YFP) increases rapidly as the amount of mRNA transcribed from YFG increases. Within a few hours, YFP is one of the most prevalent components of the cell. | ||
| + | [[Image:tu17.jpg]] | ||
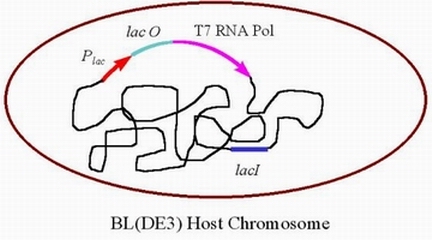
| + | *One of the most important parts of the pET Expression System involves the fact that YFG is not transcribed unless the T7 RNA polymerase is present. Prokaryotic cells do not produce this type of RNA, and therefore the T7 RNA polymerase must be added. Usually, the host cell for this expression system is a bacteria which has been genetically engineered to incorporate the gene for T7 RNA polymerase, the lac promoter and the lac operator in it's genome. When lactose or a molecule similar to lactose is present inside the cell, transcription of the T7 RNA polymerase is activated.. Typically, the host cell used is E. coli strain BL(DE3) (Blaber, 1998). The diagram below shows the genome of the host cell. | ||
| + | [[Image:tu18.jpg]] | ||
| + | *Control of the pET expression system is accomplished through the lac promoter and operator. Before YFG can be transcribed, T7 polymerase must be present. The gene on the host cell chromosome usually has an inducible promoter which is activated by IPTG. This molecule, IPTG, displaces the repressor from the lac operator. Since there are lac operators on both the gene encoding T7 polymerase and YFG, IPTG activates both genes (Science Advisory Board, 2003; Campbell, 2003). Therefore, when IPTG is added to the cell, the T7 polymerase is expressed, and quickly begins to transcribe YFG which is then translated as YFP. | ||
| + | IPTG works to displace a lac repressor since IPTG is an analogue of lactose (Blaber, 1998). The lac genes express enzymes which are involved in the breaking down of lactose, and therefore, the presence of lactose (or it's analogue) would trigger the initiation of transcription of lac genes. | ||
| + | |||
| + | '''4.2 The application of PET system in cyanobacteria.''' | ||
| + | *In order to enable cyanobacteria to generate T7 RNA polymerase, we construct the other plasmid making use of biobricks. We name it biobrick plasmid. The design proposal is as follows. | ||
| + | [[Image:tu19.jpg]] | ||
| + | *For the purpose of stable expression, the plasmid had better combined onto the geneom of cyanobacteria where homologous recombination is needed. | ||
| + | *Homologous recombination, also known as general recombination, is a type of genetic recombination in which nucleotide sequences are exchanged between two similar or identical strands of DNA. The process involves several steps of physical breaking and the eventual rejoining of DNA. Although most widely used to accurately repair double-strand breaks in DNA, homologous recombination also produces new combinations of DNA sequences during chromosomal crossover in meiosis. These new combinations of DNA produce genetic variation (e.g. new, possibly beneficial, combinations of alleles in populations as they reproduce, allowing them to evolutionarily adapt to changing environmental conditions over time. | ||
| + | In our project,firstly ,through the technique of PCR we are able to get homologous arms from Anebina 7120. Secondly, we ligate the homologous arms both at the beginning and at the end of the biobick segment. | ||
| + | [[Image:tu20.jpg]] | ||
| + | *With help of the two homologous arms, the plasmid can be easily recognized and combined onto the genome of cyanobacteria which enable it induced by T7 promoter. | ||
| + | |||
| + | ==== summary ==== | ||
| + | |||
| + | In the cyanobacteria convertor project,the construction of plasmid PA-489 and plasmid biobrick is the core work and tri-parental transformation is our unique technique. Besides our manipulation is all based on the synthetic biology. Hopefully through our design, cyanobacteria can not only generate ethanol but also increase the production and rate. | ||
Revision as of 00:47, 18 September 2009
| [[Home]|Project|Team | Protocols | Parts | Modeling |Gallery |Notebook|Ethics| |
Project
Project 1
- 1, the key enzyme for ethanol production
There is a pathway both in Saccharomyces Cerevisiae and Zymomonas.Mobilis.
pyruvic acid------> carbon dioxide + acetaldehyde ------>ethanol
- The enzyme which catalysis the first reaction is pyruvic decarboxylase (PDC)and the other enzyme catalyzing the following step is alcohol dehydrogenase(ADH)
In order to integrate this two enzyme into the genome of cyanobacteria so that it can carry on the ethanol producing pathway during the process of photosynthesis, we decided to construct a plasmid through which the ADH and PDC gene are able to express in cyanobacteria. The basic carrier we use is PRL -489. So after integrating the sequence of ADH and PDC on it , we name the new plasmid PA-489.
