Team:UNIPV-Pavia/New Parts
From 2009.igem.org

|
|
|
Our new parts |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Our new parts:
Re-built existing parts (BBa_our part code/BBa_existing part code) Existing parts from the Registry Existing parts: sequence debugging
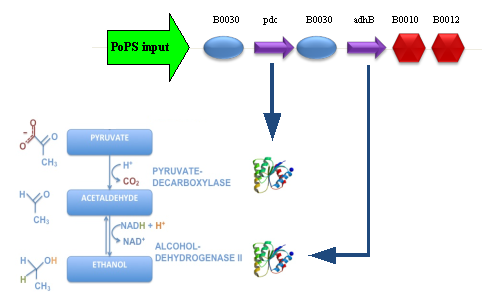
BBa_K173003 - ethanol producing deviceDescriptionThis device takes PoPS as input and produces pyruvate decarboxylase (pdc) and alcohol dehydrogenase II (adhB) enzymes. Pyruvate decarboxylase (pdc, ) catalyses the decarboxylation of pyruvic acid to acetaldehyde and carbon dioxide, while alcohol dehydrogenase II (adhB, ) catalyses the acetaldehyde reduction to ethanol. The latter enzyme, as reported in the literature, is also able to work in the opposite direction, oxidizing ethanol to acetaldehyde. The two enzymes come from Zymomonas mobilis ethanologenic bacterium and constitute an essential step of alcoholic fermentation. So, this device contains the minimum set of genes that are required to engineer a heterologous fermentation pathway. The coding sequences of pdc and adhB genes have been optimized for Escherichia coli codon usage. CharacterizationQualitative phenotype characterization PLATES RESULTS This device has been cloned downstream of 4 different promoters, one of which in two different vectors. These assemblies were transformed in TOP10 and gave the following phenotypes when bacteria were plated on LB agar plates or on LB agar plates + 2% glucose (all the plates have been incubated at 37°C for about 11 hours, then some colonies were picked with a sterile tip and plates were incubated for additional 10 hours):
These results demonstrate that a constitutive expression of pdc and adhB gives a high metabolic burden to E. coli, in fact the only normal phenotype on plates was with non-induced device. Only this assembled part has been submitted to the Registry, as .
Grown cultures of and after the FIRST INOCULUM showed different phenotypes (qualitative analysis), as a function of the used protocol. was the regulated operon, was the promoterless operon, was the promoter alone, pSB1AK3 was a high copy plasmid and pSB4C5 was a low copy plasmid.
PROTOCOL#3 RESULTS (i.e. shaken anaerobic falcon tubes, 2% glucose):
These results confirm the metabolic burden given by this BioBrick when gene expression is triggered: induced strains bearing a high copy plasmid containing do not survive, while low copy ones can survive even at 1 uM HSL, which is a high induction, according to the characterization results on (see its own page on the Registry and our our characterization experiments on it). We decided to exploit the lux promoter leakage activity for in pSB1AK3 (high copy), while we decided to induce in pSB4C5 (low copy) the with 1 uM of 3OC6-HSL during our fermentation experiments. The qualitative phenotypes of the grown cultures of and after fermentation in 10% glucose showed that pellets were higher in (in both pSB1AK3 uninduced and pSB4C5 induced) than in and in the negative control .
