Team:Kyoto/CiC/Results
From 2009.igem.org
- Home
- Cells in Cells
- Results & Discussion
Results & Discussion
Result of Subgoal A
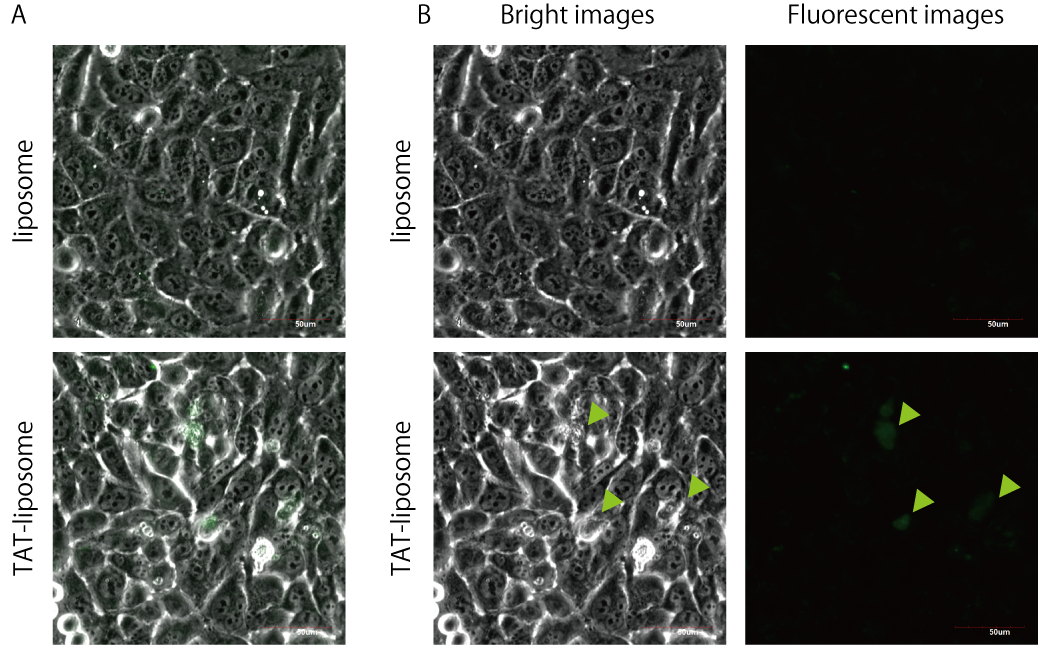
Confirmation of function of TAT-liposome
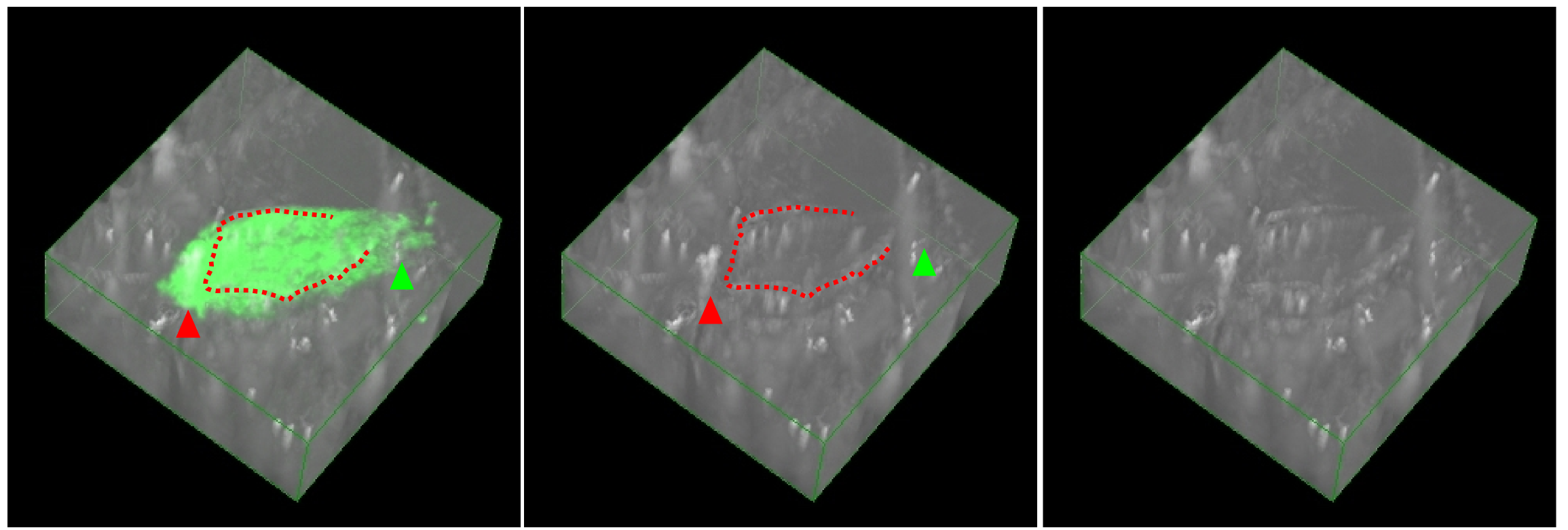
To confirm the function of the constructed TAT-anchor peptide, we prepared the liposome containing NBD-PE with or without TAT-anchor which was synthesized or translated in vitro. The size of the liposome without TAT was too small to distinguish individual particle, while that with TAT showed the large coalesced liposomes (Fig. 1-(A)). The coalesced liposomes might be caused by the aggregation between TAT peptides or electric screening effect of TAT against liposome. We, any way, added the prepared liposomes into the medium of cells. Since the cells incubated with liposome did not emit any fluorescent signal, no liposome was uptaken by cells nonspecifically. In contrast, the TAT-liposome adhered on some cells (Fig. 1-(B), arrowheads). We, thus, observed the cells by a conforcal microscopy to reveal whether the liposomes was uptaken or not. The sliced image of the cell indicates that the liposome was at least taken partially since fluorescent signal was within the outline of the cross-section of the cell (Described in Fig. 2 as red dashed-line). However, the fluorescent signal was also detected in the outside of the cellular outline around the green or red arrowhead shown in Fig. 2. The fluorescence pointed by the red arrowhead is probably a coalesced liposome which could not be uptaken by cell because of its too large size. The fluorescent signals around the green arrowhead may be within the cell because the outline of the cell around the arrowhead is difficult to determine. Consequently, we beleave our constructed TAT-liposome at least has the function of the transduction into the cells. However, the efficiency was very low and the shape of the liposome inside or outside of the cell are not sphere. We must improve the liposome by optimizing the protocol and the ratio between a lipid and a TAT peptide.
Result of Subgoal B
Result of Subgoal C
sig-GFP expression in HeLa
To confirm the function of the signal sequence for importing protein into mitochondria, we compared the expressioin pattern of sig-GFP or GFP with mitotracker signal. The GFP signal was detected throughout the cell except for the black granules in the cytoplasm or the nuclear, while sig-GFP signal showed the string-like pattern in the cytoplasm. The black granules in the cytoplasm observed in the GFP-expressing cell were stained by mitotracker, and the result indicated that the GFP was normally eliminated from mitochondria (Fig. 3, GFP and mitotracker merged). In case of sig-GFP, mitochondria stained by a mitotracker and the GFP signal showed almost the same pattern. the yellow color in the merge images (Fig. 3, sig-GFP and mitotracker merged) suggested that the sig-GFP and mitochondria were colocalized in the HeLa cell. We, consequently, our constructed signal sequence has the function of importing protein into mitochondria as expected.
 "
"