Team:BCCS-Bristol/Notebook
From 2009.igem.org
iGEM 2009
Contents |
1. Abstract
We wanted to exploit the fact that outer membrane vesicles (OMVs) were naturally produced in gram negative bacteria such as E. coli. The aim was to use them in our advantage as a directed delivery system to cells of proteins of our interest.This can potentially be used for the safe delivery of drugs into cells or for cell-cell communication purposes.We believe that the inclusion of proteins in vesicles will not only protect them from degradation in the extracellular environment but will also protect other cells if the cargo proteins packaged into vesicles are toxic or harmful.
Because the exact mechanism by which OMVs are produced in gram negative cells is not yet elucidated (although 3 models have been proposed, Lauren M. Mashburn-Warren et al, 2006) we decided to make protein fusions of proteins that were normally included in OMVs with proteins of our interest. Experiments with protein fusions to the toxin ClyA were already made with success (Jae-Young Kim et al, 2008) and the desired proteins were delivered to OMVs. But for the purposes that we would be using OMVs we thought that it would be safer to use harmless proteins that would act as carriers instead of ClyA. Hence we selected 3 possible candidate protein carriers (Eun-Young Lee et al, 2007) to be used in our project.These proteins are:
1. OsmE (Osmotically inducible lipoprotein E)
2. fiu (siderophore receptor)
3. FhuA (Ferrichrome-iron receptor)
In order to assess the production of our fusion proteins and at the same time monitor their introduction into the OMVs we used GFP (Green Fluorescent Protein) as our cargo protein fused to one of the three carriers stated above.
2. Bioscaffold
2.1 Protein Fusion Problems
Each gene that encodes a protein naturally ends with two stop codons (5'-TAATAA-3'). Moreover when two genes are fused together an additional third stop codon is inserted in the DNA scar (DNA region that connects the two genes). Hence this would prevent the expression of the second partner in the fusion protein.
2.2 Existing Solutions
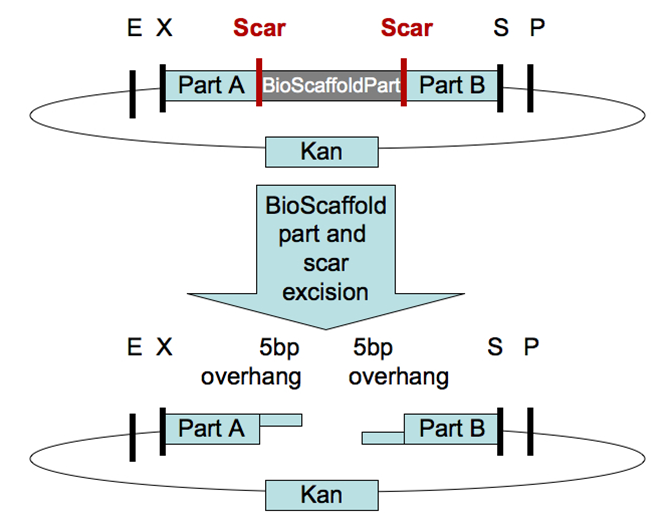
The current iGEM assembly standards do not fully support protein fusions. Only assembly standard 15, a concept known as Bioscaffold, tries to address this problem in some depth. The Bioscaffold is a DNA sequence that is ligated between the two protein encoding genes and contains restriction sites of atypical Type II restriction enzymes. The enzymes will cleave outside their recognition sequence hence removing the stop codons of the first protein in the fusion. However the current scaffolds are not compatible as biobricks or not available in biobrick format and some require further manipulation with PCR primers.
2.3 BCCS Bioscaffold
For the purposes of our project we had to construct protein fusions comprising of a carrier and a cargo and as a result we had to deal with this fusion problem in some way. This led to us to design and create a new type of [http://partsregistry.org/Part:BBa_K259002 Bioscaffold] which would keep the basic concept of bioscaffold the same but offer more flexibility in terms of protein fusions.
2.4 Bioscaffold Features
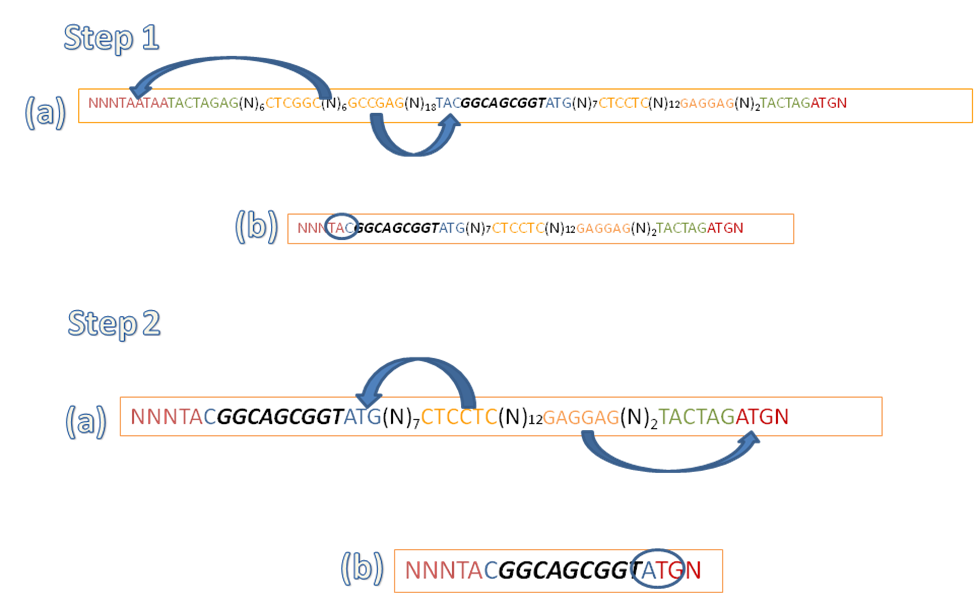
Our bioscaffold is placed between two coding biobricks and contains restriction sites of atypical Type II restriction enzymes like assembly standard 15. It has a restriction site of BpuEI (Type IIS) on the left hand side and BseRI (Type IIB) on the right hand side (both are 6 cutters).
2.5 Bioscaffold Application
2.5.1 Bioscaffold Advantages
All stop codons are removed by the action of these restriction enzymes and thus will allow the expression of all the protein partners in the fusion. Also the methionine start codon of downstream gene is maintained and hence it will be transcripted correctly. An important advantage of bioscaffold is the Gly-Ser-Gly linker that is added between the two genes after the restriction digest which provides flexibility for the correct folding of proteins in the fusion into their native 3D conformations. Furthermore the reading frame of both proteins remains unchanged maintaining their primary structure intact. BpuEI and BseRI restriction sites are not present in pSB2K3 vector so biobrick fusions can be safely carried out on this vector. Last but not least the bioscaffold is compatible with assembly standards 10, 21, 23 and 25 as mentioned above making it very useful to synthetic biologists.
 "
"