Team:Valencia/Notebook/August
From 2009.igem.org
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
August 3rd
- Yeasts haven't grown yet.
- iGEM meeting.
August 4th
Today we have had a yeast-team meeting. We suspect that our yeasts are not growing because they died during the trip between Barcelona and Valencia. We have decided to spread the yeasts we have left in YPD medium that had already been prepared, but we are skeptical about the results. If it doesn't work, Joaquin Arinyo and his teammates will send us more aequorin-transformed yeasts.
We have also made 1 liter of newn solid SD lacking Leu medium.
August 5th
Today we have prepared the Coelenterazine stock dissolution. First we "put" N2 gas through a Methanol dissolution in order to substitute the O2, an inhibitor for the coelenterazine. After 5 or 10 minutes we took 200 microL and added them to 50 microg of Coelenterazine.
Then we have distribute them in eppendorfs of 5 microL each and kept them in the fridge at -20º.
For other side, we doubt if our yeast in the YPD medium have grown or not... We think we should wait until tomorrow... T-T... Like Cristina says, our yeasts are like Peter Pan: they don't want to grow up...
August 6th
Yeast have grown. YEaaaaaaaaaaaaahhhh!!! We have spread them into the SD solid medium lacking Leu that we did yesterday, we will wait until tomorrow for the results. Also we have prepared new SD liquid medium lacking Leu and once prepared we will also spread our super-yeasts in it. Tomorrow we will start with the protocol (we hope).
August 7th
Yeast have grown in SD medium lacking Leu (bothsolid and liquid) so we have started the protocol. The OD was low (0'18) but we have prepared an 1'8 OD dissolution of the yeasts by centrifugation and dilution with SD medium lacking Leu.
Allright... well... we have mesured the "luminiscence" and we have seen nothing. We've made the experiment in an espectophotometer and in the electrophoresis gel photograph machine. Before the mesurements we checked the OD and it was 0'34 so very far from 1'8. Maybe that's the problem. We're not depressed anyway, on monday we will come back to demonstrate those yeasts what we can do. Valencia's iGEM way.
August 10th
Today, we've made a lot of things. Yeasts haven't grown after three days in SD lacking Leu liquid medium at 30º. We've found out that we were preparing the medium in the wrong way.
The right way is:
For 500ml:
- 2'5 g (NH4)2SO4
- 1 g Yeast Synthetic Drop-Out Medium Supplement Without Leucine
- 10 g Glucose
- 7'5 g Agar (only for the solid medium)
We have also found out that the minimal volume for the spectophotometer is 400 microL so we will double all the quantities of the protocol.
August 11th
We repeated the protocol and we mesured in the espectrophotometer but we saw nothing. We think it's too few sensitive and we're going to ask for a fluorimeter. We also did the experiment in GelDoc but, again, we saw nothing.
August 12th
New yeasts have arrived. We have also prepared a mechanism to mesure in the GelDoc at the same time we add the KOH with the door closed and no delay, but we haven't obtained a single flash recorded.
We have found an espectrofluormeter and we will start to use it tomorrow.
August 13th
We have mesured the new yeasts in the espectrofluorimeter, with no incident ray, but we have only recorded noise. Then we have realized that we've mesuring at 465 nm, the wave length of the normal coelenterazine, where the one that we are using emits more light, but it does it at 444nm.
Note: We have wake up really early, at 6 a.m. The sunrise in the Cavanilles is weirdly beautiful ^^
We also have recorded a freak advertisement about one of us inventions for the experimental procedure: the Pippete Enlarger (you can watch it in ....)
August 14th
Another sunrise surrounded by yeasts... We have repeated yesterday's experiment at the right wavelenght (444 nm) but we haven't obtain results.
August 17th
We have designed our primers to amplify aequorin gene, the only "physical" BioBrick we're going to do by now. We have also asked for it. Probably, them will arrive in that week.
Carles is in Banyuls (France) making a short course. Angeles and Cristina will entrust with lab work for 10 days.