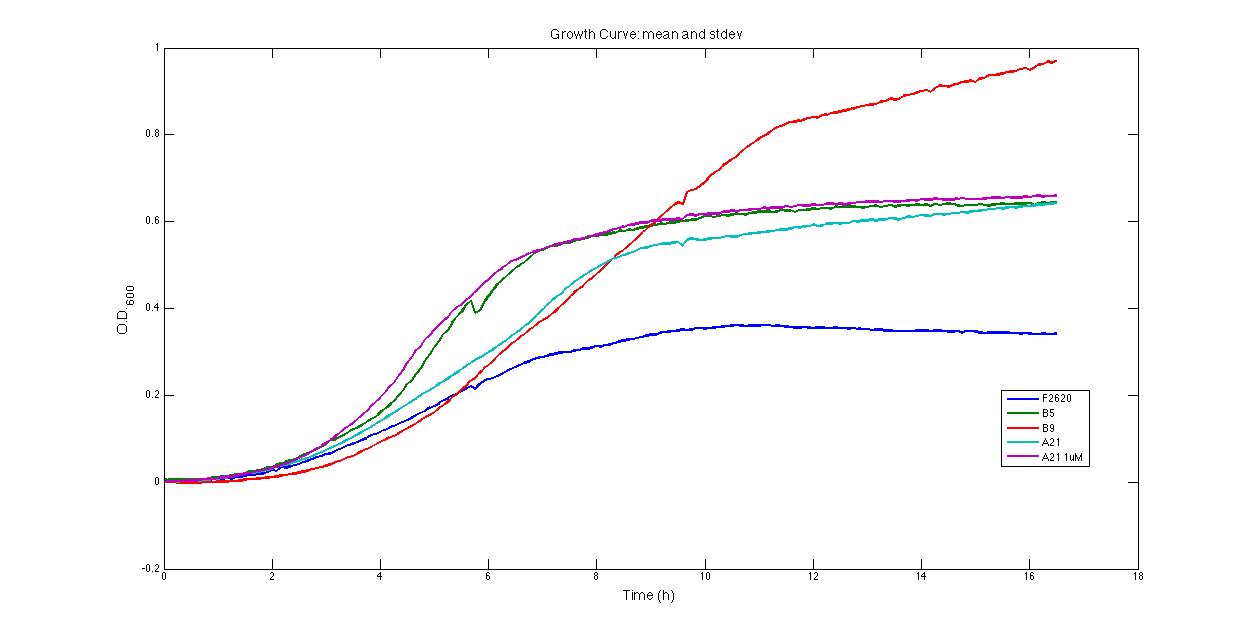
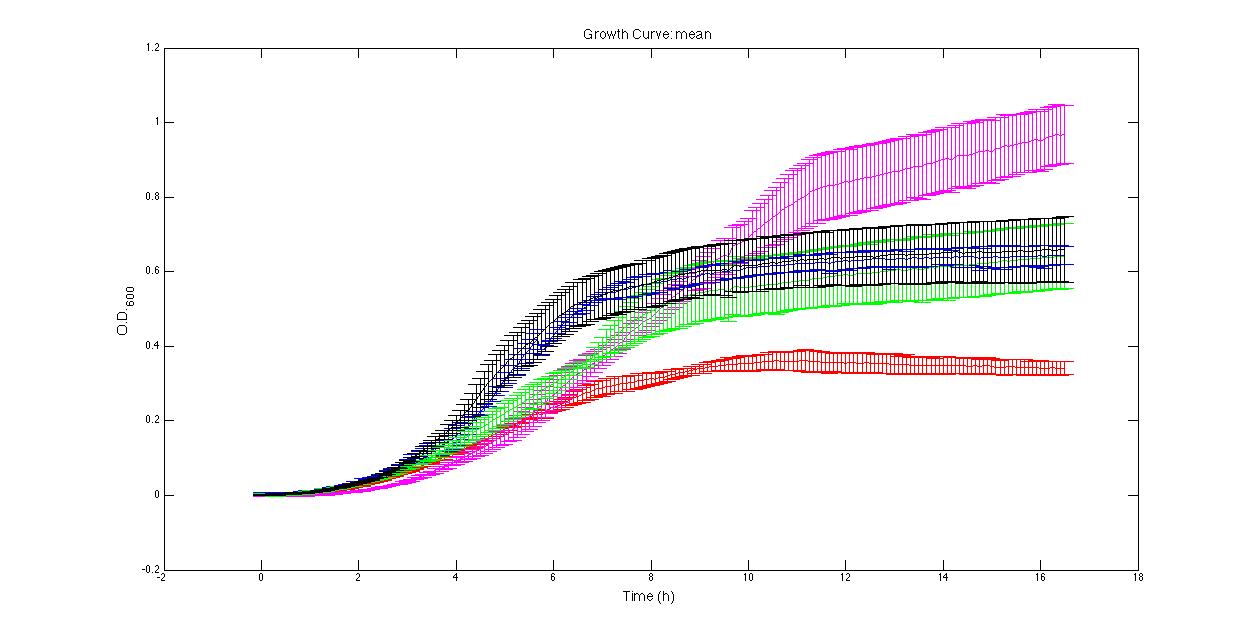
Quantitative characterization pH pH has been measured in grown cultures after the FIRST INOCULUM (in 2% glucose after 24 hours) and after the SECOND INOCULUM (in 10% glucose after 24 or 48 hours). The measurements performed are the following: PROTOCOL#1: pH at the end of fermentation (after the SECOND INOCULUM). PROTOCOL#2: pH after the FIRST and the SECOND (end of fermentation) INOCULUM PROTOCOL#3: pH after the FIRST and the SECOND (end of fermentation) INOCULUM The shown results suggest that in strains bearing expressed pdc and adhB the fermentation does not give high levels of organic acids as in negative control strains which do not have pdc and adhB. It is surprising that the pH of in pSB1AK3 (pdc and adhB without any promoter) is slightly higher than in the negative controls. This could be due to a weak spurious transcription of pdc and adhB, amplified by the high copy number plasmid. This may re-direct part of pyruvate metabolism to ethanol and not to organic acids. Of course this phenomenon should be further