- 2. The construction of PA-489
To reach this goal we designed two plans and in each plan two groups are operated parallelly.
Plan A
*Group 1:
- step 1, there are two enzyme restriction sites on the plasmid PRL-489 which is EColR1 and BamH1,through double digests, excising this piece of segment and replace it with the promoter of groESL ,namely heat shock promoter.
- Step 2, with enzyme restriction sites ,xhoI and kpnI,ADH can be inserted after promoter groESL.
- Step 3, with enzyme restriction sites, SalI and SacI, PDC can be inserted after ADH.
- After the three steps the constructed result is our target plasmid.
Group 2: All the manipulations are the same with group 1, the only difference is that we use rubisco (rbc) promoter.
Plan B
- In this plan we utilize another technique called sub-clone. In molecular biology, subcloning is a technique used to move a particular gene of interest from a parent vector to a destination vector in order to further study its functionality.
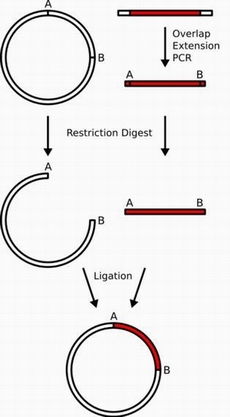 We need two carriers to construct PA-489 and also do it parallelly.
We need two carriers to construct PA-489 and also do it parallelly.
Group 1:
- step 1, we choose PSB1A3 as the basic model, with the same enzyme restriction sites EcoR1 and BamH1, heat shock promoter can be subcloned on the plasmid.
- Step 2, in the same way we subclone ADC after groESL.
- Step 3, with the restriction sites of SalI and SacI, we subclone PDC flowing to ADC.
- Step 4, the segment that contain promoter, ADH and PDC is the target sequence, according to the two sites of EcoR1 and BamH1, we digest the segment from PSB1A3 and ligate it onto PRL-489.
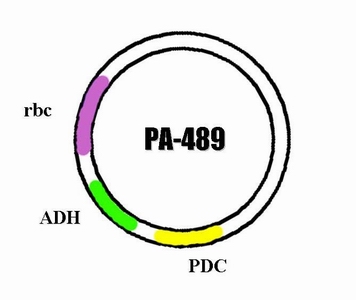 As a result, we will be able to construct the plasmid same as we design in plan A.
As a result, we will be able to construct the plasmid same as we design in plan A.
- 3. Tri-parental transformation
- After successfully constructing plasmid PA-489. The following is about expression. However the problem we are facing is that how the ethanol production pathway, existing in eukaryote, express in cyanobacteria which is a prokaryote?
- Firstly,integrating PA-489 into E-Coli with large-scale culture and screening,then we select the final invert E-Coli. Via tri-parental transformation,the plasmid will be able to transferred from E-Coli to cyanobacteria and express stably.
- Triparental mating is a form of Bacterial conjugation where a conjugative plasmid present in one bacterial strain assists the transfer of a mobilizable plasmid present in a second bacterial strain into a third bacterial strain. Plasmids are introduced into bacteria for such purposes as transformation, cloning, or transposon mutagenesis. Triparental matings can help overcome some of the barriers to efficient plasmid mobilization. For instance, if the conjugative plasmid and the mobilizable plasmid are members of the same incompatibility group they do not need to stably coexist in the second bacterial strain for the mobilizable plasmid to be transferred.
- 4, promoter
After construction the ethanol pathway into cyanobacteria,a suitable and effective promoter is needed to control the whole process. Inspired by the PET system, we hopes to build a cyanobacteria model which use T7 promoter to induse and control the express of target gene so that it can not only generate ethanol but also other bio-products.
4.1 PET System
- One expression system that was developed in 1986 by W. F. Studier and B. A. Moffatt, who created an RNA polymerase expression system which was highly selective for bacteriophage T7 RNA polymerase. The initial system involved two different methods of maintaining T7 RNA polymerase into the cell - in one method, a lambda bacteriophage was used to insert the gene which codes for T7 RNA polymerase, and in the other, the gene for T7 RNA polymerase was inserted into the host chromosome (Studier et al, 1986). This expression system has become known as the pET Expression System, and is now widely used because of its ability to mass-produce proteins, the specificity involved in the T7 promoter which only binds T7 RNA polymerase, and also the design of the system which allows for the easy manipulation of how much of the desired protein is expressed and when that expression occurs.