August 18th
We have made a solution at 50% with our three yeasts strains and glicerol. Later, we have put them in the freezer.
August 19th
We have repeated the experiment again but this time we have changed coelenterazine concentration from 2 to 5 micromolar and the incubation has been longer (8 hours). No results.
New coelenterazine (native) has arrived.
August 20th
Native coelenterazine has been resuspended as the protocol says.
Our primers have arrived and tomorrow we begin with all BioBricks work.
August 21st
We have repited our initial protocol (coelenterazine at 2 micromolar, and incubation about 5,5 hours), but now with native coelenterazine. We have started the experiment at 5.30 a.m. because we needed everything ready at 12.00. Lab work is a really hard thing... ¬¬
If we don't obtain results with the fluorimetre, I commite suicide...
Well, we have prepared three samples:
1. One acording to the original protocol with KOH.
2. One with the CaCl2 1,33M (see our protocol).
3. And, in a desesperate way, we have added KOH and CaCl2 together as a kind of "mixtured-input" (30 microlitres of everyone) to a third sample.
We haven't seen nothing... I'm gonna commite suicide...
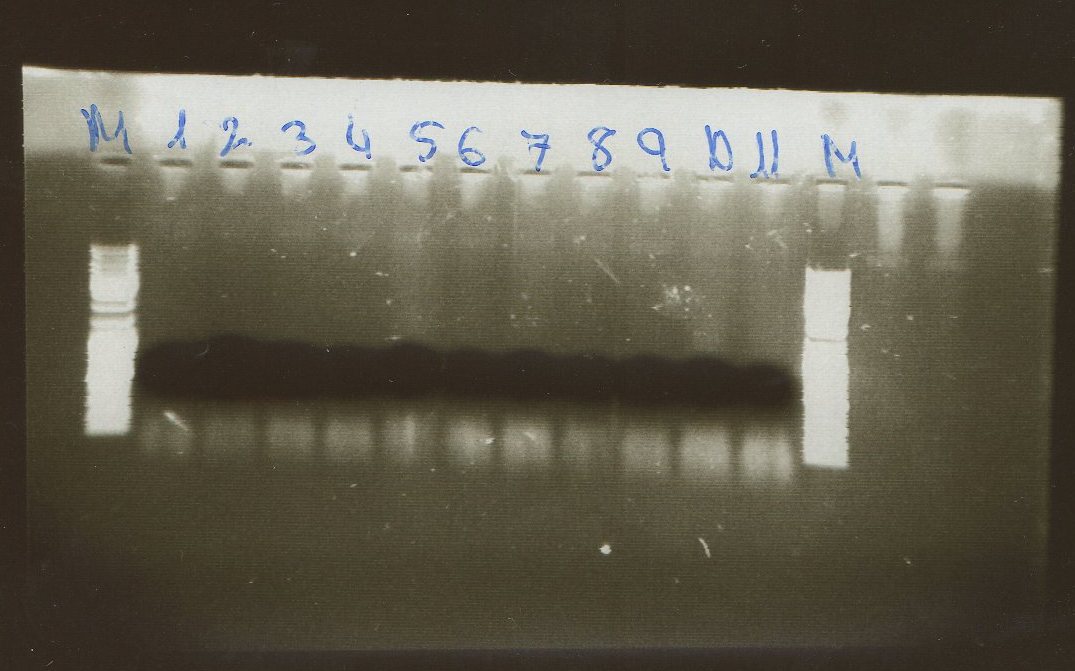
For the other side, we have started our BioBricks work. We have prepare a PCR to amplify aequorin gene. Our samples were DNA from our three yeast strains (WT, Cch1- and Mid1-). We obtein them boiling our strains for 5 minutes. After the typical 30 cycles, we have run an agarose 0,8% gel with the PCR products. We runned a Molecular Weight Marker in two little holes (GeneRuler DNA Ladder Mix #SM0333 ng/0,5micrograms %).
To increase our desesperate state, there were'nt any amplification. Only primer-primers/primer-dimers...