investigated. Growth curves and its suitable negative controls growth was tested in our microplate reader: 200 ul aliquots (3 or 4 replicates) were taken from a just inoculated 30 ml culture (LB + 10% glucose) of the SECOND INOCULUM of PROTOCOL#3 and let grow in the microplate reader according to the automatic protocol described in Microplate reader experiments. The microplate was "sealed" with parafilm in order to create an anaerobic condition. This test showed that in pSB1AK3 is able to reach much higher O.D.600 than the other cultures, reaching values of ~1. in pSB4C5 both induced and uninduced had comparable growth curves and reached O.D.600 of about 0.7. Negative controls ( in pSB1A2 and in pSB1AK3) had comparable growth curves and reached low O.D. (about 0.4). It is surprising that in pSB1AK3 reached a higher OD (~0.7) than the negative controls. The shown results are in complete accordance with the pH results, confirming that strains bearing in pSB1AK3 and in pSB4C5 produce less organic acids than the negative controls, hopefully thanks to pdc and adhB and consequently grow better.
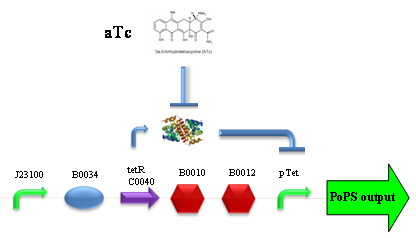
PROTOCOL#1: ethanol at the end of fermentation. PROTOCOL#2: ethanol at the end of fermentation. PROTOCOL#2: ethanol at the end of fermentation. PROTOCOL#3: ethanol at the end of fermentation. ConclusionsBBa_K173007 - aTc inducible device with J23100 promoterDescriptionThis is an aTc sensing device. promoter drives the constitutive production of tetR repressor (), which inhibits tetR promoter () activity. When aTc is added to the medium, it binds tetR and inhibits it. So, the PoPS output is a function of the aTc concentration. A tight regulation is expected for this inducible system because BBa_J23100 is a strong promoter and so tetR repressor should be produced at extremely high levels. The data below are referred to , which is the measurement system of . Characterization
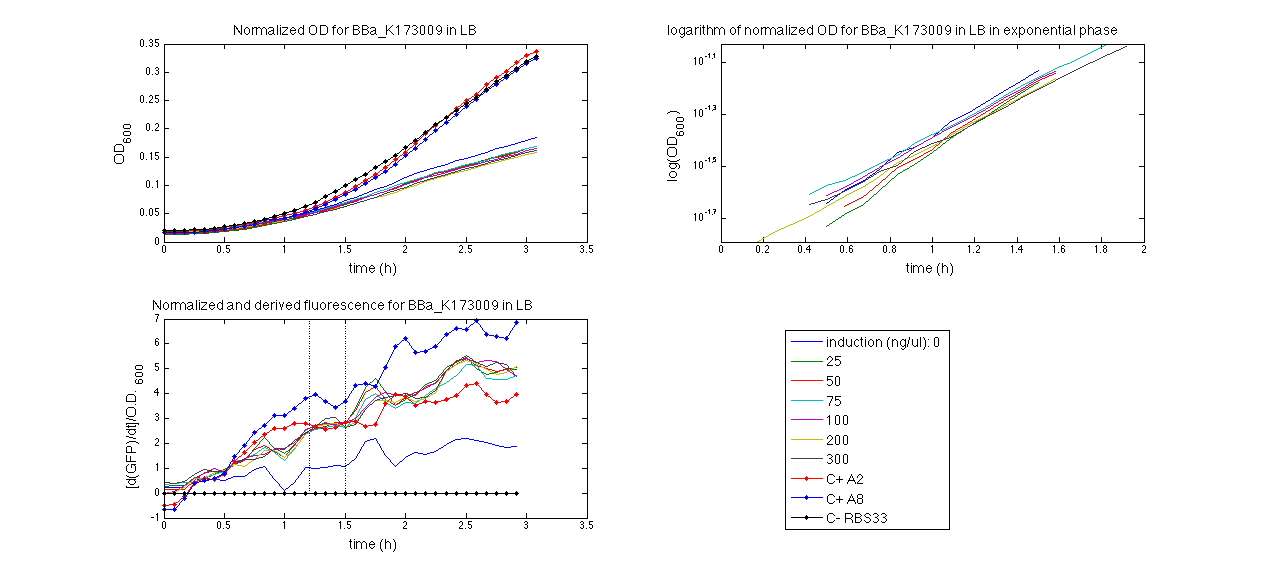
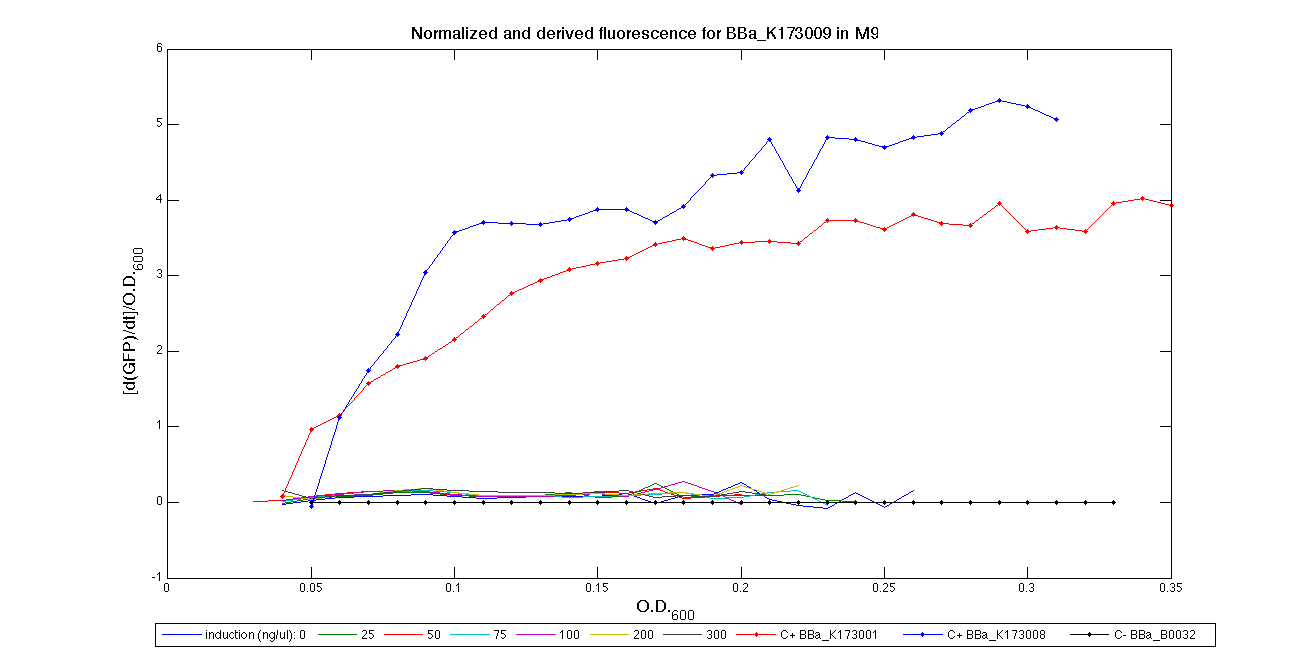
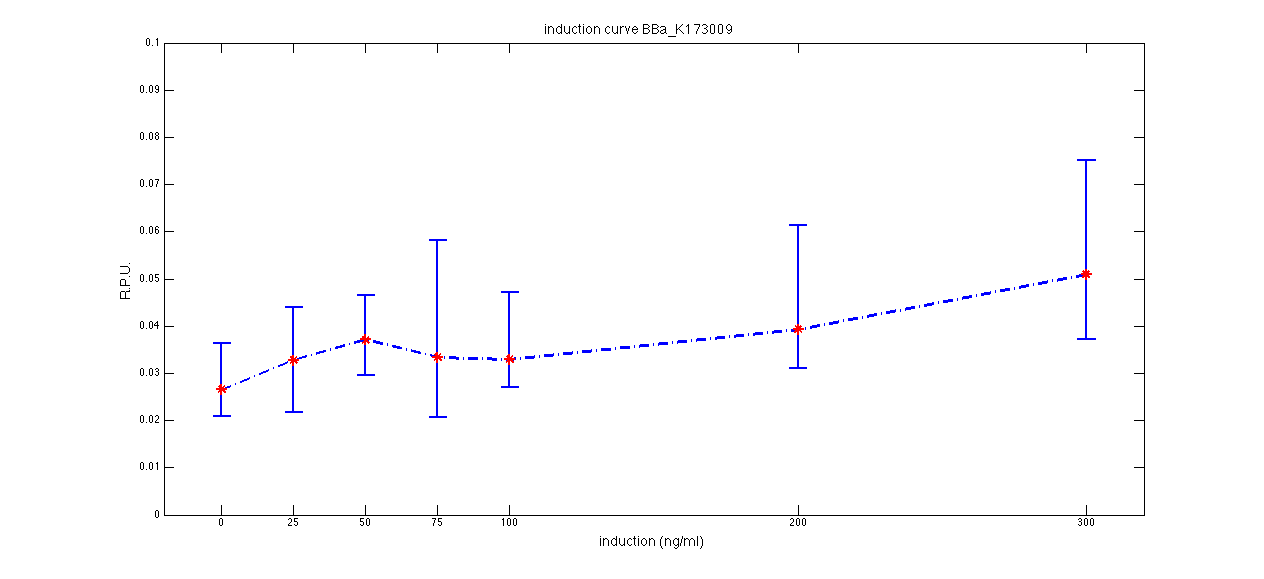
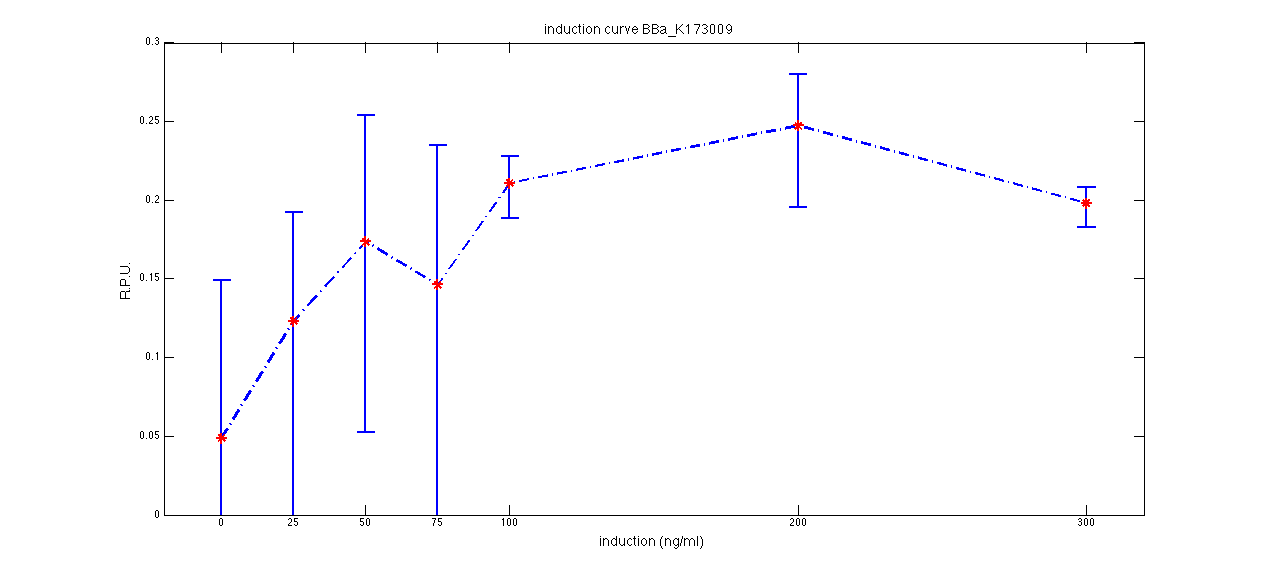
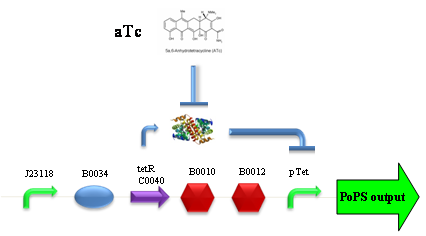