- A pET vector is a bacterial plasmid designed to enable the quick production of a large quantity of any desired protein when activated. This plasmid (pictured below) contains several important elements - a lacI gene which codes for the lac repressor protein, a T7 promoter which is specific to only T7 RNA polymerase (not bacterial RNA polymerase) and also does not occur anywhere in the prokaryotic genome, a lac operator which can block transcription, a polylinker, an f1 origin of replication (so that a single-stranded plasmid can be produced when co-infected with M13 helper phage), an ampicillin resistance gene, and a ColE1 origin of replication.
To start the process, your favorite gene (YFG) is cloned into a pET plasmid at the polylinker site. Both the T7 promoter and the lac operator are located 5' to YFG. When the T7 RNA polymerase is present and the lac operator is not repressed, the transcription of YFG proceeds rapidly. Because T7 is a viral promoter, it transcribes rapidly and profusely for as long as the T7 RNA polymerase is present (Campbell, 2003). The expression of your favorite protein (YFP) increases rapidly as the amount of mRNA transcribed from YFG increases. Within a few hours, YFP is one of the most prevalent components of the cell.
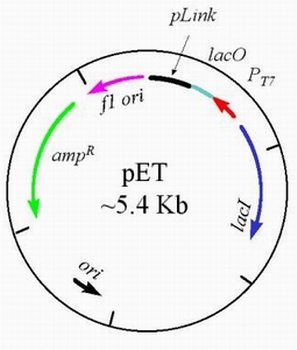
- One of the most important parts of the pET Expression System involves the fact that YFG is not transcribed unless the T7 RNA polymerase is present. Prokaryotic cells do not produce this type of RNA, and therefore the T7 RNA polymerase must be added. Usually, the host cell for this expression system is a bacteria which has been genetically engineered to incorporate the gene for T7 RNA polymerase, the lac promoter and the lac operator in it's genome. When lactose or a molecule similar to lactose is present inside the cell, transcription of the T7 RNA polymerase is activated.. Typically, the host cell used is E. coli strain BL(DE3) (Blaber, 1998). The diagram below shows the genome of the host cell.
- Control of the pET expression system is accomplished through the lac promoter and operator. Before YFG can be transcribed, T7 polymerase must be present. The gene on the host cell chromosome usually has an inducible promoter which is activated by IPTG. This molecule, IPTG, displaces the repressor from the lac operator. Since there are lac operators on both the gene encoding T7 polymerase and YFG, IPTG activates both genes (Science Advisory Board, 2003; Campbell, 2003). Therefore, when IPTG is added to the cell, the T7 polymerase is expressed, and quickly begins to transcribe YFG which is then translated as YFP.
IPTG works to displace a lac repressor since IPTG is an analogue of lactose (Blaber, 1998). The lac genes express enzymes which are involved in the breaking down of lactose, and therefore, the presence of lactose (or it's analogue) would trigger the initiation of transcription of lac genes.
4.2 The application of PET system in cyanobacteria.
- In order to enable cyanobacteria to generate T7 RNA polymerase, we construct the other plasmid making use of biobricks. We name it biobrick plasmid. The design proposal is as follows.
- For the purpose of stable expression, the plasmid had better combined onto the geneom of cyanobacteria where homologous recombination is needed.
- Homologous recombination, also known as general recombination, is a type of genetic recombination in which nucleotide sequences are exchanged between two similar or identical strands of DNA. The process involves several steps of physical breaking and the eventual rejoining of DNA. Although most widely used to accurately repair double-strand breaks in DNA, homologous recombination also produces new combinations of DNA sequences during chromosomal crossover in meiosis. These new combinations of DNA produce genetic variation (e.g. new, possibly beneficial, combinations of alleles in populations as they reproduce, allowing them to evolutionarily adapt to changing environmental conditions over time.
In our project,firstly ,through the technique of PCR we are able to get homologous arms from Anebina 7120. Secondly, we ligate the homologous arms both at the beginning and at the end of the biobick segment. File:Tu20.jpg
- With help of the two homologous arms, the plasmid can be easily recognized and combined onto the genome of cyanobacteria which enable it induced by T7 promoter.
summary
In the cyanobacteria convertor project,the construction of plasmid PA-489 and plasmid biobrick is the core work and tri-parental transformation is our unique technique. Besides our manipulation is all based on the synthetic biology. Hopefully through our design, cyanobacteria can not only generate ethanol but also increase the production and rate.
 "
"