Team:BCCS-Bristol/BSim/Case studies/Repressilators
From 2009.igem.org
iGEM 2009

| 
| 
|
Contents |
Repressilators coupled by quorum sensing - an agent based approach
The emergence of new GRNs such as the repressilator [1], which may be affected by factors on the population rather than the individual level, has sparked a new interest in modelling GRNs across a bacterial population. Synthetic clocks such as the repressilator may help to provide us with a deeper understanding of oscillatory behaviour in natural systems. Theoretical modelling of such systems across a population is an important step towards better understanding of natural oscillators such as the circadian clock.
What is a repressilator?
A repressilator is a synthetic gene regulatory network, which is generally constructed from three different genes in a feedback loop such that they repress each other in sequence. This can result in self-sustained oscillations occurring in the network. An example of an analogue in the world of electronics would be a [http://en.wikipedia.org/wiki/Ring_oscillator ring oscillator], in which a number of inverters are connected together in sequence. The original repressilator also includes coupling to a green flourescent protein (GFP) reporter so that the oscillations can be observed visually.

The single repressilator GRN can be simply extended as shown in figure 2(below) to include an autoinducer component. This would allow repressilators in individual bacteria to communicate with one another via an external chemical field.
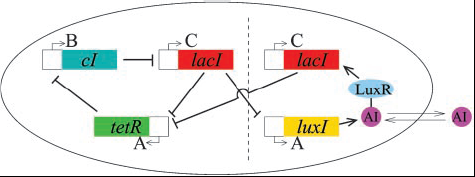
Previous work
The quorum-coupled repressilator is an excellent initial model for invesigating naturally occurring oscillatory phenomena such as the circadian clock. As a result there have already been a number of theoretical models produced to investigate the behaviour of such a system, for example [2].
Many of these previous approaches to modelling quorum coupled repressilators have assumed that the chemical is well-mixed across the population. In reality external chemical concentrations will vary across a large space, therefore it is important to consider the effects of a nonuniform chemical field on network dynamics.
Why we used BSim
Recent mathematical modelling approaches in systems biology tend to model a gene regulatory network in a single cell, and agent based models are considered in a separate context. However, some GRNs such as the repressilator can be coupled across a population of bacteria. In the case of the repressilator the GRNs are coupled by an autoinducer chemical which is free to diffuse in and out of the cell.
In [2] it was shown that communication between cells via quorum sensing can result in population level synchronisation given a large enough cellular density. By bringing together an agent based approach with the standard ordinary differential equation methods used for modelling GRNs we hope to be able to extensively study spatial factors affecting the behaviour of the repressilator in a population, as well as allowing for full spatial considerations in our chemical field model.
BSim coupled repressilators
When modelling the population of repressilator-containing bacteria in BSim, we used many of the same features as the model outlined in [2]. The repressilators themselves are modelled as a system of 7 ODEs; 3 ODEs representing the 3 different mRNA levels, 3 ODEs for the 3 corresponding proteins, and one ODE for the internal level of autoinducer. These are in turn coupled to an external spatially varying chemical field via the autoinducer term, which incorporates physically correct diffusion and degradation characteristics. For the modelling of the autoinducer behaviour, we made the assumption that the autoinducer in question was AHL as this is a common quorum signalling molecule. The parameters for diffusion (in space and through the cell wall) and decay were then set accordingly. The parameter governing the ratio between mRNA and protein degradation was chosen from a random distribution as in the first part of [2].
Overview
Due to the complex nature of the complete model, there are a large number of possible directions for an investigation to take. As a result of time constraints of the iGEM project we were only able to look into the effects of a few of the parameters of the system. However we were able to verify the validity of our model by reproducing some of the key results in [2], which we summarise below.
However, the project continues, and we have outlined some possible directions in which we plan to take our investigation in the final part of this summary.
Results
So far, we have managed to investigate some of the effects of diffusion on the level of synchronisation in the system. Using realistic parameters for AHL, when considering the very small size of the simulation region, results in almost instantaneous diffusion. This is good as it allows us to simulate closely a 'well-mixed' environment as an initial assumtion and to evaluate the validity of our model. The following series of images shows the external chemical field oscillating as a result of the rapid diffusion speed. This can also be observed in the video at the end of this section.

| 
| 
| 
|
The following video shows 200 bacteria swimming in a 100x100x100 micron volume. Each bacterium has a system of ODE's inside which model the essential dynamics of a repressilator. In this case, the individual repressilators are coupled via a diffusing autoinducer signal (in this case AHL). The colour of a bacterium represents the level of lacI mRNA in that bacterium (the lacI gene is one of the genes present in the repressilator); the bacterium will change from yellow to red as the internal level of lacI mRNA increases. In the example shown here, all 200 of the individual repressilators are initialised with random conditions, but quickly synchronise due to the effect of the AHL communication.
Due to the long time scale of repressilator oscillations the simulation was run for 10 hours in total; the video has therefore been sped up a fair amount and as a result individual bacterial motion may be difficult to see. However, a number of key features can be observed, namely the oscillation of individual repressilators and the pulsng oscillation in the external chemical field resulting from AHL diffusing into and out of the bacteria.
Of the two diffusivity parameters present in our system, it was found that cell wall diffusivity had the most noticeable effect on synchronisation. The effect of external diffusivity was negligible due to the small size of our simulation region. In the following sections we summarise some of the results that were obtained when cell wall diffusivity was varied.
Phase locking
The following images show the time series of a bacterium vs. 3 other bacteria for the final ~6 hours of the simulation. It can be clearly seen that at this point synchronisation has occurred, though phase locking is not always perfect! This is due to the randomness in our degradation ratio parameter. (Left to right: increasing degrees of phase locking):

| 
| 
|
Frequency
- plots
- as we increase the parameter X the F_coupling/synch etc increases
Synchronisation transition
- the parameter we used to measure synch
- increasing diffusion = better synch
- uniform ICs reproduces what was found in Strogatz paper
Further work
- parameters - which are important/relevant - there are many more compared to strogatz etc
- stochasticity
- control - open loop/closed loop - via injection of AI - coupling model to an actual experiment
- better measure of synch - spatial synch
- clustering - effects of control on this
- things
References
- [1] Michael B. Elowitz & Stanislas Leibler - A synthetic oscillatory network of transcriptional regulators | [http://dx.doi.org/doi:10.1038/35002125 doi:10.1038/35002125]
- [2] J. Garcia-Ojalvo, Michael B. Elowitz, Steven H. Strogatz - Modeling a synthetic multicellular clock: Repressilators coupled by quorum sensing | [http://dx.doi.org/doi:10.1073/pnas.0307095101 doi:10.1073/pnas.0307095101]
 "
"