ConclusionsWe demonstrated that this part works as expected, sensing the aTc concentration provided in the culture medium. The transfer function of this sensor has been characterized in standard units (RPUs) in two different growth media (LB and M9 supplemented with glycerol), as well as the metabolic burden (in terms of doubling time) which affects E. coli bearing this part. On the other hand, we did not expect to have a higher GFP synthesis rate per cell after the exponential growth phase than in the exponential phase itself (as reported in the 3rd plot for both LB and M9). This parts shows to have a very low leakage rate (about 0.025 RPU) but also a very low induction for high concentrations of aTc. So it can be used for tight regulation, in those systems who need very low leakage rates (no gene expressed in absence of inducer) but not for massive protein production. Unfortunately, the estimated transfer function is very noisy (probably because the measured fluorescence values are very close to blank values), and further experiments should be performed in order to validate these induction curves. BBa_K173011 - aTc inducible device with J23118 promoterDescriptionThis is an aTc sensing device. promoter drives the constitutive production of tetR repressor (), which inhibits tetR promoter () activity. When aTc is added to the medium, it binds tetR and inhibits it. So, the PoPS output is a function of the aTc concentration. A less tight regulation is expected for this inducible system than in because BBa_J23118 promoter is weaker than BBa_J23100 and so tetR repressor shold be produced at lower levels than in the other sensor. The data below are referred to , which is the measurement system of . Characterization
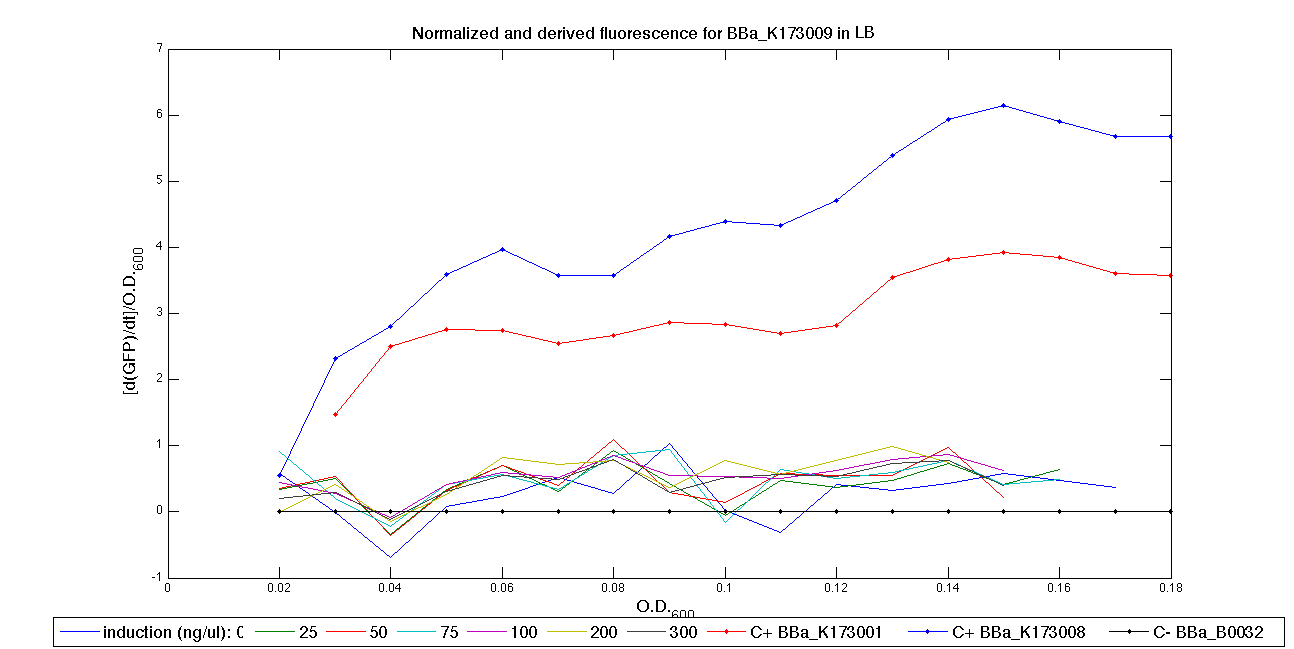
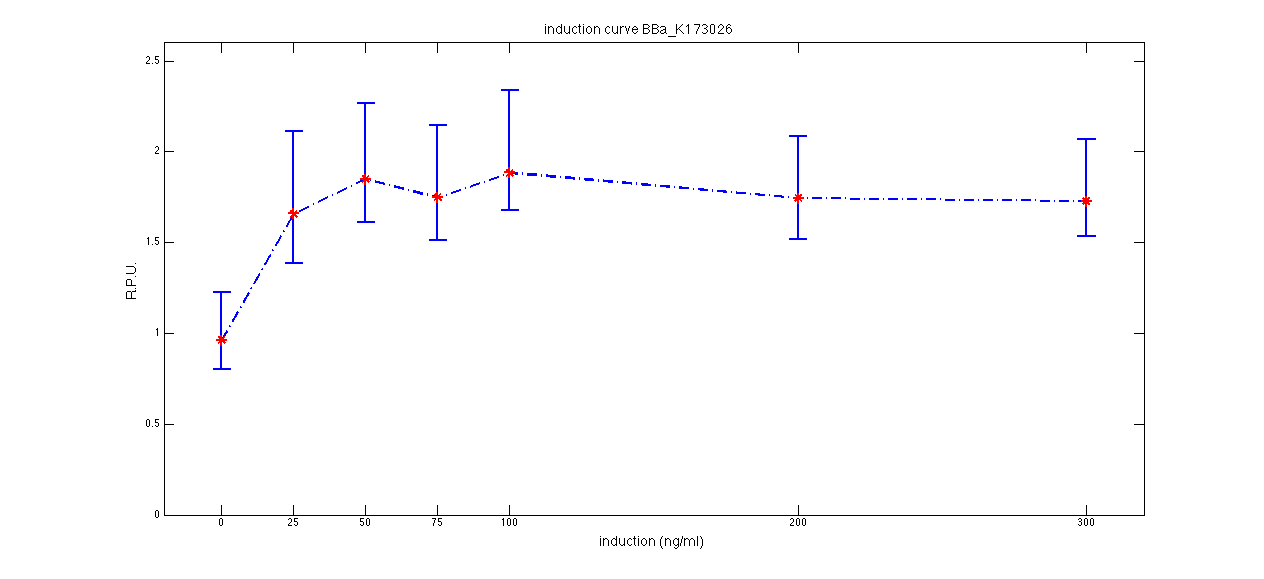
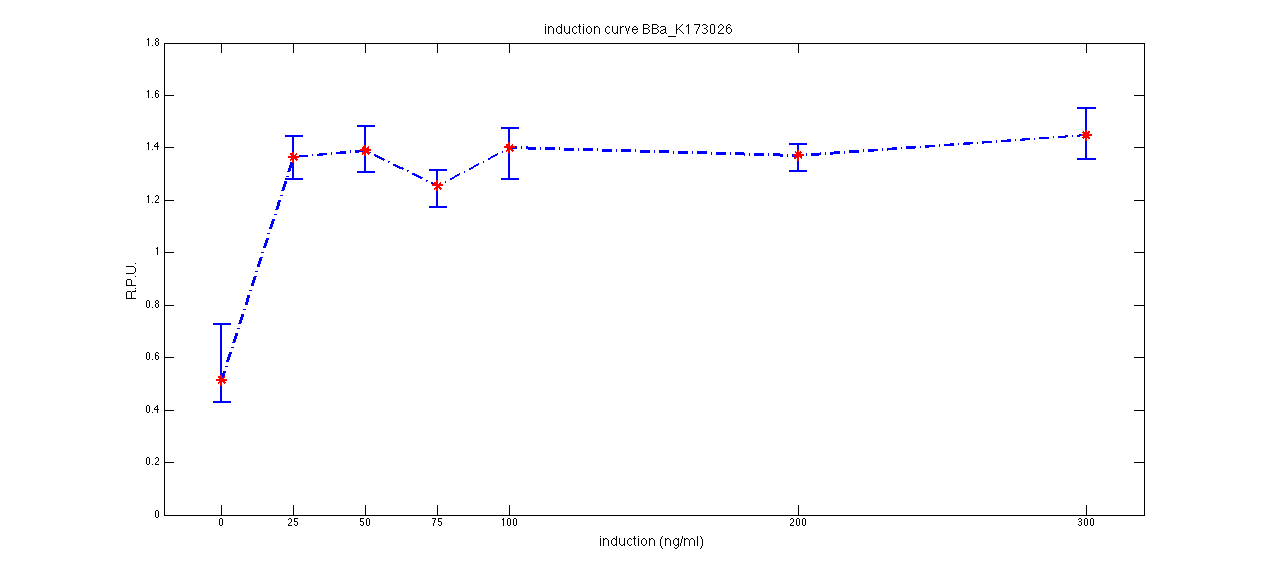
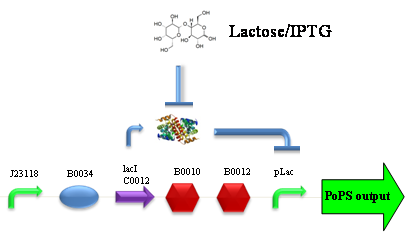
ConclusionsWe demonstrated that this part works as expected because GFP is produced as an increasing function of the aTc concentration provided in the culture medium. The transfer function of this sensor has been characterized in standard units (RPUs) in two different growth media (LB and M9 supplemented with glycerol), as well as the metabolic burden (in terms of doubling time) which affects E. coli bearing this part. On the other