1, 2, 3 = wt (1 microlitre)
4, 5, 6 = Cch1 (1 microlitre)
7, 8, 9 = Mid1 (1 microlitre)
10, 11 = Negative Control
Nevertheless, our PCR conditions were:
Components:
- 38,5 microlitres of H2O
- 5 microlitres of Buffer 10X (it contains Mg2+)
- 1 microlitre of our forward primer
- 2 microlitres of dNTP's 5 mM
- 1 microlitre of our reverse primer
- 1,5 microlitres of Taq 10 mM
- 1 microlitre of our DNA
Programme:
After 3 minutes at 94 ºC as a previous step, every of our 30 cycles has these three steps:
50s at 94 ºC
1min at 62 ºC
1min and 40s at 72 ºC
August 24th
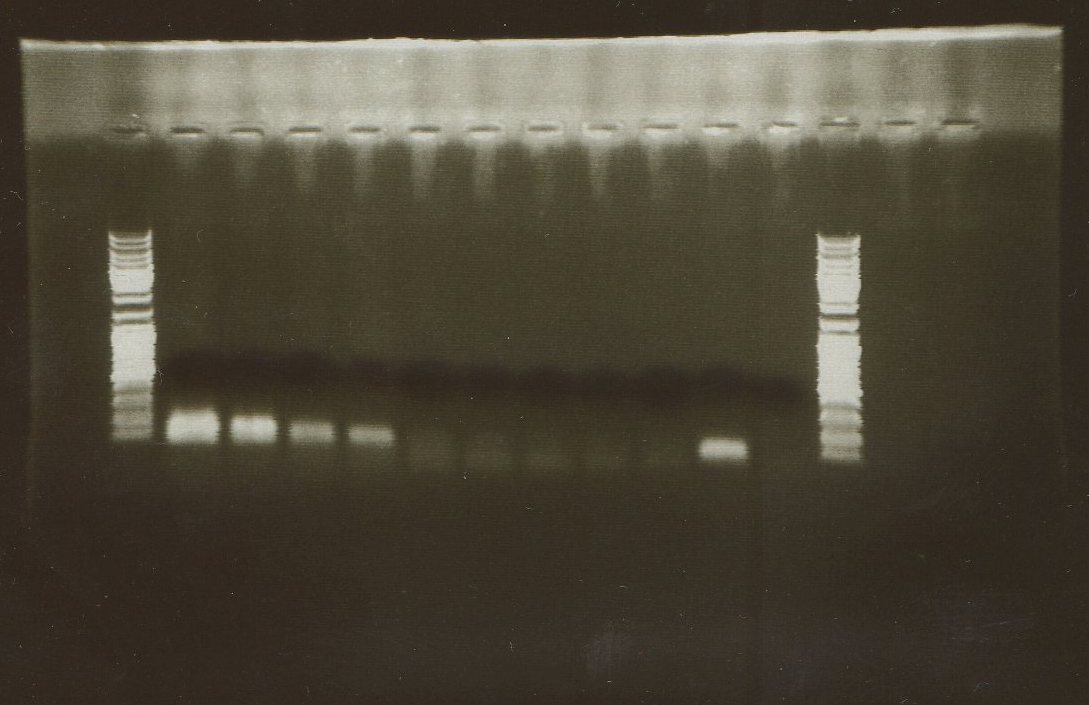
We have repited the same PCR, but we changed some points:
Programme:
After 3 minutes at 94 ºC as a previous step, every of our 35 cycles has these trhee steps:
- 50 at 94 ºC
- 1' at 52 ºC
- 1' 40 at 72 ºC
We have added one possitive control, too.
There was no amplification, even not in the possitive control. We have only seen bands of primerprimers and primer-dimers.
1, 2, 3 = wt (1 microlitre)
4, 5, 6 = Cch1 (1 microlitre)
7, 8, 9 = Mid1 (1 microlitre)
10 = Possitive Control
11 = Negative Control
(We used the same MWM)
We suspect about the termocycler. Tomorrow we will change to another one.
 "
"