hand, as for BBa_K173007, we did not expect to have a higher GFP synthesis rate per cell after the exponential growth phase than in the exponential phase itself (as reported in the 3rd plot). This part shows a high level of leakage (about 0.5 RPU in LB and 1 RPU in M9) but also high levels of induction for high concentrations of aTc (about 1.45 RPU in LB and 1.7 RPU in M9). For this reason, it can be used in those cases where an important response in terms of gene expression is required in presence of an inducer, but is not suitabe if the absence of expression has to be off when inducer is absent. Its induction range is 0.95 RPU in LB and 0.7 RPU in M9. The switch point of this device is between 0 ng/ml and 25 ng/ml of aTc in both LB and M9. BBa_K173010 - lactose/IPTG inducible device with J23118 promoterDescriptionThis should work as a lactose/IPTG sensor. promoter drives the constitutive production of lacI repressor (), which inhibits lac promoter () activity. When lactose or IPTG is added to the medium, it binds lacI and inhibits it. So, the PoPS output is a function of lactose/IPTG concentration. Thanks to the hybrid lac promoter (), designed taking the Plambda promoter () and substituting its cI () binding sites with two lacI binding sites, the behaviour of this device is not a function of glucose concentration because the wild type CAP binding sites are not present in this artificial lac promoter. The test performed to characterize this device has been done with , which is the measurement system of itself. Characterization
Results: both cultures had a comparable O.D.600, but also had the same RFU values, demonstrating that the induced culture did not produce any GFP. ConclusionsWe did not perform any standard measurement on this device because preliminary tests showed that GFP levels of induced and non induced cultures were the same and were equals to negative control. Unfortunately, we did not check if the sequence of , so we do not know if it was actually correct. Another possibility is that lacI is produced at so high levels that 2 mM of IPTG is not sufficient to induce the lac promoter. Further tests should be done for this system.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"